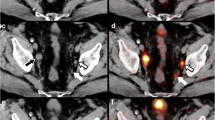
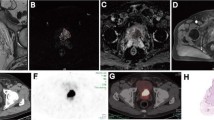
Abstract
Introduction
Radical prostatectomy with extended pelvic lymph node dissection (ePLND) is a curative treatment option for patients with clinically significant localised prostate cancer. The decision to perform an ePLND can be challenging because the overall incidence of lymph node metastasis is relatively low and ePLND is not free of complications. Using current clinical nomograms to identify patients with nodal involvement, approximately 75–85% of ePLNDs performed are negative. The aim of this study was to assess the added value of 68Ga-PSMA-11 PET in predicting lymph node metastasis in men with intermediate- or high-risk prostate cancer.
Methods
68Ga-PSMA-11 PET scans of 60 patients undergoing radical prostatectomy with ePLND were reviewed for qualitative (visual) assessment of suspicious nodes and assessment of quantitative parameters of the primary tumour in the prostate (SUVmax, total activity (PSMAtotal) and PSMA positive volume (PSMAvol)). Ability of quantitative PET parameters to predict nodal metastasis was assessed with receiver operating characteristics (ROC) analysis. A multivariable logistic regression model combining PSA, Gleason score, visual nodal status on PET and primary tumour PSMAtotal was built. Net benefit at each risk threshold was compared with five nomograms: MSKCC nomogram, Yale formula, Roach formula, Winter nomogram and Partin tables (2016).
Results
Overall, pathology of ePLND specimens revealed 31 pelvic metastatic lymph nodes in 12 patients. 68Ga-PSMA-11 PET visual analysis correctly detected suspicious nodes in 7 patients, yielding a sensitivity of 58% and a specificity of 98%. The area under the ROC curve for primary tumour SUVmax was 0.70, for PSMAtotal 0.76 and for PSMAvol 0.75. The optimal cut-off for nodal involvement was PSMAtotal > 49.1. The PET model including PSA, Gleason score and quantitative PET parameters had a persistently higher net benefit compared with all clinical nomograms.
Conclusion
Our model combining PSA, Gleason score and visual lymph node analysis on 68Ga-PSMA-11 PET with PSMAtotal of the primary tumour showed a tendency to improve patient selection for ePLND over the currently used clinical nomograms. Although this result has to be validated, 68Ga-PSMA-11 PET showed the potential to reduce unnecessary surgical procedures in patients with intermediate- or high-risk prostate cancer.





Similar content being viewed by others
Abbreviations
- RPE:
-
Radical prostatectomy
- ePLND:
-
Extended pelvic lymph node dissection
- RT:
-
Radiotherapy
- LNM:
-
Lymph node metastasis
- CT:
-
Computed tomography
- PET:
-
Positron emission tomography
- 68Ga-PSMA-11 PET:
-
Positron emission tomography with prostate-specific membrane antigen
- 68Ga-PSMA-11 PET/MRI:
-
68Ga-PSMA-11 PET/magnetic resonance imaging
- PSA:
-
Prostate-specific antigen
- GS:
-
Gleason score
- MSKCC:
-
MSKCC nomogram
- YF:
-
Yale formula
- RF:
-
Roach formula
- WN:
-
Winter nomogram
- PT (2016):
-
Partin tables
- SD:
-
Standard deviation
- SUVmax :
-
Standard uptake value
- PSMAvol :
-
PSMA positive volume
- PSMAtotal :
-
PSMA accumulation
- ROC:
-
Receiver operating characteristics
- AUC:
-
Area under the ROC curve
- NB:
-
Net benefit
- EIP:
-
Irradiation of the pelvic nodes
- CI:
-
95% Confidence interval
References
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71:618–29. https://doi.org/10.1016/j.eururo.2016.08.003.
Ploussard G, Briganti A, de la Taille A, Haese A, Heidenreich A, Menon M, et al. Pelvic lymph node dissection during robot-assisted radical prostatectomy: efficacy, limitations, and complications-a systematic review of the literature. Eur Urol. 2014;65:7–16. https://doi.org/10.1016/j.eururo.2013.03.057.
