Abstract
Background
Hepatic immunity, normally protective against neoplasia, is subverted in colorectal liver metastasis (CRLM). Here, we compare the inflammatory microenvironment of CRLM-bearing liver tissue to donor liver.
Methods
Twenty-five patients undergoing resection for CRLM were recruited, 13 of whom developed intrahepatic recurrence within 18 months. Biopsies were obtained from tumour and normal liver tissue adjacent to and distal from, the tumour. Donor liver biopsies were obtained during transplantation. Biopsies were cultured and conditioned media (CM) screened for 102 inflammatory mediators. Twelve of these were validated by Luminex assay. Transwell assays measured cancer cell chemotaxis. Polymorphonuclear leukocytes (PMN) and lymphocytes were quantified in H&E sections.
Results
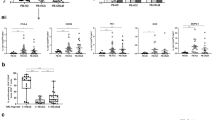
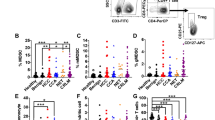
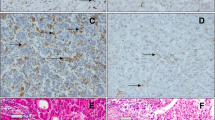
Fewer periportal tissue-resident PMN were present in metastatic liver compared to donor liver. Patients with the fewest PMN in liver tissue distal to their tumour had a shorter time to intrahepatic recurrence (P < 0.001). IL-6, CXCL1, CXCL5, G-CSF, GM-CSF, VEGF, LIF, and CCL3 were higher in liver-bearing CRLM compared to donor tissue. Consequently, cancer cells migrated equally towards CM of all regions of metastatic liver but not towards donor liver CM.
Conclusions
The local inflammatory environment may affect both immune cell infiltration and cancer cell migration contributing to recurrence following resection for CRLM.




Similar content being viewed by others
Abbreviations
- CCL:
-
Chemokine (C–C motif) ligand
- CM:
-
Conditioned media
- CRC:
-
Colorectal cancer,
- CRLM:
-
Colorectal liver metastases,
- CXCL:
-
Chemokine (C-X-C motif) ligand
- DPP4:
-
Dipeptidyl peptidase-4
- HPF:
-
High-powered field
- LIF:
-
Leukemia inhibitory factor
- MIF:
-
Macrophage migration inhibitory factor
- NLR:
-
Neutrophil-to-lymphocyte ratio
- PMN:
-
Polymorphonuclear leukocytes
- VEGF:
-
Vascular endothelial growth factor
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359–E386. https://doi.org/10.1002/ijc.29210
Sethi N, Kang Y (2011) Unravelling the complexity of metastasis—molecular understanding and targeted therapies. Nat Rev Cancer 11(10):735–748. https://doi.org/10.1038/nrc3125
Disibio G, French SW (2008) Metastatic patterns of cancers: results from a large autopsy study. Arch Pathol Lab Med 132(6):931-9. https://doi.org/10.1043/1543-2165(2008)132[931:MPOCRF]2.0.CO;2
Nathan H, de Jong MC, Pulitano C, Ribero D, Strub J, Mentha G, Gigot JF, Schulick RD, Choti MA, Aldrighetti L, Capussotti L, Pawlik TM (2010) Conditional survival after surgical resection of colorectal liver metastasis: an international multi-institutional analysis of 949 patients. J Am Coll Surg 210(5):755–764. https://doi.org/10.1016/j.jamcollsurg.2009.12.041
Hugen N, van de Velde CJ, de Wilt JH, Nagtegaal ID (2014) Metastatic pattern in colorectal cancer is strongly influenced by histological subtype. Ann Oncol 25(3):651–657. https://doi.org/10.1093/annonc/mdt591
Wurster EF, Tenckhoff S, Probst P, Jensen K, Dölger E, Knebel P, Diener MK, Büchler MW, Ulrich A (2017) A systematic review and meta-analysis of the utility of repeated versus single hepatic resection for colorectal cancer liver metastases. HPB (Oxford) 19(6):491–497. https://doi.org/10.1016/j.hpb.2017.02.440
Saiura A, Yamamoto J, Koga R, Takahashi Y, Takahashi M, Inoue Y, Ono Y, Kokudo N (2014) Favorable outcome after repeat resection for colorectal liver metastases. Ann Surg Oncol 21(13):4293–4299. https://doi.org/10.1245/s10434-014-3863-7
Mellman I, Coukos G, Dranoff G (2011) Cancer immunotherapy comes of age. Nature 480(7378):480–489. https://doi.org/10.1038/nature10673
