Abstract
Purpose
To develop a predictive model by 18F-FDG PET/CT radiomic features and to validate the predictive value of the model for distinguishing solitary lung adenocarcinoma from tuberculosis.
Methods
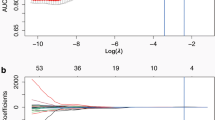
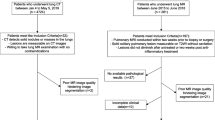
A total of 235 18F-FDG PET/CT patients with pathologically or follow-up confirmed lung adenocarcinoma (n = 131) or tuberculosis (n = 104) were retrospectively and randomly divided into a training (n = 163) and validation (n = 72) cohort. Based on the Transparent Reporting of Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD), this work was belonged to TRIPOD type 2a study. The Mann-Whitney U test and least absolute shrinkage and selection operator (LASSO) algorithm were used to select the optimal predictors from 92 radiomic features that were extracted from PET/CT, and the optimal predictors were used to build the radiomic model in the training cohort. The meaningful clinical variables comprised the clinical model, and the combination of the radiomic model and clinical model was a complex model. The performances of the models were assessed by the area under the receiver operating characteristic curve (AUC) in the training and validation cohorts.
Results
In the training cohort, 9 radiomic features were selected as optimal predictors to build the radiomic model. The AUC of the radiomic model was significantly higher than that of the clinical model in the training cohort (0.861 versus 0.686, p < 0.01), and this was similar in the validation cohort (0.889 versus 0.644, p < 0.01). The AUC of the radiomic model was slightly lower than that of the complex model in the training cohort (0.861 versus 0.884, p > 0.05) and validation cohort (0.889 versus 0.909, p > 0.05), but there was no significant difference.
Conclusion
18F-FDG PET/CT radiomic features have a significant value in differentiating solitary lung adenocarcinoma from tuberculosis.




Similar content being viewed by others
Date availability
All authors made sure that all data and materials as well as the software application supported the published claims and complied with field standards.
Code availability
All authors made sure that all the custom codes supported the published claims and complied with field standards.
References
Hsu WH, Yang JCH, Mok TS, et al. Overview of current systemic management of EGFR-mutant NSCLC. Ann Oncol. 2018;29:i3–9.
World Health Organization. Global Tuberculosis Report 2019 WHO. 2019; https://www.who.int/tb/publications/global_report/en/; Accessed 17 Oct 2019
Wang XL, Shan W. Application of dynamic CT to identify lung cancer, pulmonary tuberculosis, and pulmonary inflammatory pseudotumor. Eur Rev Med Pharmacol Sci. 2017;21:4804–9.
Qi LP, Chen KN, Zhou XJ, et al. Conventional MRI to detect the differences between mass-like tuberculosis and lung cancer. J Thorac Dis. 2018;10:5673–84.
Lim CG, Shin KM, Lim JS, et al. Predictors of conversion to thoracotomy during video-assisted thoracoscopic surgery lobectomy in lung cancer: additional predictive value of FDG-PET/CT in a tuberculosis endemic region. J Thorac Dis. 2017;9:2427–36.
Kang F, Wang S, Tian F, et al. Comparing the diagnostic potential of 68Ga-Alfatide II and 18F-FDG in differentiating between non-small cell lung cancer and tuberculosis. J Nucl Med. 2016;57:672–7.
Haas MK, Belknap RW. Diagnostic tests for latent tuberculosis infection. Clin Chest Med. 2019;40:829–37.
Tiberi S, du Plessis N, Walzl G, et al. Tuberculosis: progress and advances in development of new drugs, treatment regimens, and host-directed therapies. Lancet Infect Dis. 2018;18:e183–98.
Hatt M, Le Rest CC, Tixier F, et al. Radiomics: data are also images. J Nucl Med. 2019;60:38S–44S.
Castello A, Rossi S, Toschi L, et al. Hyper-progressive disease in patients with non-small cell lung cancer treated with checkpoint inhibitors: the role of 18F-FDG PET/CT. J Nucl Med. 2019. https://doi.org/10.2967/jnumed.119.237768.
Dissaux G, Visvikis D, Da-Ano R, et al. Pre-treatment 18F-FDG PET/CT radiomics predict local recurrence in patients treated with stereotactic radiotherapy for early-stage non-small cell lung cancer: a multicentric study. J Nucl Med. 2019. https://doi.org/10.2967/jnumed.119.228106.
Yang X, He J, Wang J, et al. CT-based radiomics signature for differentiating solitary granulomatous nodules from solid lung adenocarcinoma. Lung Cancer. 2018;125:109–14.
Wilson R, Devaraj A. Radiomics of pulmonary nodules and lung cancer. Transl Lung Cancer Res. 2017;6:86–91.
