Abstract
Background
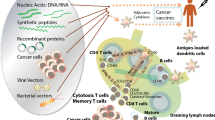
Therapeutic vaccines for cancer are an attractive alternative to conventional therapies, since the later result in serious adverse effects and in most cases are not effective against advanced disease. Human papillomavirus (HPV) is responsible for several malignancies such as cervical carcinoma. Vaccines targeting oncogenic viral proteins like HPV16-E6 and HPV16-E7 are ideal candidates to elicit strong immune responses without generating autoimmunity because: (1) these products are not expressed in normal cells and (2) their expression is required to maintain the malignant phenotype. Our group has developed peptide vaccination strategy called TriVax, which is effective in generating vast numbers of antigen-specific T cells in mice capable of persisting for long time periods.
Materials and methods
We have used two HPV-induced mouse cancer models (TC-1 and C3.43) to evaluate the immunogenicity and therapeutic efficacy of TriVax prepared with the immunodominant CD8 T-cell epitope HPV16-E749-57, mixed with poly-IC adjuvant and costimulatory anti-CD40 antibodies.
Results
TriVax using HPV16-E749-57 induced large and persistent T-cell responses that were therapeutically effective against established HPV16-E7 expressing tumors. In most cases, TriVax was successful in attaining complete rejections of 6–11-day established tumors. In addition, TriVax induced long-term immunological memory, which prevented tumor recurrences. The anti-tumor effects of TriVax were independent of NK and CD4 T cells and, surprisingly, did not rely to a great extent on type-I or type-II interferon.
Conclusions
These findings indicate that the TriVax strategy is an appealing immunotherapeutic approach for the treatment of established viral-induced tumors. We believe that these studies may help to launch more effective and less invasive therapeutic vaccines for HPV-mediated malignancies.





Similar content being viewed by others
Abbreviations
- αCD40 mAb:
-
Anti-CD40 monoclonal antibodies
- CC:
-
Cervical carcinoma
- DC:
-
Dendritic cell
- HPV:
-
Human papillomavirus
- MHC-I:
-
Major histocompatibility complex I
- IFNγ:
-
Interferon-gamma
- IFNαβR:
-
Interferon-alpha/beta receptor
- KO:
-
Knockout
- TAA:
-
Tumor-associated antigen
- TLR:
-
Toll-like receptor
References
Smith JS, Lindsay L, Hoots B, Keys J, Franceschi S, Winer R, Clifford GM (2007) Human papillomavirus type distribution in invasive cervical cancer and high-grade cervical lesions: a meta-analysis update. Int J Cancer 121:621–632
Fleurence RL, Dixon JM, Milanova TF, Beusterien KM (2007) Review of the economic and quality-of-life burden of cervical human papillomavirus disease. Am J Obstet Gynecol 196:206–212
Insinga RP, Glass AG, Rush BB (2004) The health care costs of cervical human papillomavirus–related disease. Am J Obstet Gynecol 191:114–120
Hildesheim A, Herrero R, Wacholder S, Rodriguez AC, Solomon D, Bratti MC, Schiller JT, Gonzalez P, Dubin G, Porras C, Jimenez SE, Lowy DR (2007) Effect of human papillomavirus 16/18 L1 viruslike particle vaccine among young women with preexisting infection: a randomized trial. JAMA 298:743–753
FUTURE II Study Group (2007) Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med 356:1915–1927
Kaufmann AM, Nieland JD, Jochmus I, Baur S, Friese K, Gabelsberger J, Gieseking F, Gissmann L, Glasschroder B, Grubert T, Hillemanns P, Hopfl R, Ikenberg H, Schwarz J, Karrasch M, Knoll A, Kuppers V, Lechmann M, Lelle RJ, Meissner H, Muller RT, Pawlita M, Petry KU, Pilch H, Walek E, Schneider A (2007) Vaccination trial with HPV16 L1E7 chimeric virus-like particles in women suffering from high grade cervical intraepithelial neoplasia (CIN 2/3). Int J Cancer 121:2794–2800
Bach PB (2010) Gardasil: from bench, to bedside, to blunder. Lancet 375:963–964
