Abstract
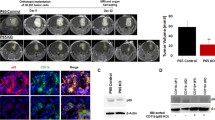
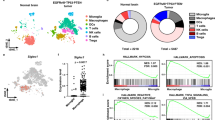
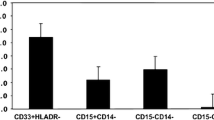
High-grade gliomas harbor abundant myeloid cells that suppress anti-tumor immunity and support tumor growth. Targeting transcription factors, such as NF-κB p50, that mediate suppressive myeloid M2 polarization may prove therapeutic. GL261-Luc glioblastoma cells were inoculated into wild-type and p50−/− mice, followed by analysis of tumor growth, survival, tumor myeloid cells, and T cells. The absence of host p50 slows tumor growth and enables regression in 30% of recipients, leading to prolonged survival. Tumors developing in p50−/− mice possess a greater concentration of tumor-infiltrating myeloid cells (TIMs) than those in wild-type mice. TIMs are predominantly F4/80hi macrophages which, along with tumor-associated microglia, express increased pro-inflammatory M1 and reduced immune-suppressive M2 markers. In p50−/− mice, total tumor CD4 T cells are threefold more abundant, whereas CD8 T-cell numbers are unchanged, and both produce increased IFNγ and Granzyme B. Naïve splenic p50−/− CD8 T cells manifest increased activation, whereas naïve p50−/− and WT CD4 T cells show similar Th1, Th2, and Th17 polarization. Antibody targeting CD4, but not CD8, fully obviates the p50−/− survival advantage. Combined CD4 and CD8 T-cell depletion reverses myeloid M2 polarization in wild-type hosts, without affecting myeloid M1 polarization in p50−/− hosts. Finally, gliomas grow similarly in p50(f/f) and p50(f/f);Lysozyme-Cre mice, the latter having reduced p50 specifically in myeloid cells and tumor microglia. Thus, high-grade glioma T cells play a key role in directing M2 polarization of tumor myeloid cells, and reducing NF-κB p50 in both tumor myeloid cells and T cells may contribute to glioma therapy.






Similar content being viewed by others
Abbreviations
- B6:
-
C57BL/6
- BMDM:
-
Bone-marrow-derived macrophage
- FC:
-
Flow cytometry
- GBM:
-
Glioblastoma
- IVIS:
-
In vivo imaging system
- MR:
-
Mannose receptor
- p50:
-
NF-κB p50
- PBS:
-
Phosphate-buffered saline
- qRT-PCR:
-
Quantitative real-time PCR
- TAM:
-
Tumor-associated macrophage
- TIM:
-
Tumor-infiltrating myeloid cell
- WT:
-
Wild type
References
Kennedy BC, Showers CR, Anderson DE, Anderson L, Canoll P, Bruce JN, Anderson SC (2013) Tumor-associated macrophages in glioma: friend or foe? J Oncol 2013:486912
Gabrusiewicz K, Ellert-Miklaszewska A, Lipko M, Sielska M, Frankowska M, Kaminska B (2011) Characteristics of the alternative phenotype of microglia/macrophages and its modulation in experimental gliomas. PLoS One 6:e23902
Ginhoux F, Greter M, Leboeuf M, Nandi S, See P, Gokhan S, Mehler MF, Conway SJ, Ng LG, Stanley ER, Samokhvalov IM, Merad M (2010) Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330:841–845
Shand FH, Ueha S, Otsuji M, Koid SS, Shichino S, Tsukui T, Kosugi-Kanaya M, Abe J, Tomura M, Ziogas J, Matsushima K (2014) Tracking intertissue migration reveals the origins of tumor-infiltrating monocytes. Proc Natl Acad Sci USA 111:7771–7776
Sica A, Bronte V (2007) Altered macrophage differentiation and immune dysfunction in tumor development. J Clin Invest 117:1155–1166
