Abstract
Purpose
To compare the difference of evaluating the microcirculatory function status of primary small HCC between DCE-MRI with two-compartmental pharmacokinetic model and IVIM-DWI.
Methods
27 patients (22 men, 5 women; mean age, 49 years; range 36–65 years) with primary single sHCC who underwent IVIM-DWI and DCE–MRI before the operation were included in this retrospective study. The MR perfusion parameters are Ktrans, Ve, Kep, D, D* and f. Pathological results include pathological grade (low grade ≤ II, high grade > II) and MVD. The perfusion parameters and pathological results of sHCC were analyzed and compared in their relevance, sensitivity and specificity. Statistical methods included Spearman and ROC curve analysis.
Results
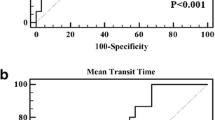
The perfusion parameters (Ktrans, Kep, D*, f) were significantly positive correlated (r = 0.892, 0.808, 0.589 and 0.543, P = 0.000, 0.000, 0.001 and 0.003 with MVD of sHCC. The parameter Ve and D values were negatively correlated (r = − 0.454 and − 0.399, P = 0.017 and 0.039, respectively) with the pathological grade. Regarding the evaluation MVD of sHCC, the evaluation of the sensitivity and specificity performance was present in descending order: Ktrans > Kep > PF > D*. In the evaluation pathological grade of sHCC, the sensitivity and specificity were better by parameters D than Ve.
Conclusion
DCE-MRI is better than IVIM-DWI for evaluation microcirculation functional status of sHCC. But for evaluating the pathological grade, IVIM-DWI is better than DCE-MRI. Combination of the two imaging techniques may provide more comprehensive evaluation in microcirculation functional status of the sHCC.




Similar content being viewed by others
References
Onaca N, Davis GL, Jennings LW, Goldstein RM, Klintmalm GB (2009) Improved results of transplantation for hepatocellular carcinoma: a report from the International Registry of Hepatic Tumors in Liver Transplantation. Liver Transpl 15 (6):574-580. https://doi.org/10.1002/lt.21738
Chebib I, Shabani-Rad MT, Chow MS, Zhang J, Gao ZH (2007) Microvessel density and clinicopathologic characteristics in hepatocellular carcinoma with and without cirrhosis. Biomark Insights 2:59-68
Weidner N (1995) Current pathologic methods for measuring intratumoral microvessel density within breast carcinoma and other solid tumors. Breast Cancer Res Treat 36 (2):169-180
Zhang Q, Chen X, Zhou J, Zhang L, Zhao Q, Chen G, Xu J, Qian F, Chen Z (2006) CD147, MMP-2, MMP-9 and MVD-CD34 are significant predictors of recurrence after liver transplantation in hepatocellular carcinoma patients. Cancer Biol Ther 5 (7):808-814
Chen J, Chen C, Xia C, Huang Z, Zuo P, Stemmer A, Song B (2018) Quantitative free-breathing dynamic contrast-enhanced MRI in hepatocellular carcinoma using gadoxetic acid: correlations with Ki67 proliferation status, histological grades, and microvascular density. Abdom Radiol (NY) 43 (6):1393-1403. https://doi.org/10.1007/s00261-017-1320-3
Munoz NM, Minhaj AA, Maldonado KL, Kingsley CV, Cortes AC, Taghavi H, Polak U, Mitchell JM, Ensor JE, Bankson JA, Rashid A, Avritscher R (2019) Comparison of dynamic contrast-enhanced magnetic resonance imaging and contrast-enhanced ultrasound for evaluation of the effects of sorafenib in a rat model of hepatocellular carcinoma. Magn Reson Imaging 57:156-164. https://doi.org/10.1016/j.mri.2018.11.012
Woo S, Lee JM, Yoon JH, Joo I, Han JK, Choi BI (2014) Intravoxel incoherent motion diffusion-weighted MR imaging of hepatocellular carcinoma: correlation with enhancement degree and histologic grade. Radiology 270 (3):758-767. https://doi.org/10.1148/radiol.13130444
Hectors SJ, Wagner M, Besa C, Bane O, Dyvorne HA, Fiel MI, Zhu H, Donovan M, Taouli B (2016) Intravoxel incoherent motion diffusion-weighted imaging of hepatocellular carcinoma: Is there a correlation with flow and perfusion metrics obtained with dynamic contrast-enhanced MRI? J Magn Reson Imaging 44 (4):856-864. https://doi.org/10.1002/jmri.25194
Zhang Y, Liu H, Xiao W, Zhu L, Wang N, He X, Jiang Z, Guan B (2017) Quantitative dynamic contrast-enhanced magnetic resonance imaging in a VX2 rabbit liver tumour model using different gadolinium-based contrast agents: comparison of DCE-MRI quantitative results between Magnevist and Eovist. BJR Case Rep 3 (3):20160099. https://doi.org/10.1259/bjrcr.20160099
Zhu SC, Liu YH, Wei Y, Li LL, Dou SW, Sun TY, Shi DP (2018) Intravoxel incoherent motion diffusion-weighted magnetic resonance imaging for predicting histological grade of hepatocellular carcinoma: Comparison with conventional diffusion-weighted imaging. World J Gastroenterol 24 (8):929-940. https://doi.org/10.3748/wjg.v24.i8.929
Kenmochi K, Sugihara S, Kojiro M (1987) Relationship of histologic grade of hepatocellular carcinoma (HCC) to tumor size, and demonstration of tumor cells of multiple different grades in single small HCC. Liver 7 (1):18-26
Wei Y, Huang Z, Tang H, Deng L, Yuan Y, Li J, Wu D, Wei X, Song B (2019) IVIM improves preoperative assessment of microvascular invasion in HCC. Eur Radiol. https://doi.org/10.1007/s00330-019-06088-w
Yamada I, Aung W, Himeno Y, Nakagawa T, Shibuya H (1999) Diffusion coefficients in abdominal organs and hepatic lesions: evaluation with intravoxel incoherent motion echo-planar MR imaging. Radiology 210 (3):617-623. https://doi.org/10.1148/radiology.210.3.r99fe17617
Kakite S, Dyvorne H, Besa C, Cooper N, Facciuto M, Donnerhack C, Taouli B (2015) Hepatocellular carcinoma: short-term reproducibility of apparent diffusion coefficient and intravoxel incoherent motion parameters at 3.0T. J Magn Reson Imaging 41 (1):149-156. https://doi.org/10.1002/jmri.24538
Witjes CD, Willemssen FE, Verheij J, van der Veer SJ, Hansen BE, Verhoef C, de Man RA, Ijzermans JN (2012) Histological differentiation grade and microvascular invasion of hepatocellular carcinoma predicted by dynamic contrast-enhanced MRI. J Magn Reson Imaging 36 (3):641-647. https://doi.org/10.1002/jmri.23681
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Song, Q., Guo, Y., Yao, X. et al. Comparative study of evaluating the microcirculatory function status of primary small HCC between the CE (DCE-MRI) and Non-CE (IVIM-DWI) MR Perfusion Imaging. Abdom Radiol 46, 2575–2583 (2021). https://doi.org/10.1007/s00261-020-02945-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-020-02945-1