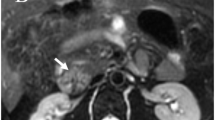
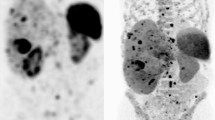
Abstract
Pancreatic neuroendocrine tumors (pNETs) are rare neoplasms that secrete peptides and neuro-amines. pNETs can be sporadic or hereditary, syndromic or non-syndromic with different clinical presentations and prognoses. The role of medical imaging includes locating the tumor, assessing its extent, and evaluating the feasibility of curative surgery or cytoreduction. Pancreatic NETs have very distinctive phenotypes on CT, MRI, and PET. PET have been demonstrated to be very sensitive to detect either well-differentiated pNETs using 68Gallium somatostatin receptor (SSTR) radiotracers, or more aggressive undifferentiated pNETS using 18F-FDG. A comprehensive interpretation of multimodal imaging guides resectability and cytoreduction in pNETs. The imaging phenotype provides information on the differentiation and proliferation of pNETs, as well as the spatial and temporal heterogeneity of tumors with prognostic and therapeutic implications. This review provides a structured approach for standardized reading and reporting of medical imaging studies with a focus on PET and MR techniques. It explains which imaging approach should be used for different subtypes of pNET and what a radiologist should be looking for and reporting when interpreting these studies.










Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- AJCC:
-
American Joint Committee on Cancer
- APUD:
-
Amine precursor uptake and decarboxylation
- CEUS:
-
Contrast-enhanced ultra sound examination
- CT:
-
Computed tomography
- DOPA:
-
Dihydroxyphenylalanine
- DOTATOC:
-
DOTA0-Phe1-Tyr3 octreotide
- DWI:
-
Diffusion-weighted imaging
- ENETs:
-
European Neuroendocrine Tumor Society
- EUS:
-
Endoscopic ultrasound examination
- 18F-FDG:
-
18Fluoro-Fluorodeoxyglucose
- 68Ga:
-
Gallium-68
- 99mTc:
-
Technetium 99m
- GLP-1:
-
Glucagon-like peptide 1
- IACIG:
-
Intra-arterial injection of calcium
- IOUS:
-
Intraoperative ultrasound examination
- LM:
-
Liver metastases
- MEN:
-
Multiple endocrine neoplasia syndrome
- MRI:
-
Magnetic resonance imaging
- NANETS:
-
North American Neuroendocrine Tumor Society
- NF:
-
Neurofibromatosis
- NET:
-
Neuroendocrine tumors
- OS:
-
Overall survival
- PDAC:
-
Pancreatic ductal adenocarcinoma
- pNET:
-
Pancreatic tumor
- SSTR:
-
Somatostatin receptor
- SSTR-PET:
-
Somatostatin receptor PET
- SSTR scintigraphy:
-
Somatostatin receptor scintigraphy
- SUV:
-
Standard uptake value
- TSC:
-
Tuberous sclerosis complex
- US:
-
Ultrasound examination
- VHL:
-
Von Hippel–Lindau syndrome
- WHO:
-
World Health Organization
References
Canellas R, Lo G, Bhowmik S, Ferrone C, Sahani D (2018) Pancreatic neuroendocrine tumor: Correlations between MRI features, tumor biology, and clinical outcome after surgery. J Magn Reson Imaging 47 (2):425-432. https://doi.org/10.1002/jmri.25756
Smith JK, Ng SC, Hill JS, Simons JP, Arous EJ, Shah SA, Tseng JF, McDade TP (2010) Complications after pancreatectomy for neuroendocrine tumors: a national study. J Surg Res 163 (1):63-68. https://doi.org/10.1016/j.jss.2010.04.017
Falconi M, Eriksson B, Kaltsas G, Bartsch DK, Capdevila J, Caplin M, Kos-Kudla B, Kwekkeboom D, Rindi G, Klöppel G, Reed N, Kianmanesh R, Jensen RT, Participants aoVCC (2016) ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology 103 (2):153-171. https://doi.org/10.1159/000443171
Vagefi PA, Razo O, Deshpande V, McGrath DJ, Lauwers GY, Thayer SP, Warshaw AL, Fernández-del Castillo C (2007) Evolving patterns in the detection and outcomes of pancreatic neuroendocrine neoplasms: the Massachusetts General Hospital experience from 1977 to 2005. Archives of Surgery 142 (4):347-354
Ellison TA, Wolfgang CL, Shi C, Cameron JL, Murakami P, Mun LJ, Singhi AD, Cornish TC, Olino K, Meriden Z, Choti M, Diaz LA, Pawlik TM, Schulick RD, Hruban RH, Edil BH (2014) A single institution’s 26-year experience with nonfunctional pancreatic neuroendocrine tumors: a validation of current staging systems and a new prognostic nomogram. Ann Surg 259 (2):204-212. https://doi.org/10.1097/sla.0b013e31828f3174
Siegel RL, Miller KD, Jemal A (2017) Cancer Statistics, 2017. CA Cancer J Clin 67 (1):7-30. https://doi.org/10.3322/caac.21387