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8:958
Gabrilovich DI, Ostrand-Rosenberg S, Bronte V (2012) Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 12:253–268
Franklin RA, Liao W, Sarkar A, Kim MV, Bivona MR, Liu K, Pamer EG, Li MO (2014) The cellular and molecular origin of tumor-associated macrophages. Science 344:921–925
Noy R, Pollard JW (2014) Tumor-associated macrophages: from mechanisms to therapy. Immunity 41:49–61
Li W, Graeber MB (2012) The molecular profile of microglia under the influence of glioma. Neuro Oncol 14:958–978
Pyonteck SM, Akkari L, Schuhmacher AJ, Bowman RL, Sevenich L, Quail DF, Olson OC, Quick ML, Huse JT, Teijeiro V, Setty M, Leslie CS, Oei Y, Pedraza A, Zhang J, Brennan CW, Sutton JC, Holland EC, Daniel D, Joyce JA (2013) CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nature Med 19:1264–1272
Sielska M, Przanowski P, Wylot B, Gabrusiewicz K, Maleszewska M, Kijewska M, Zawadzka M, Kucharska J, Vinnakota K, Kettenmann H, Kotulska K, Grajkowska W, Kaminska B (2013) Distinct roles of CSF family cytokines in macrophage infiltration and activation in glioma progression and injury response. J Pathol 230:310–321
Quail DF, Bowman RL, Akkari L, Quick ML, Schuhmacher AJ, Huse JT, Holland EC, Sutton JC, Joyce JA (2016) The tumor microenvironment underlies acquired resistance to CSF-1R inhibition in gliomas. Science 352:aad3018
Bohuslav J, Kravchenko VV, Parry GC, Erlich JH, Gerondakis S, Mackman N, Ulevitch RJ (1998) Regulation of an essential innate immune response by the p50 subunit of NF-κB. J Clin Invest 102:1645–1652
Saccani A, Schioppa T, Porta C, Biswas SK, Nebuloni M, Vago L, Bottazzi B, Colombo MP, Mantovani A, Sica A (2006) p50 nuclear factor-kB overexpression in tumor-associated macrophages inhibits M1 inflammatory responses and antitumor resistance. Cancer Res 66:11432–11440
Porta C, Rimoldi M, Raes G, Brys L, Ghezzi P, Di Liberto D, Dieli F, Ghisletti S, Natoli G, De Baetselier P, Mantovani A, Sica A (2009) Tolerance and M2 (alternative) macrophage polarization are related processes orchestrated by p50 nuclear factor kB. Proc Natl Acad Sci USA 106:14978–14983
Kono Y, Kawakami S, Higuchi Y, Yamashita F, Hashida M (2014) In vitro evaluation of inhibitory effect of nuclear factor-κB activity by small interfering RNA on pro-tumor characteristics of M2-like macrophages. Biol Pharm Bull 37:137–144
Rackov G, Hernandez-Jimenez E, Shokri R, Carmona-Rodríguez L, Mañes S, Álvarez-Mon M, López-Collazo E, Martínez-A C, Balomenos D (2016) p21 mediates macrophage reprogramming through regulation of p50–p50 NF-κB and IFN-β. J Clin Invest 126:3089–3103
Ruffell D, Mourkioti F, Gambardella A, Kirstetter P, Lopez RG, Rosenthal N, Nerlov C (2009) A CREB-C/EBPβ cascade induces M2 macrophage-specific gene expression and promotes muscle injury repair. Proc Natl Acad Sci USA 106:17475–17480
Marigo I, Bosio E, Solito S, Mesa C, Fernandez A, Dolcetti L, Ugel S, Sonda N, Bicciato S, Falisi E, Calabrese F, Basso G, Zanovello P, Cozzi E, Mandruzzato S, Bronte V (2010) Tumor-induced tolerance and immune suppression depend on the C/EBPβ transcription factor. Immunity 32:790–802
