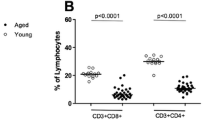
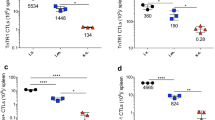
Abstract
Peptide vaccines represent an attractive alternative to conventional anti-tumor therapies, but have not yet achieved significant clinical efficacy with commonly used formulations. Combination of short antigenic peptides, synthetic melanin and TLR9 agonist (Toll-like receptor 9, CpG-28) was reported as highly efficient to trigger strong CD8 + T-cell responses. We compared this vaccine approach to the standard adjuvant formulation that combines the incomplete Freund’s adjuvant (IFA) and CpG-28, using either an ovalbumin epitope (pOVA30) or a spontaneously occurring tumor neoepitope (mAdpgk).
Melanin-based vaccine induced significantly higher cytotoxic T lymphocytes (CTL) responses than IFA-based vaccine in both pOVA30- and mAdpgk-targeted vaccines. The anti-tumor efficacy of melanin-based vaccine was further assessed in mice, grafted either with E.G7-OVA cells (E.G7 cells transfected with ovalbumin) or with MC38 cells that spontaneously express the mAdpgk neoepitope. Melanin-based vaccine induced a major inhibition of E.G7-OVA tumor growth when compared to IFA-based vaccine (p < 0.001), but tumors eventually relapsed from day 24. In the MC38 tumor model, no significant inhibition of tumor growth was observed. In both cases, tumor escape appeared related to the loss of antigen presentation by tumor cells (loss of ovalbumin expression in E.G7-OVA model; poor presentation of mAdpgk in MC38 model), although the CTL responses displayed an effector memory phenotype, a high cytolytic potential and low programmed cell death-1 (PD1) expression.
In conclusion, synthetic melanin can be efficiently used as an adjuvant to enhance T-cells response against subunit vaccine antigens and compared favorably to the classic combination of IFA and TLR9 agonist in mice.




Similar content being viewed by others
Abbreviations
- ACK:
-
Ammonium–chloride–potassium
- APC:
-
Antigen-presenting cell
- CCR7:
-
C–C chemokine receptor type 7
- CTL:
-
Cytotoxic T lymphocyte
- CTLA4:
-
Cytotoxic T-lymphocyte-associated protein 4
- DMEM:
-
Dulbecco modified Eagle medium
- EDTA:
-
Ethylenediaminetetraacetic acid
- ICI:
-
Immune check-point inhibitor
- IFA:
-
Incomplete Freund’s adjuvant
- IFNγ:
-
Interferon gamma
- LAG3:
-
Lymphocyte-activation gene 3
- MHC:
-
Major histocompatibility complex
- PBS:
-
Phosphate-buffered saline
- PD1:
-
Programmed cell death 1
- qRT-PCR:
-
Quantitative real-time reverse transcriptase–polymerase chain reaction
- RNA:
-
Ribonucleic acid
- SEM:
-
Standard error of the mean
- SFC:
-
Spot-forming cell
- TAA:
-
Tumor-associated antigens
- TIL:
-
Tumor-infiltrating lymphocyte
- TIM3:
-
T-cell immunoglobulin mucin-3
- TLR9:
-
Toll-like receptor 9
- Treg:
-
Regulatory T cells
References
Carpentier AF, Tran T, Sejalon F, Geinguenaud F, Tartour E, Motte L, Banissi C. The adjuvant effect of melanin is superior to incomplete Freund adjuvant in a tumor subunit vaccine model, Eur J Cancer, March 2018 Volume 92, Supplement 1, p S2–S3 [Abstract A4]
Schumacher TN, Schreiber RD (2015) Neoantigens in cancer immunotherapy. Science 348(6230):69–74
Kumai T, Kobayashi H, Harabuchi Y, Celis E (2017) Peptide vaccines in cancer-old concept revisited. Curr Opin Immunol 45:1–7