Halfdanarson TR, Rabe KG, Rubin J, Petersen GM (2008) Pancreatic neuroendocrine tumors (PNETs): incidence, prognosis and recent trend toward improved survival. Ann Oncol 19 (10):1727-1733. https://doi.org/10.1093/annonc/mdn351
Ambe CM, Nguyen P, Centeno BA, Choi J, Strosberg J, Kvols L, Hodul P, Hoffe S, Malafa MP (2017) Multimodality Management of “Borderline Resectable” Pancreatic Neuroendocrine Tumors: Report of a Single-Institution Experience. Cancer Control 24 (5):1073274817729076. https://doi.org/10.1177/1073274817729076
van Essen M, Sundin A, Krenning EP, Kwekkeboom DJ (2014) Neuroendocrine tumours: the role of imaging for diagnosis and therapy. Nat Rev Endocrinol 10 (2):102-114. https://doi.org/10.1038/nrendo.2013.246
Halfdanarson TR, Rabe KG, Rubin J, Petersen GM (2008) Pancreatic neuroendocrine tumors (PNETs): incidence, prognosis and recent trend toward improved survival. Annals of Oncology 19 (10):1727-1733. https://doi.org/10.1093/annonc/mdn351
Bozkurt MF, Virgolini I, Balogova S, Beheshti M, Rubello D, Decristoforo C, Ambrosini V, Kjaer A, Delgado-Bolton R, Kunikowska J, Oyen WJG, Chiti A, Giammarile F, Fanti S (2017) Guideline for PET/CT imaging of neuroendocrine neoplasms with (68)Ga-DOTA-conjugated somatostatin receptor targeting peptides and (18)F-DOPA. Eur J Nucl Med Mol Imaging 44 (9):1588-1601. https://doi.org/10.1007/s00259-017-3728-y
Gouya H, Vignaux O, Augui J, Dousset B, Palazzo L, Louvel A, Chaussade S, Legmann P (2003) CT, Endoscopic Sonography, and a Combined Protocol for Preoperative Evaluation of Pancreatic Insulinomas. American Journal of Roentgenology 181 (4):987-992. https://doi.org/10.2214/ajr.181.4.1810987
Berends FJ, Cuesta MA, Kazemier G, van Eijck CH, de Herder WW, van Muiswinkel JM, Bruining HA, Bonjer HJ (2000) Laparoscopic detection and resection of insulinomas. Surgery 128 (3):386-391. https://doi.org/10.1067/msy.2000.107413
Klöppel G, Klimstra DS, Hruban RH, Adsay V, Capella C, Couvelard A, Komminoth P, La Rosa S, Ohike N, Osamura RY, Perren A, Scoazec J-Y, Rindi G (2017) Pancreatic Neuroendocrine Tumors: Update on the New World Health Organization Classification. AJSP: Reviews & Reports 22 (5)
Tang LH, Basturk O, Sue JJ, Klimstra DS (2016) A Practical Approach to the Classification of WHO Grade 3 (G3) Well Differentiated Neuroendocrine Tumor (WD-NET) and Poorly Differentiated Neuroendocrine Carcinoma (PD-NEC) of the Pancreas. The American journal of surgical pathology 40 (9):1192-1202. https://doi.org/10.1097/pas.0000000000000662
Dasari A, Shen C, Halperin D, et al. (2017) Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the united states. JAMA Oncol 3 (10):1335-1342. https://doi.org/10.1001/jamaoncol.2017.0589
Sho S, Court CM, Winograd P, Toste PA, Pisegna JR, Lewis M, Donahue TR, Hines OJ, Reber HA, Dawson DW, Tomlinson JS (2018) A Prognostic Scoring System for the Prediction of Metastatic Recurrence Following Curative Resection of Pancreatic Neuroendocrine Tumors. J Gastrointest Surg. https://doi.org/10.1007/s11605-018-4011-7
Jensen RT, Cadiot G, Brandi ML, de Herder WW, Kaltsas G, Komminoth P, Scoazec J-Y, Salazar R, Sauvanet A, Kianmanesh R (2012) ENETS Consensus Guidelines for the Management of Patients with Digestive Neuroendocrine Neoplasms: Functional Pancreatic Endocrine Tumor Syndromes. Neuroendocrinology 95 (2):98-119. https://doi.org/10.1159/000335591
Sadowski SM, Neychev V, Millo C, Shih J, Nilubol N, Herscovitch P, Pacak K, Marx SJ, Kebebew E (2016) Prospective Study of 68Ga-DOTATATE Positron Emission Tomography/Computed Tomography for Detecting Gastro-Entero-Pancreatic Neuroendocrine Tumors and Unknown Primary Sites. J Clin Oncol 34 (6):588-596. https://doi.org/10.1200/jco.2015.64.0987
Buchmann I, Henze M, Engelbrecht S, Eisenhut M, Runz A, Schafer M, Schilling T, Haufe S, Herrmann T, Haberkorn U (2007) Comparison of 68Ga-DOTATOC PET and 111In-DTPAOC (Octreoscan) SPECT in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging 34 (10):1617-1626. https://doi.org/10.1007/s00259-007-0450-1
Oberndorfer S (1907) Karzinoide tumoren des dunndarms. Frankfurt Z Path 1:426-432
Öberg KE (2010) Gastrointestinal neuroendocrine tumors. Annals of Oncology 21 (suppl_7):vii72-vii80