Cecchini S, Carozzi F, Confortini M, Zappa M, Ciatto S (2004) Persistent human papilloma virus infection as an indicator of risk of recurrence of high-grade cervical intraepithelial neoplasia treated by the loop electrosurgical excision procedure. Tumori 90:225–228
Kaech SM, Wherry EJ, Ahmed R (2002) Effector and memory T-cell differentiation: implications for vaccine development. Nat Rev Immunol 2:251–262
Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, Celis E, Greenberg PD (2002) Adoptive T cell therapy using antigen-specific CD8 + T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci U S A 99:16168–16173
Dudley ME, Wunderlich JR, Robbins PF, Yang JC, Hwu P, Schwartzentruber DJ, Topalian SL, Sherry R, Restifo NP, Hubicki AM, Robinson MR, Raffeld M, Duray P, Seipp CA, Rogers-Freezer L, Morton KE, Mavroukakis SA, White DE, Rosenberg SA (2002) Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298:850–854
Hung CF, Ma B, Monie A, Tsen SW, Wu TC (2008) Therapeutic human papillomavirus vaccines: current clinical trials and future directions. Expert Opin Biol Ther 8:421–439
Daayana S, Elkord E, Winters U, Pawlita M, Roden R, Stern PL, Kitchener HC (2010) Phase II trial of imiquimod and HPV therapeutic vaccination in patients with vulval intraepithelial neoplasia. Br J Cancer 102:1129–1136
Kenter GG, Welters MJ, Valentijn AR, Lowik MJ, Berends-van der Meer DM, Vloon AP, Essahsah F, Fathers LM, Offringa R, Drijfhout JW, Wafelman AR, Oostendorp J, Fleuren GJ, van der Burg SH, Melief CJ (2009) Vaccination against HPV-16 oncoproteins for vulvar intraepithelial neoplasia. N Engl J Med 361:1838–1847
Zeng Q, Peng S, Monie A, Yang M, Pang X, Hung CF, Wu TC (2011) Control of cervicovaginal HPV-16 E7-expressing tumors by the combination of therapeutic HPV vaccination and vascular disrupting agents. Hum Gene Ther 22:809–819
Wu CY, Monie A, Pang X, Hung CF, Wu TC (2010) Improving therapeutic HPV peptide-based vaccine potency by enhancing CD4+ T help and dendritic cell activation. J Biomed Sci 17:88
Daftarian PM, Mansour M, Pohajdak B, Fuentes-Ortega A, Korets-Smith E, Macdonald L, Weir G, Brown RG, Kast WM (2007) Rejection of large HPV-16 expressing tumors in aged mice by a single immunization of VacciMax encapsulated CTL/T helper peptides. J Transl Med 5:26
Roman LD, Wilczynski S, Muderspach LI, Burnett AF, O’Meara A, Brinkman JA, Kast WM, Facio G, Felix JC, Aldana M, Weber JS (2007) A phase II study of Hsp-7 (SGN-00101) in women with high-grade cervical intraepithelial neoplasia. Gynecol Oncol 106:558–566
Feltkamp MC, Smits HL, Vierboom MP, Minnaar RP, de Jongh BM, Drijfhout JW, ter Schegget J, Melief CJ, Kast WM (1993) Vaccination with cytotoxic T lymphocyte epitope-containing peptide protects against a tumor induced by human papillomavirus type 16-transformed cells. Eur J Immunol 23:2242–2249
Melief CJ, van der Burg SH (2008) Immunotherapy of established (pre)malignant disease by synthetic long peptide vaccines. Nat Rev Cancer 8:351–360
van Driel WJ, Ressing ME, Kenter GG, Brandt RM, Krul EJ, van Rossum AB, Schuuring E, Offringa R, Bauknecht T, Tamm-Hermelink A, van Dam PA, Fleuren GJ, Kast WM, Melief CJ, Trimbos JB (1999) Vaccination with HPV16 peptides of patients with advanced cervical carcinoma: clinical evaluation of a phase I-II trial. Eur J Cancer 35:946–952
Assudani D, Cho HI, DeVito N, Bradley N, Celis E (2008) In vivo expansion, persistence, and function of peptide vaccine-induced CD8 T cells occur independently of CD4 T cells. Cancer Res 68:9892–9899
Cho HI, Celis E (2009) Optimized peptide vaccines eliciting extensive CD8 T-cell responses with therapeutic antitumor effects. Cancer Res 69:9012–9019
Lin KY, Guarnieri FG, Staveley-O’Carroll KF, Levitsky HI, August JT, Pardoll DM, Wu TC (1996) Treatment of established tumors with a novel vaccine that enhances major histocompatibility class II presentation of tumor antigen. Cancer Res 56:21–26
