Abstract
Purpose
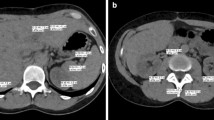
To evaluate the role of virtual monoenergetic imaging (VMI) in the detection of peritoneal metastatic disease in contrast-enhanced computed tomography (CT) of the abdomen and pelvis and to compare this technique to the conventional 120 kV mixed dataset.
Materials and methods
Institutional review board approval was obtained with no informed consent required for this retrospective analysis. 43 consecutive patients with histopathologically confirmed peritoneal disease were scanned using a standard protocol on a 128-section dual-source, dual-energy CT system (100/140 keV). Scans were retrospectively reconstructed at VMI energy levels from 40–110 keV in 10 keV increments and were analyzed both quantitatively and qualitatively. CNR values for peritoneal metastatic deposits were recorded using region of interest (ROI) analysis at each energy level for all VMI datasets. Subjective analysis was performed by two independent fellowship-trained readers with combined experience of greater than 15 years. Qualitative parameters included diagnostic acceptability, subjective noise, and contrast resolution and confidence.
Results
The contrast-to-noise ratios (CNRs) for peritoneal metastatic deposits at the different VMI energy levels were compared using a one-way ANOVA with Tukey Post Test, and the optimal CNR was observed at 40 keV (p < 0.0001). Qualitative parameters were compared using a Paired T Test. Subjective noise, diagnostic acceptability, and contrast resolution was significantly better on the conventional images, but readers reported increased confidence on VMI at 40 keV (p < 0.001).
Conclusion
VMI reconstruction of contrast-enhanced dual-energy CT scans of the abdomen and pelvis at 40 keV maximizes the conspicuity of metastatic peritoneal deposits and improves radiologists’ diagnostic confidence compared with conventional CT images. We recommend using virtual monoenergetic datasets at 40 keV as a tool for improving the detection of these lesions in routine clinical practice.





Similar content being viewed by others
References
Levy AD, Shaw JC, Sobin LH (2009) Secondary tumors and tumorlike lesions of the peritoneal cavity: imaging features with pathologic correlation. Radiographics 29(2):347–373
O’Dwyer S, Verwaal VJ, Sugarbaker PH (2015) Evolution of treatments for peritoneal metastases from colorectal cancer. J Clin Oncol 33(18):2122–2123
Klaver CEL, et al. (2015) Adjuvant hyperthermic intraperitoneal chemotherapy (HIPEC) in patients with colon cancer at high risk of peritoneal carcinomatosis; the COLOPEC randomized multicentre trial. BMC Cancer 15:428
Naffouje SA, Salti GI (2018) Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in elderly patients: complete cytoreduction is feasible and crucial for improved survival despite high carcinomatosis index. Anticancer Res 38(1):441–448
Huang Y, Alzahrani NA, Liauw W, et al. (2017) Early postoperative intraperitoneal chemotherapy is associated with survival benefit for appendiceal adenocarcinoma with peritoneal dissemination. Eur J Surg Oncol 43(12):2292–2298
Magge D, et al. (2017) Hyperthermic intraperitoneal chemoperfusion as a component of multimodality therapy for ovarian and primary peritoneal cancer. J Surg Oncol 116(3):320–328
Grabowska-Derlatka L, Derlatka P, Szeszkowski W, Cieszanowski A (2016) Diffusion-weighted imaging of small peritoneal implants in ‘potentially’ early-stage ovarian cancer. Biomed Res Int 2016:9254742
Fehniger J, et al. (2016) A prospective study evaluating diffusion weighted magnetic resonance imaging (DW-MRI) in the detection of peritoneal carcinomatosis in suspected gynecologic malignancies. Gynecol Oncol 142(1):169–175
Chen F-M, Ni J-M, Zhang Z-Y, et al. (2016) Presurgical evaluation of pancreatic cancer: a comprehensive imaging comparison of CT versus MRI. AJR Am J Roentgenol 206(3):526–535
Schmidt S, Meuli RA, Achtari C, Prior JO (2015) Peritoneal carcinomatosis in primary ovarian cancer staging: comparison between MDCT, MRI, and 18F-FDG PET/CT. Clin Nucl Med 40(5):371–377