Triponez F, Dosseh D, Goudet P, Cougard P, Bauters C, Murat A, Cadiot G, Niccoli-Sire P, Chayvialle JA, Calender A, Proye CA (2006) Epidemiology data on 108 MEN 1 patients from the GTE with isolated nonfunctioning tumors of the pancreas. Ann Surg 243 (2):265-272. https://doi.org/10.1097/01.sla.0000197715.96762.68
Corrias G, Monti S, Horvat N, Tang L, Basturk O, Saba L, Mannelli L (2018) Imaging features of malignant abdominal neuroendocrine tumors with rare presentation. Clin Imaging 51:59-64. https://doi.org/10.1016/j.clinimag.2018.02.004
Owen N, Sohaib S, Peppercorn P, Monson J, Grossman A, Besser G, Reznek R (2001) MRI of pancreatic neuroendocrine tumours. The British journal of radiology 74 (886):968-973
Kim JH, Eun HW, Kim YJ, Han JK, Choi BI (2013) Staging accuracy of MR for pancreatic neuroendocrine tumor and imaging findings according to the tumor grade. Abdominal imaging 38 (5):1106-1114
Metz DC, Jensen RT (2008) Gastrointestinal neuroendocrine tumors: pancreatic endocrine tumors. Gastroenterology 135 (5):1469-1492
Guo J, Zhao J, Bi X, Li Z, Huang Z, Zhang Y, Cai J, Zhao H (2017) Should surgery be conducted for small nonfunctioning pancreatic neuroendocrine tumors: a systematic review. Oncotarget 8 (21):35368-35375. https://doi.org/10.18632/oncotarget.15685
Raman SP, Hruban RH, Cameron JL, Wolfgang CL, Fishman EK (2012) Pancreatic imaging mimics: part 2, pancreatic neuroendocrine tumors and their mimics. AJR Am J Roentgenol 199 (2):309-318. https://doi.org/10.2214/ajr.12.8627
Foster DS, Jensen R, Norton JA (2018) Management of liver neuroendocrine tumors in 2018. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.3035
Ito T, Igarashi H, Uehara H, Berna MJ, Jensen RT (2013) Causes of death and prognostic factors in multiple endocrine neoplasia type 1: a prospective study: comparison of 106 MEN1/Zollinger-Ellison syndrome patients with 1613 literature MEN1 patients with or without pancreatic endocrine tumors. Medicine (Baltimore) 92 (3):135-181. https://doi.org/10.1097/md.0b013e3182954af1
Reznek RH (2006) CT/MRI of neuroendocrine tumours. Cancer Imaging 6 (Spec No A):S163-S177. https://doi.org/10.1102/1470-7330.2006.9037
Lipinski M, Rydzewska G, Foltyn W, Andrysiak-Mamos E, Baldys-Waligorska A, Bednarczuk T, Blicharz-Dorniak J, Bolanowski M, Boratyn-Nowicka A, Borowska M, Cichocki A, Cwikla JB, Falconi M, Handkiewicz-Junak D, Hubalewska-Dydejczyk A, Jarzab B, Junik R, Kajdaniuk D, Kaminski G, Kolasinska-Cwikla A, Kowalska A, Krol R, Krolicki L, Kunikowska J, Kusnierz K, Lampe P, Lange D, Lewczuk-Myslicka A, Lewinski A, Londzin-Olesik M, Marek B, Nasierowska-Guttmejer A, Nowakowska-Dulawa E, Pilch-Kowalczyk J, Poczkaj K, Rosiek V, Ruchala M, Sieminska L, Sowa-Staszczak A, Starzynska T, Steinhof-Radwanska K, Strzelczyk J, Sworczak K, Syrenicz A, Szawlowski A, Szczepkowski M, Wachula E, Zajecki W, Zemczak A, Zgliczynski W, Kos-Kudla B (2017) Gastroduodenal neuroendocrine neoplasms, including gastrinoma - management guidelines (recommended by the Polish Network of Neuroendocrine Tumours). Endokrynol Pol 68 (2):138-153. https://doi.org/10.5603/ep.2017.0016
O’Toole D, Salazar R, Falconi M, Kaltsas G, Couvelard A, de Herder WW, Hyrdel R, Nikou G, Krenning E, Vullierme M-P (2006) Rare functioning pancreatic endocrine tumors. Neuroendocrinology 84 (3):189-195
Lo GC, Kambadakone A (2018) MR Imaging of Pancreatic Neuroendocrine Tumors. Magn Reson Imaging Clin N Am 26 (3):391-403. https://doi.org/10.1016/j.mric.2018.03.010
Lee NJ, Hruban RH, Fishman EK (2018) Pancreatic neuroendocrine tumor: review of heterogeneous spectrum of CT appearance. Abdom Radiol (NY) 43 (11):3025-3034. https://doi.org/10.1007/s00261-018-1574-4
Bushnell DL, Baum RP (2011) Standard imaging techniques for neuroendocrine tumors. Endocrinol Metab Clin North Am 40 (1):153-162, ix. https://doi.org/10.1016/j.ecl.2010.12.002
Sundin A, Garske U, Örlefors H Nuclear imaging of neuroendocrine tumours. Best Practice & Research Clinical Endocrinology & Metabolism 21 (1):69-85. https://doi.org/10.1016/j.beem.2006.12.003
Sun H, Zhou J, Liu K, Shen T, Wang X, Wang X (2018) Pancreatic neuroendocrine tumors: MR imaging features preoperatively predict lymph node metastasis. Abdom Radiol (NY). https://doi.org/10.1007/s00261-018-1863-y