Fossati N, Willemse PM, Van den Broeck T, van den Bergh RCN, Yuan CY, Briers E, et al. The benefits and harms of different extents of lymph node dissection during radical prostatectomy for prostate cancer: a systematic review. Eur Urol. 2017;72:84–109. https://doi.org/10.1016/j.eururo.2016.12.003.
Bernstein AN, Shoag JE, Golan R, Halpern JA, Schaeffer EM, Hsu WC, et al. Contemporary incidence and outcomes of prostate cancer lymph node metastases. J Urol. 2018;199:1510–7. https://doi.org/10.1016/j.juro.2017.12.048.
Briganti A, Abdollah F, Nini A, Suardi N, Gallina A, Capitanio U, et al. Performance characteristics of computed tomography in detecting lymph node metastases in contemporary patients with prostate cancer treated with extended pelvic lymph node dissection. Eur Urol. 2012;61:1132–8. https://doi.org/10.1016/j.eururo.2011.11.008.
Hovels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL, et al. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol. 2008;63:387–95. https://doi.org/10.1016/j.crad.2007.05.022.
Evangelista L, Guttilla A, Zattoni F, Muzzio PC, Zattoni F. Utility of choline positron emission tomography/computed tomography for lymph node involvement identification in intermediate- to high-risk prostate cancer: a systematic literature review and meta-analysis. Eur Urol. 2013;63:1040–8. https://doi.org/10.1016/j.eururo.2012.09.039.
von Eyben FE, Kairemo K. Meta-analysis of (11)C-choline and (18)F-choline PET/CT for management of patients with prostate cancer. Nucl Med Commun. 2014;35:221–30. https://doi.org/10.1097/MNM.0000000000000040.
Nguyen DP, Huber PM, Metzger TA, Genitsch V, Schudel HH, Thalmann GN. A specific mapping study using fluorescence sentinel lymph node detection in patients with intermediate- and high-risk prostate cancer undergoing extended pelvic lymph node dissection. Eur Urol. 2016;70:734–7. https://doi.org/10.1016/j.eururo.2016.01.034.
Chun FK, Karakiewicz PI, Briganti A, Gallina A, Kattan MW, Montorsi F, et al. Prostate cancer nomograms: an update. Eur Urol. 2006;50:914–26; discussion 26. https://doi.org/10.1016/j.eururo.2006.07.042.
Bianchi L, Gandaglia G, Fossati N, Suardi N, Moschini M, Cucchiara V, et al. Pelvic lymph node dissection in prostate cancer: indications, extent and tailored approaches. Urologia. 2017;84:9–19. https://doi.org/10.5301/uro.5000139.
Mohler JL, Armstrong AJ, Bahnson RR, D'Amico AV, Davis BJ, Eastham JA, et al. Prostate Cancer, version 1.2016. J Natl Compr Cancer Netw. 2016;14:19–30.
Briganti A, Larcher A, Abdollah F, Capitanio U, Gallina A, Suardi N, et al. Updated nomogram predicting lymph node invasion in patients with prostate cancer undergoing extended pelvic lymph node dissection: the essential importance of percentage of positive cores. Eur Urol. 2012;61:480–7. https://doi.org/10.1016/j.eururo.2011.10.044.
Banapour P, Schumacher A, Lin JC, Finley DS. Radical prostatectomy and pelvic lymph node dissection in Kaiser Permanente Southern California: 15-year experience. Perm J. 2019;23. https://doi.org/10.7812/TPP/17-233.
Roscigno M, Nicolai M, La Croce G, Pellucchi F, Scarcello M, Sacca A, et al. Difference in frequency and distribution of nodal metastases between intermediate and high risk prostate cancer patients: results of a superextended pelvic lymph node dissection. Front Surg. 2018;5:52. https://doi.org/10.3389/fsurg.2018.00052.