Speiser DE, Liénard D, Rufer N, Rubio-Godoy V, Rimoldi D, Lejeune F, Krieg AM, Cerottini JC, Romero P (2005) Rapid and strong human CD8+ T cell responses to vaccination with peptide, IFA, and CpG oligodeoxynucleotide 7909. J Clin Invest 115(3):739–746
Obeid J, Hu Y, Slingluff CL Jr (2015) Vaccines, adjuvants, and dendritic cell activators–current status and future challenges. Semin Oncol 42:549–561
Melssen MM, Petroni GR, Chianese-Bullock KA, Wages NA, Grosh WW, Varhegyi N, Smolkin ME, Smith KT, Galeassi NV, Deacon DH, Gaughan EM, Slingluff CL Jr (2019) A multipeptide vaccine plus toll-like receptor agonists LPS or polyICLC in combination with incomplete Freund's adjuvant in melanoma patients. J Immunother Cancer 7(1):163
Souleimanian NE, Tosello V, Bhardwaj N, Adams S, O'Neill D, Pavlick A, Escalon JB, Cruz CM, Angiulli A, Angiulli F, Mears G, Vogel SM, Pan L, Jungbluth AA, Hoffmann EW, Venhaus R, Ritter G, Old LJ, Ayyoub M (2007) Vaccination with NY-ESO-1 protein and CpG in Montanide induces integrated antibody/Th1 responses and CD8 T cells through cross-priming. Proc Natl Acad Sci U S A 104(21):8947–8952
Hailemichael Y, Dai Z, Jaffarzad N, Ye Y, Medina MA, Huang XF, Dorta-Estremera SM, Greeley NR, Nitti G, Peng W, Liu C, Lou Y, Wang Z, Ma W, Rabinovicj B, Sowell RT, Schluns KS, Davis RE, Hwu P, Overwijk WW (2013) Persistent antigen at vaccination sites induces tumor-specific CD8+ T cell sequestration, dysfunction and deletion. Nat Med 19(4):465–472
Bijker MS, van den Eeden SJ, Franken KL, Melief CJ, Offringa R, van der Burg SH (2007) CD8+ CTL priming by exact peptide epitopes in incomplete Freund’s adjuvant induces a vanishing CTL response, whereas long peptides induce sustained CTL reactivity. J Immunol 179(8):5033–5040
Carpentier AF, Geinguenaud F, Tran T, Sejalon F, Martin A, Motte L, Tartour E, Banissi C (2017) Synthetic melanin bound to subunit vaccine antigens significantly enhances CD8+ T-cell responses. PLoS ONE 12(7):e0181403
Bachmann MF, Jennings GT (2010) Vaccine delivery: a matter of size, geometry, kinetics and molecular patterns. Nat Rev Immunol 10(11):787–796
Rötzschke O, Falk K, Stevanović JG, Walden P, Rammensee HG (1991) Exact prediction of a natural T cell epitope. Eur J Immunol 21(11):2891–2894
Ahonen CL, Doxsee CL, McGurran SM, Riter TR, Wade WF, Barth RJ, Vasilakos JP, Noelle RJ, Kedl RM (2004) Combined TLR and CD40 triggering induces potent CD8+ T cell expansion with variable dependence on type I IFN. J Exp Med 199:775–784
Yadav M, Jhunjhunwala S, Phung QT, Lupardus P, Tanguay J, Bumbaca S, Franci C, Cheung TK, Fritsche J, Weinschenk T, Modrusan Z, Mellman I, Lill JR, Delamarre L (2014) Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature 515:572–576
Lorenzi S, Mattei F, Sistigu A, Bracci A, Spadaro F, Sanchez M, Belardelli F, Gabriele L, Schiavoni G (2011) Type I IFNs control antigen retention and survival of CD8α+ dendritic cells after uptake of tumor apoptotic cells leading to cross-priming. J Immunol 186(9):5142–5150
Maubant S, Banissi C, Beck S, Chauvat A, Carpentier AF (2011) Adjuvant properties of cytosine-phosphateguanosine oligodeoxynucleotide in combination with various polycations in an ovalbumin-vaccine model. Nucleic Acid Ther 21:231–240