Dromain C, Deandreis D, Scoazec JY, Goere D, Ducreux M, Baudin E, Tselikas L (2016) Imaging of neuroendocrine tumors of the pancreas. Diagn Interv Imaging 97 (12):1241-1257. https://doi.org/10.1016/j.diii.2016.07.012
Boninsegna L, Partelli S, D’Innocenzio MM, Capelli P, Scarpa A, Bassi C, Pederzoli P, Falconi M (2010) Pancreatic cystic endocrine tumors: a different morphological entity associated with a less aggressive behavior. Neuroendocrinology 92 (4):246-251. https://doi.org/10.1159/000318771
Singhi AD, Chu LC, Tatsas AD, Shi C, Ellison TA, Fishman EK, Kawamoto S, Schulick RD, Wolfgang CL, Hruban RH, Edil BH (2012) Cystic Pancreatic Neuroendocrine Tumors: A Clinicopathologic Study. The American Journal of Surgical Pathology 36 (11):1666-1673. https://doi.org/10.1097/pas.0b013e31826a0048
De Robertis R, Maris B, Cardobi N, Tinazzi Martini P, Gobbo S, Capelli P, Ortolani S, Cingarlini S, Paiella S, Landoni L, Butturini G, Regi P, Scarpa A, Tortora G, D’Onofrio M (2018) Can histogram analysis of MR images predict aggressiveness in pancreatic neuroendocrine tumors? Eur Radiol. https://doi.org/10.1007/s00330-017-5236-7
Buetow PC, Buck JL, Pantongrag-Brown L, Beck KG, Ros PR, Adair CF (1996) Solid and papillary epithelial neoplasm of the pancreas: imaging-pathologic correlation on 56 cases. Radiology 199 (3):707-711
Semelka RC, Ascher SM (1993) MR imaging of the pancreas. Radiology 188 (3):593-602
Sureka B, Meena V, Khera PS (2018) Differential diagnosis of pancreatic calcifications. American Journal of Roentgenology 210 (1):W43-W43
Javadi S, Menias CO, Korivi BR, Shaaban AM, Patnana M, Alhalabi K, Elsayes KM (2017) Pancreatic calcifications and calcified pancreatic masses: pattern recognition approach on CT. American Journal of Roentgenology 209 (1):77-87
Singh R, Calhoun S, Shin M, Katz R (2008) Pancreatic Neuroendocrine Tumor with Atypical Radiologic Presentation. Radiol Case Rep 3 (3):162. https://doi.org/10.2484/rcr.v3i3.162
Cescato R, Schulz S, Waser B, Eltschinger V, Rivier JE, Wester HJ, Culler M, Ginj M, Liu Q, Schonbrunn A, Reubi JC (2006) Internalization of sst2, sst3, and sst5 receptors: effects of somatostatin agonists and antagonists. J Nucl Med 47 (3):502-511
Graham MM, Gu X, Ginader T, Breheny P, Sunderland JJ (2017) (68)Ga-DOTATOC Imaging of Neuroendocrine Tumors: A Systematic Review and Metaanalysis. J Nucl Med 58 (9):1452-1458. https://doi.org/10.2967/jnumed.117.191197
Reubi JC, Waser B, Friess H, Büchler M, Laissue J (1998) Neurotensin receptors: a new marker for human ductal pancreatic adenocarcinoma. Gut 42 (4):546
Stabin MG, Kooij PP, Bakker WH, Inoue T, Endo K, Coveney J, de Jong R, Minegishi A (1997) Radiation dosimetry for indium-111-pentetreotide. J Nucl Med 38 (12):1919-1922
Sandstrom M, Velikyan I, Garske-Roman U, Sorensen J, Eriksson B, Granberg D, Lundqvist H, Sundin A, Lubberink M (2013) Comparative biodistribution and radiation dosimetry of 68Ga-DOTATOC and 68Ga-DOTATATE in patients with neuroendocrine tumors. J Nucl Med 54 (10):1755-1759. https://doi.org/10.2967/jnumed.113.120600
Werner RA, Bluemel C, Allen-Auerbach MS, Higuchi T, Herrmann K (2015) 68 Gallium-and 90 Yttrium-/177 Lutetium:“theranostic twins” for diagnosis and treatment of NETs. Annals of nuclear medicine 29 (1):1-7
Kabasakal L, Demirci E, Ocak M, Decristoforo C, Araman A, Ozsoy Y, Uslu I, Kanmaz B (2012) Comparison of (6)(8)Ga-DOTATATE and (6)(8)Ga-DOTANOC PET/CT imaging in the same patient group with neuroendocrine tumours. Eur J Nucl Med Mol Imaging 39 (8):1271-1277. https://doi.org/10.1007/s00259-012-2123-y
Fani M, Nicolas GP, Wild D (2017) Somatostatin receptor antagonists for imaging and therapy. J Nucl Med 58 (Suppl 2):61S-66S
Kauhanen S, Seppanen M, Nuutila P (2008) Premedication with carbidopa masks positive finding of insulinoma and beta-cell hyperplasia in [(18)F]-dihydroxy-phenyl-alanine positron emission tomography. J Clin Oncol 26 (32):5307-5308; author reply 5308-5309. https://doi.org/10.1200/jco.2008.18.8581
Halbrook CJ, Lyssiotis CA (2017) Employing Metabolism to Improve the Diagnosis and Treatment of Pancreatic Cancer. Cancer Cell 31 (1):5-19. https://doi.org/10.1016/j.ccell.2016.12.006
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324 (5930):1029-1033. https://doi.org/10.1126/science.1160809