Hope TA, Goodman JZ, Allen IE, Calais J, Fendler WP, Carroll PR. Meta-analysis of (68)Ga-PSMA-11 PET accuracy for the detection of prostate Cancer validated by histopathology. J Nucl Med. 2018. https://doi.org/10.2967/jnumed.118.219501.
Thalgott M, Duwel C, Rauscher I, Heck MM, Haller B, Gafita A, et al. One-stop shop whole-body (68)Ga-PSMA-11 PET/MRI compared to clinical Nomograms for preoperative T- and N-Staging of High-Risk Prostate Cancer. J Nucl Med. 2018. https://doi.org/10.2967/jnumed.117.207696.
Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D, et al. (68)Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44:941–9. https://doi.org/10.1007/s00259-017-3631-6.
von Klot CJ, Merseburger AS, Boker A, Schmuck S, Ross TL, Bengel FM, et al. (68)Ga-PSMA PET/CT imaging predicting Intraprostatic tumor extent, extracapsular extension and seminal vesicle invasion prior to radical prostatectomy in patients with prostate cancer. Nucl Med Mol Imaging. 2017;51:314–22. https://doi.org/10.1007/s13139-017-0476-7.
Ross JS, Sheehan CE, Fisher HA, Kaufman RP Jr, Kaur P, Gray K, et al. Correlation of primary tumor prostate-specific membrane antigen expression with disease recurrence in prostate cancer. Clin Cancer Res. 2003;9:6357–62.
Bravaccini S, Puccetti M, Bocchini M, Ravaioli S, Celli M, Scarpi E, et al. PSMA expression: a potential ally for the pathologist in prostate cancer diagnosis. Sci Rep. 2018;8:4254. https://doi.org/10.1038/s41598-018-22594-1.
Uslu-Besli L, Asa S, Bakir B, Sayman H, Sager S, Khosroshahi BR, et al. Correlation of SUVmax and ADC values detected by Ga-68 PSMA PET/MRI in primary prostate lesions and their significance in lymph node metastasis. Eur J Nucl Med Mol Imaging. 2018;45:S20-S.
Hueting TA, Cornel EB, Somford DM, Jansen H, van Basten J-PA, Pleijhuis RG, et al. External validation of models predicting the probability of lymph node involvement in prostate cancer patients. Eur Urol Oncol. 2018.
Cagiannos I, Karakiewicz P, Eastham JA, Ohori M, Rabbani F, Gerigk C, et al. A preoperative nomogram identifying decreased risk of positive pelvic lymph nodes in patients with prostate cancer. J Urol. 2003;170:1798–803. https://doi.org/10.1097/01.ju.0000091805.98960.13.
Yu JB, Makarov DV, Gross C. A new formula for prostate cancer lymph node risk. Int J Radiat Oncol Biol Phys. 2011;80:69–75. https://doi.org/10.1016/j.ijrobp.2010.01.068.
Roach M 3rd, Marquez C, Yuo HS, Narayan P, Coleman L, Nseyo UO, et al. Predicting the risk of lymph node involvement using the pre-treatment prostate specific antigen and Gleason score in men with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 1994;28:33–7.
Winter A, Kneib T, Rohde M, Henke RP, Wawroschek F. First nomogram predicting the probability of lymph node involvement in prostate cancer patients undergoing radioisotope guided sentinel lymph node dissection. Urol Int. 2015;95:422–8. https://doi.org/10.1159/000431182.
Tosoian JJ, Chappidi M, Feng Z, Humphreys EB, Han M, Pavlovich CP, et al. Prediction of pathological stage based on clinical stage, serum prostate-specific antigen, and biopsy Gleason score: Partin Tables in the contemporary era. BJU Int. 2017;119:676–83. https://doi.org/10.1111/bju.13573.
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–74.
Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, et al. Ga-68-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44:1014–24. https://doi.org/10.1007/s00259-017-3670-z.