Marin D, et al. (2010) Low-tube-voltage, high-tube-current multidetector abdominal CT: improved image quality and decreased radiation dose with adaptive statistical iterative reconstruction algorithm–initial clinical experience. Radiology 254(1):145–153
Silva AC, Morse BG, Hara AK, et al. (2011) Dual-energy (spectral) CT: applications in abdominal imaging. Radiographics 31(4):1031–1050
Yu L, Christner JA, Leng S, et al. (2011) Virtual monochromatic imaging in dual-source dual-energy CT: radiation dose and image quality. Med Phys 38(12):6371–6379
Yu L, Leng S, McCollough CH (2012) Dual-energy CT-based monochromatic imaging. AJR Am J Roentgenol 199(5 Suppl):S9–S15
Srinivasan A, Parker RA, Manjunathan A, et al. (2013) Differentiation of benign and malignant neck pathologies: preliminary experience using spectral computed tomography. J Comput Assist Tomogr 37(5):666–672
Shuman WP, et al. (2014) Dual-energy liver CT: effect of monochromatic imaging on lesion detection, conspicuity, and contrast-to-noise ratio of hypervascular lesions on late arterial phase. AJR Am J Roentgenol 203(3):601–606
Wang Q, Shi G, Qi X, Fan X, Wang L (2014) Quantitative analysis of the dual-energy CT virtual spectral curve for focal liver lesions characterization. Eur J Radiol 83(10):1759–1764
Hou WS, Wu HW, Yin Y, et al. (2015) Differentiation of lung cancers from inflammatory masses with dual-energy spectral CT imaging. Acad Radiol 22(3):337–344
Yuan Y, Zhang Y, Lang N, Li J, Yuan H (2015) Differentiating malignant vertebral tumours from non-malignancies with CT spectral imaging: a preliminary study. Eur Radiol 25(10):2945–2950
Huda W, Ogden KM, Khorasani MR (2008) Converting dose-length product to effective dose at CT. Radiology 248(3):995–1003
Christner JA, Kofler JM, McCollough CH (2010) Estimating effective dose for CT using dose-length product compared with using organ doses: consequences of adopting international commission on radiological protection publication 103 or dual-energy scanning. Am J Roentgenol 194(4):881–889
Bongartz G et al. (1999) European guidelines on quality criteria for computed tomography. Eur Comm.
Fu W, et al. (2017) Optimizing window settings for improved presentation of virtual monoenergetic images in dual-energy computed tomography. Med Phys 44(11):5686–5696
GraphPad Prism. https://www.graphpad.com/scientific-software/prism/. Accessed 29 July 2018.
Morgan DE (2018) The role of dual-energy computed tomography in assessment of abdominal oncology and beyond. Radiol Clin North Am 56(4):565–585
McNamara MM, Little MD, Alexander LF, et al. (2015) Multireader evaluation of lesion conspicuity in small pancreatic adenocarcinomas: complimentary value of iodine material density and low keV simulated monoenergetic images using multiphasic rapid kVp-switching dual energy CT. Abdom Imaging 40(5):1230–1240
Sodickson A, Primak A, Keraliya A. Workflow evaluation of a new fully automated dual-energy CT post-processing configuration. In: Radiological Society of North America (RSNA) Scientific Assembly and Annual Meeting
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by a 2015 Research Grant from the Canadian Radiological Foundation (CRF).
Conflict of interest
Savvas Nicolaou: Institutional research agreement, Siemens AG. All the other authors have declared that they have no competing interests.
Human ethics
Institutional review board approval (University of British Columbia) was obtained for this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent from individual participants was not required for this retrospective study as per Institutional Review Board approvals.
Rights and permissions
About this article
Cite this article
Darras, K.E., Clark, S.J., Kang, H. et al. Virtual monoenergetic reconstruction of contrast-enhanced CT scans of the abdomen and pelvis at 40 keV improves the detection of peritoneal metastatic deposits. Abdom Radiol 44, 422–428 (2019). https://doi.org/10.1007/s00261-018-1733-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1733-7