Abstract
Purpose
68Ga-FAPI-04 is a rapidly evolving PET tracer for whole-body imaging in a variety of cancers. We aimed to evaluate the diagnostic performance of 68Ga-FAPI-04 for detecting and characterizing hepatic nodules in patients with suspected carcinoma.
Methods
Twenty-five patients showing suspicious hepatic lesions for malignancy underwent 68Ga-FAPI-04 PET. The maximum and mean standardised uptake values (SUVmax, SUVmean) were measured for all detected lesions and normal hepatic tissues, respectively. The target-to-background ratio (TBR) was calculated by dividing the lesion SUVmax with the SUVmean of non-tumour liver tissue. Lesion uptake value was correlated with the in vitro hepatic FAP expression determined by immunohistochemistry (IHC).
Results
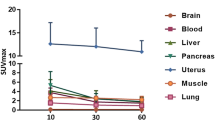
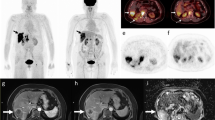
In total, 17 patients who underwent surgery or biopsy were recruited for the final analysis. A total of 28 intrahepatic malignant lesions were detected in 16 patients; the mean SUVmax was 8.36 ± 4.21 (range 2.21 to 15.86), and mean TBR was 13.15 ± 9.48 (range 2.79 to 38.12) in all detected lesions (n = 28). One benign patient showed negligible hepatic uptake (SUVmax = 0.47), whereas 75% of the primary intrahepatic hepatocellular carcinoma (HCC) lesions (n = 6) showed prominent FAP expression, 12.5% of the lesions (n = 1) showed moderate expression in stromal cells, and one showed negligible expression.
Conclusions
68Ga-FAPI-04 showed high sensitivity in detecting hepatic malignancies, particularly in poorly differentiated forms with concordantly elevated FAP expression.






Similar content being viewed by others
References
Alsaleh M, Leftley Z, Barbera TA, Sithithaworn P, Khuntikeo N, Loilome W, et al. Cholangiocarcinoma: a guide for the nonspecialist. Int J Gen Med. 2019;12:13–23. https://doi.org/10.2147/ijgm.S186854.
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301–14. https://doi.org/10.1016/s0140-6736(18)30010-2.
Mertens JC, Rizvi S, Gores GJ. Targeting cholangiocarcinoma. Biochim Biophys Acta Mol Basis Dis. 1864;2018:1454–60. https://doi.org/10.1016/j.bbadis.2017.08.027.
Levy MT, McCaughan GW, Abbott CA, Park JE, Cunningham AM, Muller E, et al. Fibroblast activation protein: a cell surface dipeptidyl peptidase and gelatinase expressed by stellate cells at the tissue remodelling interface in human cirrhosis. Hepatology. 1999;29:1768–78. https://doi.org/10.1002/hep.510290631.
Affo S, Yu LX, Schwabe RF. The role of cancer-associated fibroblasts and fibrosis in liver cancer. Annu Rev Pathol. 2017;12:153–86. https://doi.org/10.1146/annurev-pathol-052016-100322.
Zou B, Liu X, Zhang B, Gong Y, Cai C, Li P, et al. The expression of FAP in hepatocellular carcinoma cells is induced by hypoxia and correlates with poor clinical outcomes. J Cancer. 2018;9:3278–86. https://doi.org/10.7150/jca.25775.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60:801–5. https://doi.org/10.2967/jnumed.119.227967.
Clinical Practice Guidelines EASL. Management of hepatocellular carcinoma. J Hepatol. 2018;69:182–236. https://doi.org/10.1016/j.jhep.2018.03.019.
Kesler M, Levine C, Hershkovitz D, Mishani E, Menachem Y, Lerman H, et al. (68)Ga-PSMA is a novel PET-CT tracer for imaging of hepatocellular carcinoma: a prospective pilot study. J Nucl Med. 2018. https://doi.org/10.2967/jnumed.118.214833.
Loktev A, Lindner T, Mier W, Debus J, Altmann A, Jager D, et al. A tumor-imaging method targeting cancer-associated fibroblasts. J Nucl Med. 2018;59:1423–9. https://doi.org/10.2967/jnumed.118.210435.
Lindner T, Loktev A, Altmann A, Giesel F, Kratochwil C, Debus J, et al. Development of quinoline-based theranostic ligands for the targeting of fibroblast activation protein. J Nucl Med. 2018;59:1415–22. https://doi.org/10.2967/jnumed.118.210443.
Klimstra DS, Kloppell G, La Rosa S, Rindi G. Hepatocellular carcinoma. 5th ed. International Agency for Research on Cancer, Lyon: WHO Classification of Tumours Editorial Board (Ed); 2019.
El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365:1118–27. https://doi.org/10.1056/NEJMra1001683.