Binderup T, Knigge U, Loft A, Federspiel B, Kjaer A (2010) 18F-fluorodeoxyglucose positron emission tomography predicts survival of patients with neuroendocrine tumors. Clin Cancer Res 16 (3):978-985. https://doi.org/10.1158/1078-0432.ccr-09-1759
Hindie E (2017) The NETPET Score: Combining FDG and Somatostatin Receptor Imaging for Optimal Management of Patients with Metastatic Well-Differentiated Neuroendocrine Tumors. Theranostics 7 (5):1159-1163. https://doi.org/10.7150/thno.19588
Bucau M, Laurent-Bellue A, Poté N, Hentic O, Cros J, Mikail N, Rebours V, Ruszniewski P, Lebtahi R, Couvelard A (2018) 18F-FDG Uptake in Well-Differentiated Neuroendocrine Tumors Correlates with Both Ki-67 and VHL Pathway Inactivation. Neuroendocrinology 106 (3):274-282
Zimny M, Bares R, Fass J, Adam G, Cremerius U, Dohmen B, Klever P, Sabri O, Schumpelick V, Buell U (1997) Fluorine-18 fluorodeoxyglucose positron emission tomography in the differential diagnosis of pancreatic carcinoma: a report of 106 cases. European journal of nuclear medicine 24 (6):678-682
Korner M, Stockli M, Waser B, Reubi JC (2007) GLP-1 receptor expression in human tumors and human normal tissues: potential for in vivo targeting. J Nucl Med 48 (5):736-743. https://doi.org/10.2967/jnumed.106.038679
Sowa-Staszczak A, Pach D, Mikolajczak R, Macke H, Jabrocka-Hybel A, Stefanska A, Tomaszuk M, Janota B, Gilis-Januszewska A, Malecki M, Kaminski G, Kowalska A, Kulig J, Matyja A, Osuch C, Hubalewska-Dydejczyk A (2013) Glucagon-like peptide-1 receptor imaging with [Lys40(Ahx-HYNIC- 99mTc/EDDA)NH2]-exendin-4 for the detection of insulinoma. Eur J Nucl Med Mol Imaging 40 (4):524-531. https://doi.org/10.1007/s00259-012-2299-1
Sowa-Staszczak A, Trofimiuk-Müldner M, Stefańska A, Tomaszuk M, Buziak-Bereza M, Gilis-Januszewska A, Jabrocka-Hybel A, Głowa B, Małecki M, Bednarczuk T (2016) 99mTc labeled glucagon-like peptide-1-analogue (99mTc-GLP1) scintigraphy in the management of patients with occult insulinoma. PloS one 11 (8):e0160714
Christ E, Wild D, Ederer S, Béhé M, Nicolas G, Caplin ME, Brändle M, Clerici T, Fischli S, Stettler C (2013) Glucagon-like peptide-1 receptor imaging for the localisation of insulinomas: a prospective multicentre imaging study. The Lancet Diabetes & Endocrinology 1 (2):115-122
Doppman JL, Miller DL, Chang R, Shawker TH, Gorden P, Norton JA (1991) Insulinomas: localization with selective intraarterial injection of calcium. Radiology 178 (1):237-241
Braatvedt G, Jennison E, Holdaway IM (2014) Comparison of two low-dose calcium infusion schedules for localization of insulinomas by selective pancreatic arterial injection with hepatic venous sampling for insulin. Clinical endocrinology 80 (1):80-84
Kato M, Doi R, Imamura M, Furutani M, Hosotani R, Shimada Y (1997) Calcium-evoked insulin release from insulinoma cells is mediated via calcium-sensing receptor. Surgery 122 (6):1203-1211
Itami A, Kato M, Komoto I, Doi R, Hosotani R, Shimada Y, Imamura M (2001) Human gastrinoma cells express calcium-sensing receptor. Life Sciences 70 (2):119-129. https://doi.org/10.1016/s0024-3205(01)01380-7
Morganstein D, Lewis D, Jackson J, Isla A, Lynn J, Devendra D, Meeran K, Todd J (2009) The role of arterial stimulation and simultaneous venous sampling in addition to cross-sectional imaging for localisation of biochemically proven insulinoma. Eur Radiol 19 (10):2467-2473
Yeh R, Dercle L, Garg I, Wang ZJ, Hough DM, Goenka AH (2018) The Role of 18F-FDG PET/CT and PET/MRI in Pancreatic Ductal Adenocarcinoma. Abdominal Radiology 43 (2):415-434
Attenberger U, Catana C, Chandarana H, Catalano OA, Friedman K, Schonberg SA, Thrall J, Salvatore M, Rosen BR, Guimaraes AR (2015) Whole-body FDG PET-MR oncologic imaging: pitfalls in clinical interpretation related to inaccurate MR-based attenuation correction. Abdominal Imaging 40 (6):1374-1386. https://doi.org/10.1007/s00261-015-0455-3
Kamisawa T, Takum K, Anjiki H, Egawa N, Kurata M, Honda G, Tsuruta K (2010) FDG-PET/CT findings of autoimmune pancreatitis. Hepato-gastroenterology 57 (99-100):447-450
Kato K, Nihashi T, Ikeda M, Abe S, Iwano S, Itoh S, Shimamoto K, Naganawa S (2013) Limited Efficacy of 18F-FDG PET/CT for Differentiation Between Metastasis-Free Pancreatic Cancer and Mass-Forming Pancreatitis. Clinical Nuclear Medicine 38 (6):417. https://doi.org/10.1097/rlu.0b013e3182817d9d