Sadovnikova E, Zhu X, Collins SM, Zhou J, Vousden K, Crawford L, Beverley P, Stauss HJ (1994) Limitations of predictive motifs revealed by cytotoxic T lymphocyte epitope mapping of the human papilloma virus E7 protein. Int Immunol 6:289–296
Nava-Parada P, Forni G, Knutson KL, Pease LR, Celis E (2007) Peptide vaccine given with a Toll-like receptor agonist is effective for the treatment and prevention of spontaneous breast tumors. Cancer Res 67:1326–1334
Cho HI, Lee YR, Celis E (2011) Interferon gamma limits the effectiveness of melanoma peptide vaccines. Blood 117:135–144
von Knebel DoeberitzM, Rittmuller C, zur Hausen H, Durst M (1992) Inhibition of tumorigenicity of cervical cancer cells in nude mice by HPV E6-E7 anti-sense RNA. Int J Cancer 51:831–834
Hu G, Liu W, Hanania EG, Fu S, Wang T, Deisseroth AB (1995) Suppression of tumorigenesis by transcription units expressing the antisense E6 and E7 messenger RNA (mRNA) for the transforming proteins of the human papilloma virus and the sense mRNA for the retinoblastoma gene in cervical carcinoma cells. Cancer Gene Ther 2:19–32
Bennett SR, Carbone FR, Toy T, Miller JF, Heath WR (1998) B cells directly tolerize CD8(+) T cells. J Exp Med 188:1977–1983
Suhrbier A, Burrows SR, Fernan A, Lavin MF, Baxter GD, Moss DJ (1993) Peptide epitope induced apoptosis of human cytotoxic T lymphocytes. Implications for peripheral T cell deletion and peptide vaccination. J Immunol 150:2169–2178
Toes RE, van der Voort EI, Schoenberger SP, Drijfhout JW, van Bloois L, Storm G, Kast WM, Offringa R, Melief CJ (1998) Enhancement of tumor outgrowth through CTL tolerization after peptide vaccination is avoided by peptide presentation on dendritic cells. J Immunol 160:4449–4456
Borrow P, Tough DF, Eto D, Tishon A, Grewal IS, Sprent J, Flavell RA, Oldstone MB (1998) CD40 ligand-mediated interactions are involved in the generation of memory CD8(+) cytotoxic T lymphocytes (CTL) but are not required for the maintenance of CTL memory following virus infection. J Virol 72:7440–7449
Ishii KJ, Koyama S, Nakagawa A, Coban C, Akira S (2008) Host innate immune receptors and beyond: making sense of microbial infections. Cell Host Microbe 3:352–363
Kawai T, Akira S (2008) Toll-like receptor and RIG-I-like receptor signaling. Ann N Y Acad Sci 1143:1–20
Xiao Z, Casey KA, Jameson SC, Curtsinger JM, Mescher MF (2009) Programming for CD8 T cell memory development requires IL-12 or type I IFN. J Immunol 182:2786–2794
Billiau A, Matthys P (2009) Interferon-gamma: a historical perspective. Cytokine Growth Factor Rev 20:97–113
Dunn GP, Ikeda H, Bruce AT, Koebel C, Uppaluri R, Bui J, Chan R, Diamond M, White JM, Sheehan KC, Schreiber RD (2005) Interferon-gamma and cancer immunoediting. Immunol Res 32:231–245
Apte SH, Groves P, Olver S, Baz A, Doolan DL, Kelso A, Kienzle N (2010) IFN-gamma inhibits IL-4-induced type 2 cytokine expression by CD8 T cells in vivo and modulates the anti-tumor response. J Immunol 185:998–1004
Acknowledgments
We gratefully acknowledge Dr. T-C Wu and Dr. W. M. Kast for providing us with the tumor cell lines and are indebted to Dr. A. Salazar for providing large amounts of Poly-ICLC. We also thank Moffitt Cancer Center Flow Cytometry Core, especially J. Kroger for her help in flow cytometer training. This work was supported by NIH grants R01CA136828 and R01CA157303 to EC.
Conflict of interest
Esteban Celis has filed a patent application based on the use of synthetic peptides and poly-IC combinatorial complexes for vaccination. The rights of the patent application have been transferred to the Moffitt Cancer Center. Kelly Barrios declares no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barrios, K., Celis, E. TriVax-HPV: an improved peptide-based therapeutic vaccination strategy against human papillomavirus-induced cancers. Cancer Immunol Immunother 61, 1307–1317 (2012). https://doi.org/10.1007/s00262-012-1259-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1259-8