Sirica AE. The role of cancer-associated myofibroblasts in intrahepatic cholangiocarcinoma. Nat Rev Gastroenterol Hepatol. 2011;9:44–54. https://doi.org/10.1038/nrgastro.2011.222.
Huo L, Dang Y, Lv J, Xing H, Li F. Application of dual phase imaging of 11C-acetate positron emission tomography on differential diagnosis of small hepatic lesions. PLoS One. 2014;9:e96517. https://doi.org/10.1371/journal.pone.0096517.
Lopci E, Torzilli G, Poretti D, de Neto LJ, Donadon M, Rimassa L, et al. Diagnostic accuracy of (1)(1)C-choline PET/CT in comparison with CT and/or MRI in patients with hepatocellular carcinoma. Eur J Nucl Med Mol Imaging. 2015;42:1399–407. https://doi.org/10.1007/s00259-015-3079-5.
Kuyumcu S, Has-Simsek D, Iliaz R, Sanli Y, Buyukkaya F, Akyuz F, et al. Evidence of prostate-specific membrane antigen expression in hepatocellular carcinoma using 68Ga-PSMA PET/CT. Clin Nucl Med. 2019;44:702–6. https://doi.org/10.1097/rlu.0000000000002701.
Pellicoro A, Ramachandran P, Iredale JP, Fallowfield JA. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol. 2014;14:181–94. https://doi.org/10.1038/nri3623.
Wang XM, Yu DM, McCaughan GW, Gorrell MD. Fibroblast activation protein increases apoptosis, cell adhesion, and migration by the LX-2 human stellate cell line. Hepatology. 2005;42:935–45. https://doi.org/10.1002/hep.20853.
Levy MT, McCaughan GW, Marinos G, Gorrell MD. Intrahepatic expression of the hepatic stellate cell marker fibroblast activation protein correlates with the degree of fibrosis in hepatitis C virus infection. Liver. 2002;22:93–101. https://doi.org/10.1034/j.1600-0676.2002.01503.x.
Cox G, Kable E, Jones A, Fraser I, Manconi F, Gorrell MD. 3-dimensional imaging of collagen using second harmonic generation. J Struct Biol. 2003;141:53–62.
Kim YJ, Rhee H, Yoo JE, Alves VAF, Kim GJ, Kim HM, et al. Tumour epithelial and stromal characteristics of hepatocellular carcinomas with abundant fibrous stroma: fibrolamellar versus scirrhous hepatocellular carcinoma. Histopathology. 2017;71:217–26. https://doi.org/10.1111/his.13219.
Gao MQ, Kim BG, Kang S, Choi YP, Park H, Kang KS, et al. Stromal fibroblasts from the interface zone of human breast carcinomas induce an epithelial-mesenchymal transition-like state in breast cancer cells in vitro. J Cell Sci. 2010;123:3507–14. https://doi.org/10.1242/jcs.072900.
Kim GJ, Rhee H, Yoo JE, Ko JE, Lee JS, Kim H, et al. Increased expression of CCN2, epithelial membrane antigen, and fibroblast activation protein in hepatocellular carcinoma with fibrous stroma showing aggressive behavior. PLoS One. 2014;9:e105094. https://doi.org/10.1371/journal.pone.0105094.
Yin Z, Dong C, Jiang K, Xu Z, Li R, Guo K, et al. Heterogeneity of cancer-associated fibroblasts and roles in the progression, prognosis, and therapy of hepatocellular carcinoma. J Hematol Oncol. 2019;12:101. https://doi.org/10.1186/s13045-019-0782-x.
Funding
This work was sponsored in part by the National Natural Science Foundation of China (Grant Nos. 81571713, No. 81671722, No. 81801741), Fundamental Research Funds for the Central Universities (3332020011) and the CAMS Innovation Fund for Medical Sciences (Grant Nos. 2016-I2M-4-003, 2017-I2M-3-001, 2018-I2M-3-001).
Author information
Authors and Affiliations
Contributions
Ximin Shi, Li Huo and Xiang Li designed the study, interpreted the data and led the writing and review of the manuscript. Ximin Shi, Haiqun Xing and Shaobo Yao synthesised the 68Ga-FAPI-4 and performed the examination. Xiaobo Yang and Haitao Zhao collected clinical data. Hui Zhang evaluated the FAP IHC images. Fang Li and Marcus Hacker participated in the review of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
This prospective study was approved by the Peking Union Medical College Hospital ethics committee (IRB protocol #ZS1810).
Consent to participate
All subjects provided written informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - General
Rights and permissions
About this article
Cite this article
Shi, X., Xing, H., Yang, X. et al. Fibroblast imaging of hepatic carcinoma with 68Ga-FAPI-04 PET/CT: a pilot study in patients with suspected hepatic nodules. Eur J Nucl Med Mol Imaging 48, 196–203 (2021). https://doi.org/10.1007/s00259-020-04882-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-020-04882-z