Kranzbuhler B, Nagel H, Becker AS, Muller J, Huellner M, Stolzmann P, et al. Clinical performance of (68)Ga-PSMA-11 PET/MRI for the detection of recurrent prostate cancer following radical prostatectomy. Eur J Nucl Med Mol Imaging. 2018;45:20–30. https://doi.org/10.1007/s00259-017-3850-x.
Hofman MS, Hicks RJ, Maurer T, Eiber M. Prostate-specific membrane antigen PET: clinical utility in prostate cancer, normal patterns, pearls, and pitfalls. Radiographics. 2018;38:200–17. https://doi.org/10.1148/rg.2018170108.
Fendler WP, Calais J, Allen-Auerbach M, Bluemel C, Eberhardt N, Emmett L, et al. (68)Ga-PSMA-11 PET/CT interobserver agreement for prostate cancer assessments: an international multicenter prospective study. J Nucl Med. 2017;58:1617–23. https://doi.org/10.2967/jnumed.117.190827.
Feicke A, Baumgartner M, Talimi S, Schmid DM, Seifert HH, Muntener M, et al. Robotic-assisted laparoscopic extended pelvic lymph node dissection for prostate cancer: surgical technique and experience with the first 99 cases. Eur Urol. 2009;55:876–83. https://doi.org/10.1016/j.eururo.2008.12.006.
Steyerberg EW, Vickers AJ, Cook NR, Gerds T, Gonen M, Obuchowski N, et al. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology. 2010;21:128–38. https://doi.org/10.1097/EDE.0b013e3181c30fb2.
Vickers AJ, Elkin EB. Decision curve analysis: a novel method for evaluating prediction models. Med Decis Mak. 2006;26:565–74. https://doi.org/10.1177/0272989X06295361.
Kerr KF, Brown MD, Zhu K, Janes H. Assessing the clinical impact of risk prediction models with decision curves: guidance for correct interpretation and appropriate use. J Clin Oncol. 2016;34:2534–40. https://doi.org/10.1200/JCO.2015.65.5654.
Van Calster B, Wynants L, Verbeek JFM, Verbakel JY, Christodoulou E, Vickers AJ, et al. Reporting and interpreting decision curve analysis: a guide for investigators. Eur Urol. 2018;74:796–804. https://doi.org/10.1016/j.eururo.2018.08.038.
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA, et al. The 2014 International Society of Urological Pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol. 2016;40:244–52. https://doi.org/10.1097/PAS.0000000000000530.
Berglund E, Maaskola J, Schultz N, Friedrich S, Marklund M, Bergenstrahle J, et al. Spatial maps of prostate cancer transcriptomes reveal an unexplored landscape of heterogeneity. Nat Commun. 2018;9:2419. https://doi.org/10.1038/s41467-018-04724-5.
Tu SM, Lin SH, Logothetis CJ. Stem-cell origin of metastasis and heterogeneity in solid tumours. Lancet Oncol. 2002;3:508–13.
Hupe MC, Philippi C, Roth D, Kumpers C, Ribbat-Idel J, Becker F, et al. Expression of Prostate-Specific Membrane Antigen (PSMA) on biopsies is an independent risk stratifier of prostate cancer patients at time of initial diagnosis. Front Oncol. 2018;8:623. https://doi.org/10.3389/fonc.2018.00623.
Blanchard P, Faivre L, Lesaunier F, Salem N, Mesgouez-Nebout N, Deniau-Alexandre E, et al. Outcome according to elective pelvic radiation therapy in patients with high-risk localized prostate cancer: a secondary analysis of the GETUG 12 phase 3 randomized trial. Int J Radiat Oncol Biol Phys. 2016;94:85–92. https://doi.org/10.1016/j.ijrobp.2015.09.020.
Seaward SA, Weinberg V, Lewis P, Leigh B, Phillips TL, Roach M 3rd. Improved freedom from PSA failure with whole pelvic irradiation for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 1998;42:1055–62.
Aizer AA, Yu JB, McKeon AM, Decker RH, Colberg JW, Peschel RE. Whole pelvic radiotherapy versus prostate only radiotherapy in the management of locally advanced or aggressive prostate adenocarcinoma. Int J Radiat Oncol Biol Phys. 2009;75:1344–9. https://doi.org/10.1016/j.ijrobp.2008.12.082.