Matsumoto I, Shirakawa S, Shinzeki M, Asari S, Goto T, Ajiki T, Fukumoto T, Kitajima K, Ku Y (2013) 18-Fluorodeoxyglucose Positron Emission Tomography Does Not Aid in Diagnosis of Pancreatic Ductal Adenocarcinoma. Clinical Gastroenterology and Hepatology 11 (6):712-718. https://doi.org/10.1016/j.cgh.2012.12.033
Rijkers AP, Valkema R, Duivenvoorden HJ, van Eijck CHJ (2014) Usefulness of F-18-fluorodeoxyglucose positron emission tomography to confirm suspected pancreatic cancer: A meta-analysis. European Journal of Surgical Oncology (EJSO) 40 (7):794-804. https://doi.org/10.1016/j.ejso.2014.03.016
Beiderwellen K, Geraldo L, Ruhlmann V, Heusch P, Gomez B, Nensa F, Umutlu L, Lauenstein TC (2015) Accuracy of [18F]FDG PET/MRI for the Detection of Liver Metastases. PLoS One 10 (9):e0137285. https://doi.org/10.1371/journal.pone.0137285
Joo I, Lee JM, Lee DH, Lee ES, Paeng JC, Lee SJ, Jang JY, Kim SW, Ryu JK, Lee KB (2017) Preoperative Assessment of Pancreatic Cancer with FDG PET/MR Imaging versus FDG PET/CT Plus Contrast-enhanced Multidetector CT: A Prospective Preliminary Study. Radiology 282 (1):149-159. https://doi.org/10.1148/radiol.2016152798
Schaarschmidt BM, Grueneisen J, Heusch P, Gomez B, Umutlu L, Ruhlmann V, Rosenbaum-Krumme S, Antoch G, Buchbender C (2015) Does 18F-FDG PET/MRI reduce the number of indeterminate abdominal incidentalomas compared with 18F-FDG PET/CT? Nucl Med Commun 36 (6):588-595. https://doi.org/10.1097/mnm.0000000000000298
Chen BB, Tien YW, Chang MC, Cheng MF, Chang YT, Yang SH, Wu CH, Kuo TC, Shih IL, Yen RF, Shih TT (2018) Multiparametric PET/MR imaging biomarkers are associated with overall survival in patients with pancreatic cancer. Eur J Nucl Med Mol Imaging 45 (7):1205-1217. https://doi.org/10.1007/s00259-018-3960-0
Dercle L, Deandreis D, Terroir M, Leboulleux S, Lumbroso J, Schlumberger M (2016) Evaluation of 124 I PET/CT and 124 I PET/MRI in the management of patients with differentiated thyroid cancer. European journal of nuclear medicine and molecular imaging 43 (6):1006-1010
Kolbitsch C, Neji R, Fenchel M, Mallia A, Marsden P, Schaeffter T (2018) Fully integrated 3D high-resolution multicontrast abdominal PET-MR with high scan efficiency. Magn Reson Med 79 (2):900-911. https://doi.org/10.1002/mrm.26757
Fuin N, Catalano OA, Scipioni M, Canjels LPW, Izquierdo-Garcia D, Pedemonte S, Catana C (2018) Concurrent Respiratory Motion Correction of Abdominal PET and Dynamic Contrast-Enhanced-MRI Using a Compressed Sensing Approach. J Nucl Med 59 (9):1474-1479. https://doi.org/10.2967/jnumed.117.203943
Yang J, Liu J, Wiesinger F, Menini A, Zhu X, Hope TA, Seo Y, Larson PEZ (2018) Developing an efficient phase-matched attenuation correction method for quiescent period PET in abdominal PET/MRI. Phys Med Biol 63 (18):185002. https://doi.org/10.1088/1361-6560/aada26
Wagenknecht G, Kaiser HJ, Mottaghy FM, Herzog H (2013) MRI for attenuation correction in PET: methods and challenges. MAGMA 26 (1):99-113. https://doi.org/10.1007/s10334-012-0353-4
Keereman V, Mollet P, Berker Y, Schulz V, Vandenberghe S (2013) Challenges and current methods for attenuation correction in PET/MR. MAGMA 26 (1):81-98. https://doi.org/10.1007/s10334-012-0334-7
Visvikis D, Monnier F, Bert J, Hatt M, Fayad H (2014) PET/MR attenuation correction: where have we come from and where are we going? Eur J Nucl Med Mol Imaging 41 (6):1172-1175. https://doi.org/10.1007/s00259-014-2748-0
Attenberger U, Catana C, Chandarana H, Catalano OA, Friedman K, Schonberg SA, Thrall J, Salvatore M, Rosen BR, Guimaraes AR (2015) Whole-body FDG PET-MR oncologic imaging: pitfalls in clinical interpretation related to inaccurate MR-based attenuation correction. Abdom Imaging 40 (6):1374-1386. https://doi.org/10.1007/s00261-015-0455-3
Martinez-Moller A, Souvatzoglou M, Delso G, Bundschuh RA, Chefd’hotel C, Ziegler SI, Navab N, Schwaiger M, Nekolla SG (2009) Tissue classification as a potential approach for attenuation correction in whole-body PET/MRI: evaluation with PET/CT data. J Nucl Med 50 (4):520-526. https://doi.org/10.2967/jnumed.108.054726