Chen ZY, Raghav K, Lieu CH, Jiang ZQ, Eng C, Vauthey JN, Chang GJ, Qiao W, Morris J, Hong D, Hoff P, Tran H, Menter DG, Heymach J, Overman M, Kopetz S (2015) Cytokine profile and prognostic significance of high neutrophil-lymphocyte ratio in colorectal cancer. Br J Cancer 112(6):1088–1097. https://doi.org/10.1038/bjc.2015.61
Robinson MW, Harmon C, O’Farrelly C (2016) Liver immunology and its role in inflammation and homeostasis. Cell Mol Immunol 13(3):267–276. https://doi.org/10.1038/cmi.2016.3
Albillos A, Lario M, Álvarez-Mon M (2014) Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J Hepatol 61(6):1385–1396. https://doi.org/10.1016/j.jhep.2014.08.010
Gajewski TF, Schreiber H, Fu YX (2013) Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol 14(10):1014–1022. https://doi.org/10.1038/ni.2703
Anders HJ, Romagnani P, Mantovani A (2014) Pathomechanisms: homeostatic chemokines in health, tissue regeneration, and progressive diseases. Trends Mol Med 20(3):154–165. https://doi.org/10.1016/j.molmed.2013.12.002
Eggert T, Wolter K, Ji J, Ma C, Yevsa T, Klotz S, Medina-Echeverz J, Longerich T, Forgues M, Reisinger F, Heikenwalder M, Wang XW, Zender L, Greten TF (2016) Distinct Functions of senescence-associated immune responses in liver tumor surveillance and tumor progression. Cancer Cell 30(4):533–547. https://doi.org/10.1016/j.ccell.2016.09.003
Kelly AM, Golden-Mason L, McEntee G, Traynor O, Doherty DG, Hegarty JE, O’Farrelly C (2004) Interleukin 12 (IL-12) is increased in tumour bearing human liver and expands CD8(+) and CD56(+) T cells in vitro but not in vivo. Cytokine 25(6):273–282. https://doi.org/10.1016/j.cyto.2003.11.012
Kelly AM, Golden-Mason L, Traynor O, Geoghegan J, McEntee G, Hegarty JE, O’Farrelly C (2006) Changes in hepatic immunoregulatory cytokines in patients with metastatic colorectal carcinoma: implications for hepatic anti-tumour immunity. Cytokine 35(3–4):171–179. https://doi.org/10.1016/j.cyto.2006.07.019
O’Toole A, Michielsen AJ, Nolan B, Tosetto M, Sheahan K, Mulcahy HE, Winter DC, Hyland JM, O’Connell PR, Fennelly D, O’Donoghue D, O’Sullivan J, Doherty GA, Ryan EJ (2014) Tumour microenvironment of both early- and late-stage colorectal cancer is equally immunosuppressive. Br J Cancer 111(5):927–932. https://doi.org/10.1038/bjc.2014.367
Michielsen AJ, Hogan AE, Marry J, Tosetto M, Cox F, Hyland JM, Sheahan KD, O’Donoghue DP, Mulcahy HE, Ryan EJ, O’Sullivan JN (2011) Tumour tissue microenvironment can inhibit dendritic cell maturation in colorectal cancer. PLoS One 6(11):e27944. https://doi.org/10.1371/journal.pone.0027944
Weiskirchen R, Tacke F (2014) Cellular and molecular functions of hepatic stellate cells in inflammatory responses and liver immunology. Hepatobiliary Surg Nutr 3(6):344–363. https://doi.org/10.3978/j.issn.2304-3881.2014.11.03
Harmon C, Robinson MW, Fahey R, Whelan S, Houlihan DD, Geoghegan J, O’Farrelly C (2016) Tissue-resident Eomes(hi) T-bet(lo) CD56(bright) NK cells with reduced proinflammatory potential are enriched in the adult human liver. Eur J Immunol 46(9):2111–2120. https://doi.org/10.1002/eji.201646559
dos Santos AC, Barsante MM, Arantes RM, Bernard CC, Teixeira MM, Carvalho-Tavares J (2005) CCL2 and CCL5 mediate leukocyte adhesion in experimental autoimmune encephalomyelitis—an intravital microscopy study. J Neuroimmunol 162(1–2):122–129. https://doi.org/10.1016/j.jneuroim.2005.01.020
Vilela MC, Mansur DS, Lacerda-Queiroz N, Rodrigues DH, Lima GK, Arantes RM, Kroon EG, da Silva Campos MA, Teixeira MM, Teixeira AL (2009) The chemokine CCL5 is essential for leukocyte recruitment in a model of severe Herpes simplex encephalitis. Ann N Y Acad Sci 1153:256–263. https://doi.org/10.1111/j.1749-6632.2008.03959.x