Roach M, Moughan J, Lawton CAF, Dicker AP, Zeitzer KL, Gore EM, et al. Sequence of hormonal therapy and radiotherapy field size in unfavourable, localised prostate cancer (NRG/RTOG 9413): long-term results of a randomised, phase 3 trial. Lancet Oncol. 2018;19:1504–15. https://doi.org/10.1016/S1470-2045(18)30528-X.
Daoud MA, Aboelnaga EM, Alashry MS, Fathy S, Aletreby MA. Clinical outcome and toxicity evaluation of simultaneous integrated boost pelvic IMRT/VMAT at different dose levels combined with androgen deprivation therapy in prostate cancer patients. Onco Targets Ther. 2017;10:4981–8. https://doi.org/10.2147/OTT.S141224.
Ishii K, Ogino R, Hosokawa Y, Fujioka C, Okada W, Nakahara R, et al. Comparison of dosimetric parameters and acute toxicity after whole-pelvic vs prostate-only volumetric-modulated arc therapy with daily image guidance for prostate cancer. Br J Radiol. 2016;89:20150930. https://doi.org/10.1259/bjr.20150930.
White KL, Varrassi E, Routledge JA, Barraclough LH, Livsey JE, McLaughlin J, et al. Does the use of volumetric modulated arc therapy reduce gastrointestinal symptoms after pelvic radiotherapy? Clin Oncol (R Coll Radiol). 2018;30:e22–e8. https://doi.org/10.1016/j.clon.2017.10.016.
Choo MS, Kim M, Ku JH, Kwak C, Kim HH, Jeong CW. Extended versus standard pelvic lymph node dissection in radical prostatectomy on oncological and functional outcomes: a systematic review and meta-analysis. Ann Surg Oncol. 2017;24:2047–54. https://doi.org/10.1245/s10434-017-5822-6.
Acknowledgements
The authors acknowledge the technicians Marlena Hofbauer and Josephine Trinckauf and their team for the excellent work on high-quality PET images.
Funding
The Department of Nuclear Medicine holds an institutional Research Contract with GE Healthcare. This study was financially supported by the Sick legat and the Iten-Kohaut foundation
Author information
Authors and Affiliations
Contributions
DAF—data collection, data analysis and manuscript writing.
UJM—data analysis, statistics and manuscript writing.
HIGS and TH—patient selection, manuscript writing.
NJR, EEGWV, MM, JHR and MH—manuscript editing and revision.
IAB—study design and manuscript writing.
All authors reviewed and agreed to the manuscript content.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The local ethics committee approved the study protocol and all patients gave a general written informed consent for retrospective use of their data (BASEC Nr. 2018-01284).
Consent for publication
Not applicable.
Availability of data and material
Patient imaging was done in the scope of the routine clinical diagnostic studies, and the raw data are stored in the hospital archiving system at the Zurich University Hospital, Zurich, Switzerland.
Competing interests
IAB has received research grants and speaker honorarium from GE Healthcare, research grants from Swiss Life and speaker honorarium from Bayer Health Care and Astellas Pharma AG. TH holds an advisory function for MSD and Bayer. MH received an Investigator-Initiated Study grant from GE Healthcare. MM received speaker fees from GE Healthcare. Authors DAF, UJM, HIGS, NJR, JHR and EEGWV declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology—Genitourinary
Daniela A Ferraro and Urs J. Muehlematter shared first authorship
Electronic supplementary material
ESM 1
(PNG 182 kb)
Rights and permissions
About this article
Cite this article
Ferraro, D.A., Muehlematter, U.J., Garcia Schüler, H.I. et al. 68Ga-PSMA-11 PET has the potential to improve patient selection for extended pelvic lymph node dissection in intermediate to high-risk prostate cancer. Eur J Nucl Med Mol Imaging 47, 147–159 (2020). https://doi.org/10.1007/s00259-019-04511-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04511-4