Feinberg MW, Wara AK, Cao Z, Lebedeva MA, Rosenbauer F, Iwasaki H, Hirai H, Katz JP, Haspel RL, Gray S, Akashi K, Segre J, Kaestner KH, Tenen DG, Jain MK (2007) The Kruppel-like factor KLF4 is a critical regulator of monocyte differentiation. EMBO J 26:4138–4148
Tugal D, Liao X, Jain MK (2013) Transcriptional control of macrophage polarization. Arterioscler Thromb Vasc Biol 33:1135–1144
Strauss L, Sangaletti S, Consonni FM, Szebeni G, Morlacchi S, Totaro MG, Porta C, Anselmo A, Tartari S, Doni A, Zitelli F, Tripodo C, Colombo MP, Sica A (2015) RORC1 regulates tumor-promoting “emergency” granulo-monocytopoiesis. Cancer Cell 28:253–269
Newcomb EW, Zagzag D (2009) The murine GL261 glioma experimental model to assess novel brain tumor treatments. In: Van Meir EG (ed) CNS cancer, models, markers, prognostic factors, targets, and therapeutic approaches. Humana Press, New York, pp 228–241
Zeng J, See AP, Phallen J, Jackson CM, Belcaid Z, Ruzevick J, Durham N, Meyer C, Harris TJ, Albesiano E, Pradilla G, Ford E, Wong J, Hammers HJ, Mathios D, Tyler B, Brem H, Tran PT, Pardoll D, Drake CG, Lim M (2013) Anti-PD-1 blockade and stereotactic radiation produces long term survivors in mice with intracranial glioma. Int J Radiat Oncol Biol Phys 86:343–349
Flaherty S, Reynolds JM (2015) Mouse naïve CD4+ T cell isolation and in vitro differentiation into T cell subsets. J Vis Exp 98:52739
Badie B, Schartner JM (2000) Flow cytometric characterization of tumor-associated macrophages in experimental gliomas. Neurosurgery 46:957–961
Movahedi K, Laoui D, Gysemans C, Baeten M, Stangé G, Van den Bossche J, Mack M, Pipeleers D, In’t Veld P, De Baetselier P, Van Ginderachter JA (2010) Different tumor microenvironments contain functionally distinct subsets of macrophages derived from Ly6C(high) monocytes. Cancer Res 70:5728–5739
Georgoudaki AM, Prokopec KE, Boura VF, Hellqvist E, Sohn S, Östling J, Dahan R, Harris RA, Rantalainen M, Klevebring D, Sund M, Brage SE, Fuxe J, Rolny C, Li F, Ravetch JV, Karlsson MC (2016) Reprogramming tumor-associated macrophages by antibody targeting inhibits cancer progression and metastasis. Cell Rep 15:2000–2011
Muller A, Brandenburg S, Turkowski K, Muller S, Vajkoczy (2015) Resident microglia, and not peripheral macrophages, are the main source of brain tumor mononuclear cells. Int J Cancer 137:278–288
Marigo I, Zilio S, Desantis G, Mlecnik B, Agnellini AH, Ugel S, Sasso MS, Qualls JE, Kratochvill F, Zanovello P, Molon B, Ries CH, Runza V, Hoves S, Bilocq AM, Bindea G, Mazza EM, Bicciato S, Galon J, Murray PJ, Bronte V (2016) T cell cancer therapy requires CD40-40L activation of tumor necrosis factor and inducible nitric-oxide-synthase-producing dendritic cells. Cancer Cell 30:377–390
Kokubu Y, Tabu K, Muramatsu N, Wang W, Murota Y, Nobuhisa I, Jinushi M, Taga T (2016) Induction of protumoral CD11c(high) macrophages by glioma cancer stem cells through GM-CSF. Genes Cells 21:241–251
Abram CL, Roberge GL, Hu Y, Lowell CA (2014) Comparative analysis of the efficiency and specificity of myeloid-Cre deleting strains using ROSA-EYFP reporter mice. J Immunol Methods 408:89–100