Carpentier A, Metellus P, Ursu R, Zohar S, Lafitte F, Barrie´ M, Meng Y, Richard M, Parizot C, Laigle-Donadey F, Gorochov G, Psimaras D, Sanson M, Tibi A, Chinot O, Carpentier AF (2010) Intracerebral administration of CpG oligonucleotide for patients with recurrent glioblastoma, a phase II study. Neuro Oncol 12:401
Carpentier AF, Chen L, Maltonti F, Delattre JY (1999) Oligodeoxynucleotides containing CpG motifs can induce rejection of a neuroblastoma in mice. Cancer Res 59(21):5429–5432
Tran T, Diniz MO, Dransart E, Gey A, Merillon N, Lone YC, Godefroy S, Sibley C, Ferreira LC, Medioni J, Oudard S, Johannes L, Tartour E (2016) A Therapeutic her2/neu vaccine targeting dendritic cells preferentially inhibits the growth of low her2/neu-expressing tumor in HLA-A2 transgenic mice. Clin Cancer Res 22(16):4133–4144
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Sahin U, Tureci O (2018) Personalized vaccines for cancer immunotherapy. Science 359(6382):1355–1360
Makinen SR, Zhu Q, Davis HL, Weeratna RD (2016) CpG-mediated augmentation of CD8+ T-cell responses in mice is attenuated by a water-in-oil emulsion (Montanide ISA-51) but enhanced by an oil-in-water emulsion (IDRI SE). Int Immunol 28(9):453–461
Kuball J, de Boer K, Wagner E, Wattad M, Antunes E, Weeratna RD, Vicari AP, Lotz C, van Dorp S, Hol S, Greenberg PD, Heit W, Davis HL, Theobald M (2011) Pitfalls of vaccinations with WT1-, proteinase3- and MUC1-derived peptides in combination with montanideisa51 and CpG7909. Cancer Immunol Immunother 60(2):161–172
Hailemichael Y, Dai Z, Jaffarzad N, Ye Y, Medina MA, Huang XF, Dorta-Estremera SM, Greeley NR, Nitti G, Peng W, Liu C, Lou Y, Wang Z, Ma W, Rabinovich B, Sowell RT, Schluns KS, Davis RE, Hwu P, Overwijk WW (2013) Persistent antigen at vaccination sites induces tumor-specific CD8+ T cell sequestration, dysfunction and deletion. Nat Med 19(4):465–472
Hemmi H, Yoshino M, Yamazaki H, Naito M, Iyoda T, Omatsu Y, Shimoyama S, Letterio JJ, Nakabayashi T, Tagaya H, Yamane T, Ogawa M, Nishikawa S, Ryoke K, Inaba K, Hayashi S, Kunisada T (2001) Skin antigens in the steady state are trafficked to regional lymph nodes by transforming growth factor-beta1-dependent cells. Int Immunol 13(5):695–704
Yoshino M, Yamazaki H, Shultz LD, Hayashi S (2006) Constant rate of steady-state self-antigen trafficking from skin to regional lymph nodes. Int Immunol. 18(11):1541–1548
Wang W, Liu Z, Zhou X, Guo Z, Zhang J, Zhu P, Yao S, Zhu M (2019) Ferritin nanoparticle-based SpyTag/SpyCatcher-enabled click vaccine for tumor immunotherapy. Nanomedicine 16:69–78
Heße C, Kollenda S, Rotan O, Pastille E, Adamczyk A, Wenzek C, Hansen W, Epple M, Buer J, Westendorf AM, Knuschke T (2019) A tumor-peptide-based nanoparticle vaccine elicits efficient tumor growth control in antitumor immunotherapy. Mol Cancer Ther 18(6):1069–1080
Andrews LP, Marciscano AE, Drake CG, Vignali DA (2017) LAG3 (CD223) as a cancer immunotherapy target. Immunol Rev 276(1):80–96
Tu L, Guan R, Yang H, Zhou Y, Hong W, Ma L, Zhao G, Yu M (2019) Assessment of the expression of the immune checkpoint molecules PD-1, CTLA4, TIM-3 and LAG-3 across different cancers in relation to treatment response, tumor-infiltrating immune cells and survival. Int J Cancer 147(2):423–439