Muthuswamy R, Berk E, Junecko BF, Zeh HJ, Zureikat AH, Normolle D, Luong TM, Reinhart TA, Bartlett DL, Kalinski P (2012) NF-κB hyperactivation in tumor tissues allows tumor-selective reprogramming of the chemokine microenvironment to enhance the recruitment of cytolytic T effector cells. Cancer Res 72(15):3735–3743. https://doi.org/10.1158/0008-5472.CAN-11-4136
Turner MD, Nedjai B, Hurst T, Pennington DJ (2014) Cytokines and chemokines: at the crossroads of cell signalling and inflammatory disease. Biochim Biophys Acta 1843(11):2563–2582. https://doi.org/10.1016/j.bbamcr.2014.05.014
Dupaul-Chicoine J, Arabzadeh A, Dagenais M, Douglas T, Champagne C, Morizot A, Rodrigue-Gervais IG, Breton V, Colpitts SL, Beauchemin N, Saleh M (2015) The Nlrp3 inflammasome suppresses colorectal cancer metastatic growth in the liver by promoting natural killer cell tumoricidal activity. Immunity 43(4):751–763. https://doi.org/10.1016/j.immuni.2015.08.013
Brackett CM, Kojouharov B, Veith J, Greene KF, Burdelya LG, Gollnick SO, Abrams SI, Gudkov AV (2016) Toll-like receptor-5 agonist, entolimod, suppresses metastasis and induces immunity by stimulating an NK-dendritic-CD8 + T-cell axis. Proc Natl Acad Sci USA 113(7):E874–83. https://doi.org/10.1073/pnas.1521359113
White DA, Su Y, Kanellakis P, Kiriazis H, Morand EF, Bucala R, Dart AM, Gao XM, Du XJ (2014) Differential roles of cardiac and leukocyte derived macrophage migration inhibitory factor in inflammatory responses and cardiac remodelling post myocardial infarction. J Mol Cell Cardiol 69:32–42. https://doi.org/10.1016/j.yjmcc.2014.01.015
Wagner L, Klemann C, Stephan M, von Hörsten S (2016) Unravelling the immunological roles of dipeptidyl peptidase 4 (DPP4) activity and/or structure homologue (DASH) proteins. Clin Exp Immunol 184(3):265–283. https://doi.org/10.1111/cei.12757
Moschen AR, Adolph TE, Gerner RR, Wieser V, Tilg H (2017) Lipocalin-2: a master mediator of intestinal and metabolic inflammation. Trends Endocrinol Metab 28(5):388–397. https://doi.org/10.1016/j.tem.2017.01.003
Zhou SL, Dai Z, Zhou ZJ, Wang XY, Yang GH, Wang Z, Huang XW, Fan J, Zhou J (2012) Overexpression of CXCL5 mediates neutrophil infiltration and indicates poor prognosis for hepatocellular carcinoma. Hepatology 56(6):2242-54. https://doi.org/10.1002/hep.25907
Fridlender ZG, Sun J, Kim S, Kapoor V, Cheng G, Ling L, Worthen GS, Albelda SM (2009) Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 16(3):183–194. https://doi.org/10.1016/j.ccr.2009.06.017
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51. https://doi.org/10.1016/j.cell.2010.03.014
Halama N, Zoernig I, Berthel A, Kahlert C, Klupp F, Suarez-Carmona M, Suetterlin T, Brand K, Krauss J, Lasitschka F, Lerchl T, Luckner-Minden C, Ulrich A, Koch M, Weitz J, Schneider M, Buechler MW, Zitvogel L, Herrmann T, Benner A, Kunz C, Luecke S, Springfeld C, Grabe N, Falk CS, Jaeger D (2016) Tumoral immune cell exploitation in colorectal cancer metastases can be targeted effectively by anti-CCR5 therapy in cancer patients. Cancer Cell 29(4):587–601. https://doi.org/10.1016/j.ccell.2016.03.005
Furze RC, Rankin SM (2008) Neutrophil mobilization and clearance in the bone marrow. Immunology 125(3):281–288. https://doi.org/10.1111/j.1365-2567.2008.02950.x
Ahuja N, Andres-Hernando A, Altmann C, Bhargava R, Bacalja J, Webb RG, He Z, Edelstein CL, Faubel S (2012) Circulating IL-6 mediates lung injury via CXCL1 production after acute kidney injury in mice. Am J Physiol Renal Physiol 303(6):F864-72. https://doi.org/10.1152/ajprenal.00025.2012
Asfaha S, Dubeykovskiy AN, Tomita H, Yang X, Stokes S, Shibata W, Friedman RA, Ariyama H, Dubeykovskaya ZA, Muthupalani S, Ericksen R, Frucht H, Fox JG, Wang TC (2013) Mice that express human interleukin-8 have increased mobilization of immature myeloid cells, which exacerbates inflammation and accelerates colon carcinogenesis. Gastroenterology 144(1):155–166. https://doi.org/10.1053/j.gastro.2012.09.057