Johanns TM, Ward JP, Miller CA, Wilson C, Kobayashi DK, Bender D, Fu Y, Alexandrov A, Mardis ER, Artyomov MN, Schreiber RD, Dunn GP (2016) Endogenous neoantigen-specific CD8 T cells identified in two glioblastoma models using a cancer immunogenomics approach. Cancer Immunol Res 4:1007–1015
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived-suppressor cells as regulators of the immune system. Nat Rev Immunol 9:162–174
Kaneda MM, Messer KS, Ralainirina N, Li H, Leem CJ, Gorjestani S, Woo G, Nguyen AV, Figueiredo CC, Foubert P, Schmid MC, Pink M, Winkler DG, Rausch M, Palombella VJ, Kutok J, McGovern K, Frazer KA, Wu X, Karin M, Sasik R, Cohen EE, Varner JA (2016) PI3Kγ is a molecular switch that controls immune suppression. Nature 539:437–442
Belcaid Z, Phallen JA, Zeng J, See AP, Mathios D, Gottschalk C, Nicholas S, Kellett M, Ruzevick J, Jackson C, Albesiano E, Durham NM, Ye X, Tran PT, Tyler B, Wong JW, Brem H, Pardoll DM, Drake CG, Lim M (2014) Focal radiation therapy combined with 4-1BB activation and CTLA-4 blockade yields long-term survival and a protective antigen-specific memory response in a murine glioma model. PLoS One 9:e101764
Dorand RD, Nthale J, Myers JT, Barkauskas DS, Avril S, Chirieleison SM, Pareek TK, Abbott DW, Stearns DS, Letterio JJ, Huang AY, Petrosiute A (2016) Cdk5 disruption attenuates tumor PD-L1 expression and promotes antitumor immunity. Science 353:399–403
Ellsworth S, Balmanoukian A, Kos F, Nirschl CJ, Nirschl TR, Grossman SA, Luznik L, Drake CG (2014) Sustained CD4+ T cell-driven lymphopenia without a compensatory IL-7/IL-15 response among high-grade glioma patients treated with radiation and temozolomide. Oncoimmunology 3:e27357
Grossman SA, Ye X, Lesser G, Sloan A, Carraway H, Desideri S, Piantadosi S, NABTT CNS Consortium (2011) Immunosuppression in patients with high-grade gliomas treated with radiation and temozolomide. Clin Cancer Res 17:5473–5480
Funding
This study was supported by grants from Alex’s Lemonade Stand Foundation, Hyundai Hope on Wheels, the Allegheny Health Network-Johns Hopkins Cancer Research Fund, the National Institutes of Health (T32 CA60441 and P30 CA006973), and by the Giant Food Children’s Cancer Research Fund.
Author information
Authors and Affiliations
Contributions
TB, AM, RS, DJB, and SH-B conducted experiments. CGD, ML, and ADF guided the conduct of experiments. TB and ADF wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval and ethical standards
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Animal source
WT C57BL/6 (B6) mice were obtained from Charles River Laboratories, Nfkb1(p50)−/−, Lys-Cre, and ROSA26-FLPo mice from Jackson Laboratory (6097, 4781, 12930), and p50(flacZ/+) mice from the Knockout Mouse Project (CSD29053).
Cell line authentication
GL261-Luc cells were obtained from Perkin Elmer, who had authenticated it as C57BL/6 in origin by mouse STR analysis.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Barberi, T., Martin, A., Suresh, R. et al. Absence of host NF-κB p50 induces murine glioblastoma tumor regression, increases survival, and decreases T-cell induction of tumor-associated macrophage M2 polarization. Cancer Immunol Immunother 67, 1491–1503 (2018). https://doi.org/10.1007/s00262-018-2184-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-018-2184-2