Jeong JH, Cho IH, Kong EJ, Chun KA (2014) Evaluation of Dixon Sequence on Hybrid PET/MR Compared with Contrast-Enhanced PET/CT for PET-Positive Lesions. Nucl Med Mol Imaging 48 (1):26-32. https://doi.org/10.1007/s13139-013-0240-6
Dercle L, Lu L, Lichtenstein P, Yang H, Wang D, Zhu J, Wu F, Piessevaux H, Schwartz LH, Zhao B (2017) Impact of Variability in Portal Venous Phase Acquisition Timing in Tumor Density Measurement and Treatment Response Assessment: Metastatic Colorectal Cancer as a Paradigm. JCO Clinical Cancer Informatics (1):1-8. https://doi.org/10.1200/cci.17.00108
Dercle L, Chisin R, Ammari S, Gillebert Q, Ouali M, Jaudet C, Delord JP, Dierickx L, Zerdoud S, Schlumberger M, Courbon F (2014) Nonsurgical giant cell tumour of the tendon sheath or of the diffuse type: Are MRI or F-FDG PET/CT able to provide an accurate prediction of long-term outcome? Eur J Nucl Med Mol Imaging. https://doi.org/10.1007/s00259-014-2938-9
Dercle L, Ammari S, Bateson M, Durand PB, Haspinger E, Massard C, Jaudet C, Varga A, Deutsch E, Soria JC, Ferte C (2017) Limits of radiomic-based entropy as a surrogate of tumor heterogeneity: ROI-area, acquisition protocol and tissue site exert substantial influence. Sci Rep 7 (1):7952. https://doi.org/10.1038/s41598-017-08310-5
Dierickx LO, Dercle L, Chaltiel L, Caselles O, Brillouet S, Zerdoud S, Courbon F (2017) Evaluation of 2 diuretic 18Fluorine-Fluorodeoxyglucose positron emission tomography/computed tomography imaging protocols for intra-pelvic cancer. Q J Nucl Med Mol Imaging. https://doi.org/10.23736/s1824-4785.17.02912-0
Sun R, Limkin EJ, Dercle L, Reuze S, Zacharaki EI, Chargari C, Schernberg A, Dirand AS, Alexis A, Paragios N, Deutsch E, Ferte C, Robert C (2017) [Computational medical imaging (radiomics) and potential for immuno-oncology]. Cancer Radiother 21 (6-7):648-654. https://doi.org/10.1016/j.canrad.2017.07.035
Dercle L, Hartl D, Rozenblum-Beddok L, Mokrane FZ, Seban RD, Yeh R, Bidault F, Ammari S (2018) Diagnostic and prognostic value of 18F-FDG PET, CT, and MRI in perineural spread of head and neck malignancies. Eur Radiol 28 (4):1761-1770. https://doi.org/10.1007/s00330-017-5063-x
Chi W, Warner RRP, Chan DL, Singh S, Segelov E, Strosberg J, Wisnivesky J, Kim MK (2018) Long-term Outcomes of Gastroenteropancreatic Neuroendocrine Tumors. Pancreas 47 (3):321. https://doi.org/10.1097/mpa.0000000000001005
Patel BN, Olcott E, Jeffrey RB (2018) Extrapancreatic perineural invasion in pancreatic adenocarcinoma. Abdom Radiol (NY) 43 (2):323-331. https://doi.org/10.1007/s00261-017-1343-9
De Robertis R, Paiella S, Cardobi N, Landoni L, Tinazzi Martini P, Ortolani S, De Marchi G, Gobbo S, Giardino A, Butturini G, Tortora G, Bassi C, D’Onofrio M (2018) Tumor thrombosis: a peculiar finding associated with pancreatic neuroendocrine neoplasms. A pictorial essay. Abdom Radiol (NY) 43 (3):613-619. https://doi.org/10.1007/s00261-017-1243-z
Kulke MH, Anthony LB, Bushnell DL, De Herder WW, Goldsmith SJ, Klimstra DS, Marx SJ, Pasieka JL, Pommier RF, Yao JC (2010) NANETS treatment guidelines: well-differentiated neuroendocrine tumors of the stomach and pancreas. Pancreas 39 (6):735
Sundin A, Arnold R, Baudin E, Cwikla JB, Eriksson B, Fanti S, Fazio N, Giammarile F, Hicks RJ, Kjaer A, Krenning E, Kwekkeboom D, Lombard-Bohas C, O’Connor JM, O’Toole D, Rockall A, Wiedenmann B, Valle JW, Vullierme M-P, all other Antibes Consensus Conference p (2017) ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: Radiological, Nuclear Medicine and Hybrid Imaging. Neuroendocrinology 105 (3):212-244. https://doi.org/10.1159/000471879
Schraml C, Schwenzer NF, Sperling O, Aschoff P, Lichy MP, Muller M, Brendle C, Werner MK, Claussen CD, Pfannenberg C (2013) Staging of neuroendocrine tumours: comparison of [(6)(8)Ga]DOTATOC multiphase PET/CT and whole-body MRI. Cancer Imaging 13:63-72. https://doi.org/10.1102/1470-7330.2013.0007
Yu F, Venzon DJ, Serrano J, Goebel SU, Doppman JL, Gibril F, Jensen RT (1999) Prospective study of the clinical course, prognostic factors, causes of death, and survival in patients with long-standing Zollinger-Ellison syndrome. J Clin Oncol 17 (2):615-630. https://doi.org/10.1200/jco.1999.17.2.615
