Abstract
Background
Technetium-99m macroaggregated albumin is used to estimate lung shunt fraction (LSF) prior to yttrium-90 (Y90). Studies have debated the safety and efficacy of Y90 in patients with LSF > 15%. We aimed to assess the role of Y90 in hepatocellular carcinoma (HCC) with LSF > 15%.
Methods
With IRB approval, we searched our prospectively acquired database of HCC patients with Y90 treated with LSF > 15%. Median LSF and liver and lung doses were calculated. The response was assessed using RECIST. Overall survival (OS) was calculated from date of first Y90.
Results
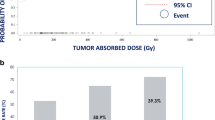
A total of 103 HCC patients underwent Y90. The median baseline LSF was 24.4% (IQR 18.1–28.8). Patients exhibited multifocal disease (59/103, 60%) and median tumor size of 7.85 cm (IQR 5.2, 10.57). BCLC class was A, B, C, and D in 7 (7%), 5 (5%), 85 (83%), and 6 (6%) patients, respectively. The median liver dose was 84.6 Gy (IQR 57.4, 107.55). The median lung dose per session and cumulatively was 22.9 Gy (IQR 15–28) and 29.5 Gy (IQR 20.5–44.3). Thirty-three patients (32%) demonstrated partial response, 57 stable disease, and 13 (13%) had progressive disease. The median OS was 7.3 months (95% CI 5.3, 11.47). Twenty patients (19%) had non-specific pulmonary symptoms (cough, shortness of breath, wheezing) in the 1-year post-Y90. The median time to the appearance of non-specific pulmonary symptoms was 63 days (range 7–224). Thoracic imaging demonstrated no pulmonary fibrosis/injury following treatment in any patient.
Conclusion
Y90 can be performed in patients with LSF > 15%. The RECIST response was identified in 32% of the patients. In isolation, LSF > 15% should not deter from treatment with Y90.


Similar content being viewed by others
References
Parkin DM. Global cancer statistics in the year 2000. Lancet Oncol. 2001;2:533–43. https://doi.org/10.1016/S1470-2045(01)00486-7.
El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557–76. https://doi.org/10.1053/j.gastro.2007.04.061.
Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol. 2013;47(Suppl):S2–6. https://doi.org/10.1097/MCG.0b013e3182872f29.
Cha CH, Ruo L, Fong Y, Jarnagin WR, Shia J, Blumgart LH, et al. Resection of hepatocellular carcinoma in patients otherwise eligible for transplantation. Ann Surg. 2003;238:315–21; discussion 21-3. https://doi.org/10.1097/01.sla.0000086548.84705.ef.
Salem R, Lewandowski RJ, Mulcahy MF, Riaz A, Ryu RK, Ibrahim S, et al. Radioembolization for hepatocellular carcinoma using Yttrium-90 microspheres: a comprehensive report of long-term outcomes. Gastroenterology. 2010;138:52–64. https://doi.org/10.1053/j.gastro.2009.09.006.
Lin M. Radiation pneumonitis caused by yttrium-90 microspheres: radiologic findings. AJR Am J Roentgenol. 1994;162:1300–2.
Sangro B, Martinez-Urbistondo D, Bester L, Bilbao JI, Coldwell DM, Flamen P, et al. Prevention and treatment of complications of selective internal radiation therapy: expert guidance and systematic review. Hepatology. 2017;66:969–82. https://doi.org/10.1002/hep.29207.
Wright CL, Werner JD, Tran JM, Gates VL, Rikabi AA, Shah MH, et al. Radiation pneumonitis following yttrium-90 radioembolization: case report and literature review. J Vasc Interv Radiol. 2012;23:669–74. https://doi.org/10.1016/j.jvir.2012.01.059.
Leung TW, Lau WY, Ho SK, Ward SC, Chow JH, Chan MS, et al. Radiation pneumonitis after selective internal radiation treatment with intraarterial 90yttrium-microspheres for inoperable hepatic tumors. Int J Radiat Oncol Biol Phys. 1995;33:919–24.