Feng B, Li Y, Guo D, et al. Research on the value of the T cell spot test for tuberculosis for the diagnosis of lung cancer combined with pulmonary tuberculosis. Thorac Cancer. 2018;9:1231–4.
Collins GS, Reitsma JB, Altman DG, et al. Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD): the TRIPOD statement. Eur Urol. 2015;67:1142–51.
Zwanenburg A, Leger S, Vallières M, Löck S, for the Image Biomarker Standardisation Initiative (IBSI). Image biomarker standardisation initiative-feature definitions. 2019 [Current version V11 2019]. https://arxiv.org/abs/1612.07003. Accessed 17 Dec 2019.
Boellaard R, Delgado-Bolton R, Oyen WJ, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–54.
Nioche C, Orlhac F, Boughdad S, et al. LIFEx: a freeware for radiomic feature calculation in multimodality imaging to accelerate advances in the characterization of tumor heterogeneity. Cancer Res. 2018;78:4786–9.
Bashir U, Azad G, Siddique MM, et al. The effects of segmentation algorithms on the measurement of F-FDG PET texture parameters in non-small cell lung cancer. EJNMMI Res. 2017;7:60.
She Y, Zhang L, Zhu H, et al. The predictive value of CT-based radiomics in differentiating indolent from invasive lung adenocarcinoma in patients with pulmonary nodules. Eur Radiol. 2018;28:5121–8.
Ter Voert E, Muehlematter UJ, Delso G, et al. Quantitative performance and optimal regularization parameter in block sequential regularized expectation maximization reconstructions in clinical 68Ga-PSMA PET/MR. EJNMMI Res. 2018;8:70.
Slifstein M, Laruelle M. Effects of statistical noise on graphic analysis of PET neuroreceptor studies. J Nucl Med. 2000;41:2083–8.
Wagner F, Hakami YA, Warnock G, et al. Comparison of contrast-enhanced CT and [F]FDG PET/CT analysis using kurtosis and skewness in patients with primary colorectal cancer. Mol Imaging Biol. 2017;19:795–803.
Chandarana H, Rosenkrantz AB, Mussi TC, et al. Histogram analysis of whole-lesion enhancement in differentiating clear cell from papillary subtype of renal cell cancer. Radiology. 2012;265:790–8.
Pantic I, Dacic S, Brkic P, et al. Application of fractal and grey level co-occurrence matrix analysis in evaluation of brain corpus callosum and cingulum architecture. Microsc Microanal. 2014;20:1373–81.
Cosma CL, Sherman DR, Ramakrishnan L. The secret lives of the pathogenic mycobacteria. Annu Rev Microbiol. 2003;57:641–76.
Lee JW, Kim BS, Lee DS, et al. 18F-FDG PET/CT in mediastinal lymph node staging of non-small-cell lung cancer in a tuberculosis-endemic country: consideration of lymph node calcification and distribution pattern to improve specificity. Eur J Nucl Med Mol Imaging. 2009;36:1794–802.
Zhang J, Zhao X, Zhao Y, et al. Value of pre-therapy 18F-FDG PET/CT radiomics in predicting EGFR mutation status in patients with non-small cell lung cancer. Eur J Nucl Med Mol Imaging. 2020;47:1137–46.
Wu J, Bai J, Wang W, et al. ATBdiscrimination: an in silico tool for identification of active tuberculosis disease based on routine blood test and T-SPOT.TB detection results. J Chem Inf Model. 2019;59:4561–8.
Kang W, Wu M, Yang K, et al. Factors associated with negative T-SPOT.TB results among smear-negative tuberculosis patients in China. Sci Rep. 2018;8:4236.
Acknowledgements
The authors would like to acknowledge all the coworkers who participated in this study.
Funding
This study was supported by the Foundation of Science and Technology Department of Hebei Province, China (grant number 15277776D).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Xinming Zhao, Yujing Hu, Jianyuan Zhang, Jingya Han and Meng Dai. The first draft of the manuscript was written by Yujing Hu, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
All procedures performed in the study and involving human participants were carried out in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The study was approved by the Institutional Review Board of the Fourth Hospital of Hebei Medical University.
Informed consent
This retrospective analysis was approved by the Institutional Review Board of the Fourth Hospital of Hebei Medical University, and the requirement of informed consent was waived.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – Chest
Rights and permissions
About this article
Cite this article
Hu, Y., Zhao, X., Zhang, J. et al. Value of 18F-FDG PET/CT radiomic features to distinguish solitary lung adenocarcinoma from tuberculosis. Eur J Nucl Med Mol Imaging 48, 231–240 (2021). https://doi.org/10.1007/s00259-020-04924-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-020-04924-6