Mantovani A, Cassatella MA, Costantini C, Jaillon S (2011) Neutrophils in the activation and regulation of innate and adaptive immunity. Nat Rev Immunol 11(8):519–531. https://doi.org/10.1038/nri3024
Lee YS, Choi I, Ning Y, Kim NY, Khatchadourian V, Yang D, Chung HK, Choi D, LaBonte MJ, Ladner RD, Nagulapalli Venkata KC, Rosenberg DO, Petasis NA, Lenz HJ, Hong YK (2012) Interleukin-8 and its receptor CXCR2 in the tumour microenvironment promote colon cancer growth, progression and metastasis. Br J Cancer 106(11):1833–1841. https://doi.org/10.1038/bjc.2012.177
Wang D, Sun H, Wei J, Cen B, DuBois RN (2017) CXCL1 is critical for pre-metastatic niche formation and metastasis in colorectal cancer. Cancer Res 77(13):3655–3665. https://doi.org/10.1158/0008-5472.CAN-16-3199
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoué F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pagès F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964. https://doi.org/10.1126/science.1129139
Pugh SA, Harrison RJ, Primrose JN, Khakoo SI (2014) T cells but not NK cells are associated with a favourable outcome for resected colorectal liver metastases. BMC Cancer. https://doi.org/10.1186/1471-2407-14-180
Berthel A, Zoernig I, Valous NA, Kahlert C, Klupp F, Ulrich A, Weitz J, Jaeger D, Halama N (2017) Detailed resolution analysis reveals spatial T cell heterogeneity in the invasive margin of colorectal cancer liver metastases associated with improved survival. Oncoimmunology 6(3):e1286436. https://doi.org/10.1080/2162402X.2017.1286436
Jaillon S, Galdiero MR, Del Prete D, Cassatella MA, Garlanda C, Mantovani A (2013) Neutrophils in innate and adaptive immunity. Semin Immunopathol 35(4):377–394. https://doi.org/10.1007/s00281-013-0374-8
Galdiero MR, Bianchi P, Grizzi F, Di Caro G, Basso G, Ponzetta A, Bonavita E, Barbagallo M, Tartari S, Polentarutti N, Malesci A, Marone G, Roncalli M, Laghi L, Garlanda C, Mantovani A, Jaillon S (2016) Occurrence and significance of tumor-associated neutrophils in patients with colorectal cancer. Int J Cancer 139(2):446–456. https://doi.org/10.1002/ijc.30076
Wculek SK, Malanchi I (2015) Neutrophils support lung colonization of metastasis-initiating breast cancer cells. Nature 528(7582):413–417. https://doi.org/10.1038/nature16140
Tohme S, Yazdani HO, Al-Khafaji AB, Chidi AP, Loughran P, Mowen K, Wang Y, Simmons RL, Huang H, Tsung A (2016) Neutrophil extracellular traps promote the development and progression of liver metastases after surgical stress. Cancer Res 76(6):1367–1380. https://doi.org/10.1158/0008-5472.CAN-15-1591
Treffers LW, Hiemstra IH, Kuijpers TW, van den Berg TK, Matlung HL (2016) Neutrophils in cancer. Immunol Rev 273(1):312–328. https://doi.org/10.1111/imr.12444
Fridlender ZG, Albelda SM (2012) Tumor-associated neutrophils: friend or foe? Carcinogenesis 33(5):949 – 55. https://doi.org/10.1093/carcin/bgs123
Mishalian I, Bayuh R, Eruslanov E, Michaeli J, Levy L, Zolotarov L, Singhal S, Albelda SM, Granot Z, Fridlender ZG (2014) Neutrophils recruit regulatory T-cells into tumors via secretion of CCL17—a new mechanism of impaired antitumor immunity. Int J Cancer 135(5):1178–1186. https://doi.org/10.1002/ijc.28770
Shaul ME, Levy L, Sun J, Mishalian I, Singhal S, Kapoor V, Horng W, Fridlender G, Albelda SM, Fridlender ZG (2016) Tumor-associated neutrophils display a distinct N1 profile following TGFβ modulation: a transcriptomics analysis of pro- vs. antitumor TANs. Oncoimmunology 5(11):e1232221. https://doi.org/10.1080/2162402X.2016.1232221