Zelba H, Bedke J, Hennenlotter J, Mostböck S, Zettl M, Zichner T, Chandran A, Stenzl A, Rammensee HG, Gouttefangeas C (2019) PD-1 and LAG-3 dominate checkpoint receptor-mediated T-cell inhibition in renal cell carcinoma. Cancer Immunol Res 7(11):1891–1899
Matsushita H, Vesely MD, Koboldt DC, Rickert CG, Uppaluri R, Magrini VJ, Arthur CD, White JM, Chen Y-S, Shea LK, Hundal J, Wendl MC, Demeter R, Wylie T, Allison JP, Smyth MJ, Old LJ, Mardis ER, Schreiber RD (2012) Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 482(7385):400–404
Dharmaraj N, Piotrowski SL, Huang C, Newton JM, Golfman LS, Hanoteau A, Koshy ST, Li AW, Pulikkathara MX, Zhang B, Burks JK, Mooney DJ, Lei YL, Sikora AG, Young S (2019) Anti-tumor immunity induced by ectopic expression of viral antigens is transient and limited by immune escape. Oncoimmunology 8(4):e1568809
Hos B, Camps MGM, van den Bulk J, Tondinin E, van den Ende T, Ruano D, Franken K, Janssen GMC, de Ru AH, Filippov DV, Arens R, van Veelen PA, de Miranda NFCC, Ossendorp F (2019) Identification of a neo-epitope dominating endogenous CD8 T cell responses to MC-38 colorectal cancer. Oncoimmunology. https://doi.org/10.1080/2162402X.2019.1673125
Acknowledgments
The authors thank the Association Oligocyte and the Association pour le development des neurosciences a Avicenne (ADNA) for supporting our research.
Funding
No funding sources were used for this report.
Author information
Authors and Affiliations
Contributions
SC, CT, ET and AFC contributed to the study concept and design of experiments. SC, CB, MSR and TT performed the experiments. CB and TT helped in methodology and software analysis. SC and CB were involved in data analysis and interpretation of results. SC and AFC wrote the preliminary version of the paper. All authors participated in critical review and revision of the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The AP/HP (Assistance Publique de Hopitaux de Paris) filed a provisional patent application on this method. AF Carpentier & C Banissi are listed as inventors. AF Carpentier holds shares in Altevax inc. and is consultant for BMS. The authors declare that there are no other conflicts of interest.
Ethical approval and ethical standards.
Experiments were conducted on female C57BL/6 mice. All animal experiments were approved by the ethics committee of Paris Descartes University (Project APAFIS #5337 N° 2016021517305775) and performed in accordance with European Union guidelines for animal experiments.
Human and animal rights
Animal source: Female C57BL/6 aged 5 to 6 week old were purchased from Janvier Labs (Le Genest-Saint-Isle, France) and kept under specific-pathogen-free conditions.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published as a poster abstract at the 5th Immunotherapy of Cancer Conference: [1]
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cuzzubbo, S., Banissi, C., Rouchon, M.S. et al. The adjuvant effect of melanin is superior to incomplete Freund’s adjuvant in subunit/peptide vaccines in mice. Cancer Immunol Immunother 69, 2501–2512 (2020). https://doi.org/10.1007/s00262-020-02631-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02631-7