Abstract
Purpose
To compare diagnostic performance of PI-RADSv2 with ADC parameters to identify clinically significant prostate cancer (csPC) and to determine the impact of csPC definitions on diagnostic performance of ADC and PI-RADSv2.
Methods
We retrospectively identified treatment-naïve pathology-proven peripheral zone PC patients who underwent 3T prostate MRI, using high b-value diffusion-weighted imaging from 2011 to 2015. Using 3D slicer, areas of suspected tumor (T) and normal tissue (N) on ADC (b = 0, 1400) were outlined volumetrically. Mean ADCT, mean ADCN, ADCratio (ADCT/ADCN) were calculated. PI-RADSv2 was assigned. Three csPC definitions were used: (A) Gleason score (GS) ≥ 4 + 3; (B) GS ≥ 3 + 4; (C) MRI-based tumor volume >0.5 cc. Performances of ADC parameters and PI-RADSv2 in identifying csPC were measured using nonparametric comparison of receiver operating characteristic curves using the area under the curve (AUC).
Results
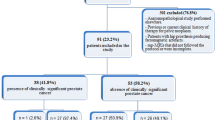
Eighty five cases met eligibility requirements. Diagnostic performances (AUC) in identifying csPC using three definitions were: (A) ADCT (0.83) was higher than PI-RADSv2 (0.65, p = 0.006); (B) ADCT (0.86) was higher than ADCratio (0.68, p < 0.001), and PI-RADSv2 (0.70, p = 0.04); (C) PI-RADSv2 (0.73) performed better than ADCratio (0.56, p = 0.02). ADCT performance was higher when csPC was defined by A or B versus C (p = 0.038 and p = 0.01, respectively). ADCratio performed better when csPC was defined by A versus C (p = 0.01). PI-RADSv2 performance was not affected by csPC definition.
Conclusions
When csPC was defined by GS, ADC parameters provided better csPC discrimination than PI-RADSv2, with ADCT providing best result. When csPC was defined by MRI-calculated volume, PI-RADSv2 provided better discrimination than ADCratio. csPC definition did not affect PI-RADSv2 diagnostic performance.

Similar content being viewed by others
References
Miller KD, Siegel RL, Lin CC, et al. (2016) Cancer treatment and survivorship statistics. CA Cancer J Clin 66(4):271–289
Loeb S, Bjurlin MA, Nicholson J, et al. (2014) Overdiagnosis and overtreatment of prostate cancer. Eur Urol 65(6):1046–1055
Etzioni R, Penson DF, Legler JM, et al. (2002) Overdiagnosis due to prostate-specific antigen screening: lessons from U.S. prostate cancer incidence trends. J Natl Cancer Inst 94(13):981–990
Ahmed HU, Hu Y, Carter T, et al. (2011) Characterizing clinically significant prostate cancer using template prostate mapping biopsy. J Urol 186(2):458–464
Rasiah KK, Stricker PD, Haynes A-M, et al. (2003) Prognostic significance of Gleason pattern in patients with Gleason score 7 prostate carcinoma. Cancer 98(12):2560–2565
Wolters T, Roobol MJ, van Leeuwen PJ, et al. (2011) A critical analysis of the tumor volume threshold for clinically insignificant prostate cancer using a data set of a randomized screening trial. J Urol 185(1):121–125
Kumar V, Jagannathan NR, Kumar R, et al. (2007) Apparent diffusion coefficient of the prostate in men prior to biopsy: determination of a cut-off value to predict malignancy of the peripheral zone. NMR Biomed 20(5):505–511
Woo S, Kim SY, Cho JY, et al. (2016) Preoperative evaluation of prostate cancer aggressiveness: using ADC and ADC ratio in determining Gleason score. AJR 207(1):114–120
Hambrock T, Somford DM, Huisman HJ, et al. (2011) Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259(2):453–461
Vargas HA, Akin O, Franiel T, et al. (2011) Diffusion-weighted endorectal MR imaging at 3 T for prostate cancer: tumor detection and assessment of aggressiveness. Radiology 259(3):775–784
Metens T, Miranda D, Absil J, et al. (2012) What is the optimal b value in diffusion-weighted MR imaging to depict prostate cancer at 3T? Eur Radiol 22(3):703–709
Kim CK, Park BK (2010) Kim B (2010) High-b-value diffusion-weighted imaging at 3 T to detect prostate cancer: comparisons between b values of 1,000 and 2,000 s/mm2. AJR 194(1):W33–W37
Weinreb JC, Barentsz JO, Choyke PL, et al. (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69(1):16–40
Bratan F, Niaf E, Melodelima C, et al. (2013) Influence of imaging and histological factors on prostate cancer detection and localisation on multiparametric MRI: a prospective study. Eur Radiol 23(7):2019–2029
Jambor I, Boström PJ, Taimen P, et al. (2017) Novel biparametric MRI and targeted biopsy improves risk stratification in men with a clinical suspicion of prostate cancer (IMPROD Trial). J Magn Reson Imaging. doi:10.1002/jmri.25641
Ahmed HU, El-Shater Bosaily A, Brown LC, et al. (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389(10071):815–822
Kozlowski P, Chang SD, Goldenberg SL (2008) Diffusion-weighted MRI in prostate cancer—comparison between single-shot fast spin echo and echo planar imaging sequences. Magn Reson Imaging 26(1):72–76
Barentsz JO, Richenberg J, Clements R, et al. (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757
McNeal JE, Redwine EA, Freiha FS, et al. (1988) Zonal distribution of prostatic adenocarcinoma. Correlation with histologic pattern and direction of spread. Am J Surg Pathol 12(12):897–906