Ho S, Lau WY, Leung TW, Chan M, Johnson PJ, Li AK. Clinical evaluation of the partition model for estimating radiation doses from yttrium-90 microspheres in the treatment of hepatic cancer. Eur J Nucl Med. 1997;24:293–8.
Salem R, Parikh P, Atassi B, Lewandowski RJ, Ryu RK, Sato KT, et al. Incidence of radiation pneumonitis after hepatic intra-arterial radiotherapy with yttrium-90 microspheres assuming uniform lung distribution. Am J Clin Oncol. 2008;31:431–8. https://doi.org/10.1097/COC.0b013e318168ef65.
Salem R, Thurston KG. Radioembolization with 90Yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 1: Technical and methodologic considerations. J Vasc Interv Radiol. 2006;17:1251–78. https://doi.org/10.1097/01.RVI.0000233785.75257.9A.
Salem R, Thurston KG. Radioembolization with 90yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 2: special topics. J Vasc Interv Radiol. 2006;17:1425–39. https://doi.org/10.1097/01.RVI.0000235779.88652.53.
Kulik L, Vouche M, Koppe S, Lewandowski RJ, Mulcahy MF, Ganger D, et al. Prospective randomized pilot study of Y90+/−sorafenib as bridge to transplantation in hepatocellular carcinoma. J Hepatol. 2014;61:309–17. https://doi.org/10.1016/j.jhep.2014.03.023.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Riaz A, Memon K, Miller FH, Nikolaidis P, Kulik LM, Lewandowski RJ, et al. Role of the EASL, RECIST, and WHO response guidelines alone or in combination for hepatocellular carcinoma: radiologic-pathologic correlation. J Hepatol. 2011;54:695–704. https://doi.org/10.1016/j.jhep.2010.10.004.
Xing M, Lahti S, Kokabi N, Schuster DM, Camacho JC, Kim HS. 90Y radioembolization lung shunt fraction in primary and metastatic liver cancer as a biomarker for survival. Clin Nucl Med. 2016;41:21–7. https://doi.org/10.1097/RLU.0000000000000915.
Ludwig JM, Ambinder EM, Ghodadra A, Xing M, Prajapati HJ, Kim HS. Lung shunt fraction prior to yttrium-90 radioembolization predicts survival in patients with neuroendocrine liver metastases: single-center prospective analysis. Cardiovasc Intervent Radiol. 2016;39:1007–14. https://doi.org/10.1007/s00270-016-1323-4.
Zhu AX, Kang YK, Yen CJ, Finn RS, Galle PR, Llovet JM, et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:282–96. https://doi.org/10.1016/s1470-2045(18)30937-9.
Author information
Authors and Affiliations
Contributions
Study concept and design: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Ali Al Asadi, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Acquisition of data: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Ali Al Asadi, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Analysis and interpretation of data: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Drafting of the manuscript: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Ali Al Asadi, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Critical revision of the manuscript for important intellectual content: all authors
Statistical analysis: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Administrative, technical, or material support: all authors
Study supervision: Arighno Das, Rehan Ali, Ahmed Gabr, Ronald Mora, Samdeep Mouli, Robert Lewandowski, Riad Salem, Ahsun Riaz
Corresponding author
Ethics declarations
Conflict of interest
AR, RL, and RS are advisors to BTG. None of the other authors have a conflict of interest.
Ethical approval
This retrospective study was approved by our institutional review board and the requirement to obtain informed consent was waived.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – Digestive tract
Rights and permissions
About this article
Cite this article
Das, A., Riaz, A., Gabr, A. et al. Safety and efficacy of radioembolization with glass microspheres in hepatocellular carcinoma patients with elevated lung shunt fraction: analysis of a 103-patient cohort. Eur J Nucl Med Mol Imaging 47, 807–815 (2020). https://doi.org/10.1007/s00259-019-04517-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04517-y