Galon J, Mlecnik B, Bindea G, Angell HK, Berger A, Lagorce C, Lugli A, Zlobec I, Hartmann A, Bifulco C, Nagtegaal ID, Palmqvist R, Masucci GV, Botti G, Tatangelo F, Delrio P, Maio M, Laghi L, Grizzi F, Asslaber M, D’Arrigo C, Vidal-Vanaclocha F, Zavadova E, Chouchane L, Ohashi PS, Hafezi-Bakhtiari S, Wouters BG, Roehrl M, Nguyen L, Kawakami Y, Hazama S, Okuno K, Ogino S, Gibbs P, Waring P, Sato N, Torigoe T, Itoh K, Patel PS, Shukla SN, Wang Y, Kopetz S, Sinicrope FA, Scripcariu V, Ascierto PA, Marincola FM, Fox BA, Pagès F (2014) Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J Pathol 232(2):199–209. https://doi.org/10.1002/path.4287
Kirilovsky A, Marliot F, El Sissy C, Haicheur N, Galon J, Pagès F (2016) Rational bases for the use of the Immunoscore in routine clinical settings as a prognostic and predictive biomarker in cancer patients. Int Immunol 28(8):373–382. https://doi.org/10.1093/intimm/dxw021
Kwak Y, Koh J, Kim DW, Kang SB, Kim WH, Lee HS (2016) Immunoscore encompassing CD3+ and CD8+ T cell densities in distant metastasis is a robust prognostic marker for advanced colorectal cancer. Oncotarget 7(49):81778–81790. https://doi.org/10.18632/oncotarget.13207
Proctor MJ, Talwar D, Balmar SM, O’Reilly DS, Foulis AK, Horgan PG, Morrison DS, McMillan DC (2010) The relationship between the presence and site of cancer, an inflammation-based prognostic score and biochemical parameters. Initial results of the Glasgow Inflammation Outcome Study. Br J Cancer 103(6):870–876. https://doi.org/10.1038/sj.bjc.6605855
Tang H, Li B, Zhang A, Lu W, Xiang C, Dong J (2016) Prognostic Significance of Neutrophil-to-Lymphocyte Ratio in Colorectal Liver Metastasis: A Systematic Review and Meta-Analysis. PLoS One 11(7):e0159447. https://doi.org/10.1371/journal.pone.0159447 (eCollection 2016)
Funding
This work was supported by grants from the Health Research Board of Ireland (RP 2008/189) to C.O’Farrelly and HRA-POR-2013-281 to E.J. Ryan and a Science Foundation Ireland Investigator Award (132/IA/1667) to C. O’Farrelly.
Author information
Authors and Affiliations
Contributions
FH: study design and conception, data collection, analysis and interpretation, drafted, reviewed, edited, and approved manuscript. CH: study design and conception, data analysis and interpretation, drafted, and approved manuscript. LAE: data collection and analysis, and reviewed and approved manuscript. FC: data collection and analysis, and reviewed and approved manuscript. AL: data analysis, and reviewed and approved manuscript. DM: provided clinical samples, and reviewed and approved manuscript. EH: provided clinical samples, and reviewed and approved manuscript. NN: data collection and analysis, and reviewed and approved manuscript. JGG: study design and conception, provided clinical samples, and reviewed and approved manuscript. EJR: study conception, design and supervision, data collection, and analysis, and drafted and approved manuscript. CO’F: study conception, design and supervision, and drafted and approved manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval and ethical standards
All protocols were approved by St. Vincent’s University Hospital Ethics Committee in accordance with the ethical guidelines of the 1975 Declaration of Helsinki.
Informed consent
Informed consent was provided by all patients prior to undergoing resection. A patient information leaflet was provided and all queries addressed by F. Hand before consent was provided. Where donor liver was obtained, informed consent was given by the donor’s family prior to sampling. Donor families were approached by a dedicated transplant coordinator. All protocols and study information were provided to the family prior to consenting.
Rights and permissions
About this article
Cite this article
Hand, F., Harmon, C., Elliott, L.A. et al. Depleted polymorphonuclear leukocytes in human metastatic liver reflect an altered immune microenvironment associated with recurrent metastasis. Cancer Immunol Immunother 67, 1041–1052 (2018). https://doi.org/10.1007/s00262-018-2149-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-018-2149-5