Vargas HA, Hötker AM, Goldman DA, et al. (2016) Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: critical evaluation using whole-mount pathology as standard of reference. Eur Radiol 26(6):1606–1612
Hegde JV, Mulkern RV, Panych LP, et al. (2013) Multiparametric MRI of prostate cancer: an update on state-of-the-art techniques and their performance in detecting and localizing prostate cancer. J Magn Reson Imaging 37(5):1035–1054
Srigley JR, Humphrey PA, Amin MB, et al. (2009) Protocol for the examination of specimens from patients with carcinoma of the prostate gland. Arch Pathol Lab Med 133(10):1568–1576
Fedorov A, Beichel R, Kalpathy-Cramer J, et al. (2012) 3D Slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging 30(9):1323–1341
Fedorov A, Vangel MG, Tempany CM, Fennessy FM (2017) Multiparametric magnetic resonance imaging of the prostate: repeatability of volume and apparent diffusion coefficient quantification. Investig Radiol 52(9):538–546
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44(3):837–845
Stensland KD, Coutinho K, Hobbs AR, et al. (2016) Are magnetic resonance imaging undetectable prostate tumours clinically significant? Results of histopathological analyses. Arab J Urol 14(4):256–261
Truong M, Hollenberg G, Weinberg E, et al. (2017) Impact of Gleason subtype on prostate cancer detection using multiparametric MRI: correlation with final histopathology. J Urol. doi:10.1016/j.juro.2017.01.077
Turkbey B, Mani H, Aras O, et al. (2012) Correlation of magnetic resonance imaging tumor volume with histopathology. J Urol 188(4):1157–1163
Priester A, Natarajan S, Khoshnoodi P, et al. (2017) Magnetic resonance imaging underestimation of prostate cancer geometry: use of patient specific molds to correlate images with whole mount pathology. J Urol 197(2):320–326
Zhao C, Gao G, Fang D, et al. (2016) The efficiency of multiparametric magnetic resonance imaging (mpMRI) using PI-RADS version 2 in the diagnosis of clinically significant prostate cancer. Clin Imaging 40(5):885–888
Lin WC, Westphalen AC, Silva GE, et al. (2016) Comparison of PI-RADS 2, ADC histogram-derived parameters, and their combination for the diagnosis of peripheral zone prostate cancer. Abdom Radiol (NY) 41(11):2209–2217
Glazer DI, Hassanzadeh E, Fedorov A, et al. (2017) Diffusion-weighted endorectal MR imaging at 3T for prostate cancer: correlation with tumor cell density and percentage Gleason pattern on whole mount pathology. Abdom Radiol (NY) 42(3):918–925
Donati OF, Mazaheri Y, Afaq A, et al. (2014) Prostate cancer aggressiveness: assessment with whole-lesion histogram analysis of the apparent diffusion coefficient. Radiology 271(1):143–152
Lebovici A, Sfrangeu SA, Feier D, et al. (2014) Evaluation of the normal-to-diseased apparent diffusion coefficient ratio as an indicator of prostate cancer aggressiveness. BMC Med Imaging 14:15
Boesen L, Chabanova E, Løgager V, et al. (2015) Apparent diffusion coefficient ratio correlates significantly with prostate cancer gleason score at final pathology. J Magn Reson Imaging 42(2):446–453
Jones AC, Antillon KS, Jenkins SM, et al. (2015) Prostate field cancerization: deregulated expression of macrophage inhibitory cytokine 1 (MIC-1) and platelet derived growth factor A (PDGF-A) in tumor adjacent tissue. PLoS One 10(3):e0119314
Mulkern RV, Barnes AS, Haker SJ, et al. (2006) Biexponential characterization of prostate tissue water diffusion decay curves over an extended b-factor range. Magn Reson Imaging 24(5):563–568
Agarwal HK, Mertan FV, Sankineni S, et al. (2017) Optimal high b-value for diffusion weighted MRI in diagnosing high risk prostate cancers in the peripheral zone. J Magn Reson Imaging 45(1):125–131
Ukimura O, Coleman JA, de la Taille A, et al. (2013) Contemporary role of systematic prostate biopsies: indications, techniques, and implications for patient care. Eur Urol 63(2):214–230
Purysko AS, Bittencourt LK, Bullen JA, et al. (2017) Accuracy and interobserver agreement for prostate imaging reporting and data system, version 2, for the characterization of lesions identified on multiparametric MRI of the prostate. AJR 209(2):339–349
Acknowledgements
The authors thank all the technologists, especially Nancy Trane, RT for her dedication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by National Institute of Health – National Cancer Institute: U01 CA151261 (Fiona M Fennessy, Andriy Fedorov); R25 CA089017 (Fiona M Fennessy, Daniel I Glazer, Clare M Tempany); NIH P41 EB 015898 (Clare M Tempany, Fiona M Fennessy, Andriy Fedorov).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Hassanzadeh, E., Alessandrino, F., Olubiyi, O.I. et al. Comparison of quantitative apparent diffusion coefficient parameters with prostate imaging reporting and data system V2 assessment for detection of clinically significant peripheral zone prostate cancer. Abdom Radiol 43, 1237–1244 (2018). https://doi.org/10.1007/s00261-017-1297-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-017-1297-y