Jensen RT, Niederle B, Mitry E, Ramage JK, Steinmuller T, Lewington V, Scarpa A, Sundin A, Perren A, Gross D, O’Connor JM, Pauwels S, Kloppel G, Frascati Consensus C, European Neuroendocrine Tumor S (2006) Gastrinoma (duodenal and pancreatic). Neuroendocrinology 84 (3):173-182. https://doi.org/10.1159/000098009
Ellison EC, Johnson JA (2009) The Zollinger-Ellison syndrome: a comprehensive review of historical, scientific, and clinical considerations. Current problems in surgery 46 (1):13-106
d’Assignies G, Fina P, Bruno O, Vullierme MP, Tubach F, Paradis V, Sauvanet A, Ruszniewski P, Vilgrain V (2013) High sensitivity of diffusion-weighted MR imaging for the detection of liver metastases from neuroendocrine tumors: comparison with T2-weighted and dynamic gadolinium-enhanced MR imaging. Radiology 268 (2):390-399. https://doi.org/10.1148/radiol.13121628
Pavel M, Baudin E, Couvelard A, Krenning E, Oberg K, Steinmuller T, Anlauf M, Wiedenmann B, Salazar R, Barcelona Consensus Conference p (2012) ENETS Consensus Guidelines for the management of patients with liver and other distant metastases from neuroendocrine neoplasms of foregut, midgut, hindgut, and unknown primary. Neuroendocrinology 95 (2):157-176. https://doi.org/10.1159/000335597
Hope TA, Bergsland EK, Bozkurt MF, Graham M, Heaney AP, Herrmann K, Howe JR, Kulke MH, Kunz PL, Mailman J, May L, Metz DC, Millo C, O’Dorisio S, Reidy-Lagunes DL, Soulen MC, Strosberg JR (2018) Appropriate Use Criteria for Somatostatin Receptor PET Imaging in Neuroendocrine Tumors. J Nucl Med 59 (1):66-74. https://doi.org/10.2967/jnumed.117.202275
Orditura M, Petrillo A, Ventriglia J, Diana A, Laterza MM, Fabozzi A, Savastano B, Franzese E, Conzo G, Santini L, Ciardiello F, De Vita F (2016) Pancreatic neuroendocrine tumors: Nosography, management and treatment. Int J Surg 28 Suppl 1:S156-162. https://doi.org/10.1016/j.ijsu.2015.12.052
Dromain C, de Baere T, Lumbroso J, Caillet H, Laplanche A, Boige V, Ducreux M, Duvillard P, Elias D, Schlumberger M, Sigal R, Baudin E (2005) Detection of liver metastases from endocrine tumors: a prospective comparison of somatostatin receptor scintigraphy, computed tomography, and magnetic resonance imaging. J Clin Oncol 23 (1):70-78. https://doi.org/10.1200/jco.2005.01.013
de Baère T, Aupérin A, Deschamps F, Chevallier P, Gaubert Y, Boige V, Fonck M, Escudier B, Palussiére J (2015) Radiofrequency ablation is a valid treatment option for lung metastases: experience in 566 patients with 1037 metastases. Annals of Oncology 26 (5):987-991. https://doi.org/10.1093/annonc/mdv037
Eriksson J, Stålberg P, Nilsson A, Krause J, Lundberg C, Skogseid B, Granberg D, Eriksson B, Åkerström G, Hellman P (2008) Surgery and Radiofrequency Ablation for Treatment of Liver Metastases from Midgut and Foregut Carcinoids and Endocrine Pancreatic Tumors. World J Surg 32 (5):930-938. https://doi.org/10.1007/s00268-008-9510-3
de Baere T, Palussiere J, Auperin A, Hakime A, Abdel-Rehim M, Kind M, Dromain C, Ravaud A, Tebboune N, Boige V, Malka D, Lafont C, Ducreux M (2006) Midterm local efficacy and survival after radiofrequency ablation of lung tumors with minimum follow-up of 1 year: prospective evaluation. Radiology 240 (2):587-596. https://doi.org/10.1148/radiol.2402050807
Kennedy A, Bester L, Salem R, Sharma RA, Parks RW, Ruszniewski P (2015) Role of hepatic intra-arterial therapies in metastatic neuroendocrine tumours (NET): guidelines from the NET-Liver-Metastases Consensus Conference. HPB (Oxford) 17 (1):29-37. https://doi.org/10.1111/hpb.12326
Vogl TJ, Gruber T, Naguib NNN, Hammerstingl R, Nour-Eldin N-EA (2009) Liver metastases of neuroendocrine tumors: treatment with hepatic transarterial chemotherapy using two therapeutic protocols. American Journal of Roentgenology 193 (4):941-947
Funding
L Dercle’s work was funded by a grant from Fondation Philanthropia, Geneva, Switzerland, and the Fondation Nuovo-Soldati.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rozenblum, L., Mokrane, FZ., Yeh, R. et al. The role of multimodal imaging in guiding resectability and cytoreduction in pancreatic neuroendocrine tumors: focus on PET and MRI. Abdom Radiol 44, 2474–2493 (2019). https://doi.org/10.1007/s00261-019-01994-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-019-01994-5