Abstract
Purpose
Anti-PD-1/PD-L1 blockade can restore tumour-specific T-cell immunity and is an emerging therapy in non-small-cell lung cancer (NSCLC). We investigated the correlation between 18F-FDG PET/CT-based markers and tumour tissue expression of PD-L1, necrosis and clinical outcome in patients receiving checkpoint inhibitor treatment.
Methods
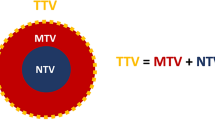
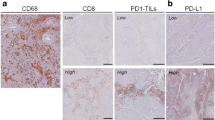
PD-Li expression in biopsy or resection specimens from 49 patients with confirmed NSCLC was investigated by immunohistochemistry. Maximum standardized uptake value (SUVmax), mean SUV (SUVmean), metabolic tumour volume (MTV) and total lesion glycolysis (TLG) were obtained from 18F-FDG PET/CT images. The ratio of metabolic to morphological lesion volumes (MMVR) and its association with PD-L1 expression in each lesion were calculated. The associations between histologically reported necrosis and 18F-FDG PET imaging patterns and radiological outcome (evaluated by iRECIST) following anti-PD-1/PD-L1 therapy were also analysed. In 14 patients, the association between necrosis and MMVR and tumour immune contexture were analysed by multiple immunofluorescent (IF) staining for CD8, PD-1, granzyme B (GrzB) and NFATC2.
Results
In total, 25 adenocarcinomas and 24 squamous cell carcinomas were analysed. All tumours showed metabolic 18F-FDG PET uptake. MMVR was correlated inversely with PD-L1 expression in tumour cells. Furthermore, PD-L1 expression and low MMVR were significantly correlated with clinical benefit. Necrosis was correlated negatively with MMVR. Multiplex IF staining showed a greater frequency of activated CD8+ cells in necrotic tumours than in nonnecrotic tumours in both stromal and epithelial tumour compartments.
Conclusion
This study introduces MMVR as a new imaging biomarker and its ability to noninvasively capture increased PD-L1 tumour expression and predict clinical benefit from checkpoint blockade in NSCLC should be further evaluated.






Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7–30. https://doi.org/10.3322/caac.21332.
Didkowska J, Wojciechowska U, Manczuk M, Lobaszewski J. Lung cancer epidemiology: contemporary and future challenges worldwide. Ann Transl Med. 2016;4:150. https://doi.org/10.21037/atm.2016.03.11.
Scagliotti GV, Bironzo P, Vansteenkiste JF. Addressing the unmet need in lung cancer: the potential of immuno-oncology. Cancer Treat Rev. 2015;41:465–75. https://doi.org/10.1016/j.ctrv.2015.04.001.
Brahmer JR, Pardoll DM. Immune checkpoint inhibitors: making immunotherapy a reality for the treatment of lung cancer. Cancer Immunol Res. 2013;1:85–91. https://doi.org/10.1158/2326-6066.cir-13-0078.
Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–54. https://doi.org/10.1056/NEJMoa1200690.
Brahmer J, Reckamp KL, Baas P, Crino L, Eberhardt WE, Poddubskaya E, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015;373:123–35. https://doi.org/10.1056/NEJMoa1504627.
Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373:1627–39. https://doi.org/10.1056/NEJMoa1507643.
Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med. 2015;372:2018–28. https://doi.org/10.1056/NEJMoa1501824.
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ, Topalian SL, Hwu P, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366:2455–65. https://doi.org/10.1056/NEJMoa1200694.
Soria JC, Marabelle A, Brahmer JR, Gettinger S. Immune checkpoint modulation for non-small cell lung cancer. Clin Cancer Res. 2015;21:2256–62. https://doi.org/10.1158/1078-0432.ccr-14-2959.
Barsoum IB, Smallwood CA, Siemens DR, Graham CH. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res. 2014;74:665–74. https://doi.org/10.1158/0008-5472.can-13-0992.
Anagnostou VK, Brahmer JR. Cancer immunotherapy: a future paradigm shift in the treatment of non-small cell lung cancer. Clin Cancer Res. 2015;21:976–84. https://doi.org/10.1158/1078-0432.ccr-14-1187.
Remon J, Chaput N, Planchard D. Predictive biomarkers for programmed death-1/programmed death ligand immune checkpoint inhibitors in nonsmall cell lung cancer. Curr Opin Oncol. 2016;28:122–9. https://doi.org/10.1097/cco.0000000000000263.
Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–50. https://doi.org/10.1016/S0140-6736(15)01281-7.
Pai-Scherf L, Blumenthal GM, Li H, Subramaniam S, Mishra-Kalyani PS, He K, et al. FDA approval summary: Pembrolizumab for treatment of metastatic non-small cell lung cancer: first-line therapy and beyond. Oncologist. 2017;22(11):1392–9. https://doi.org/10.1634/theoncologist.2017-0078.
Hellmann MD, Ciuleanu TE, Pluzanski A, Lee JS, Otterson GA, Audigier-Valette C, et al. Nivolumab plus Ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med. 2018;378(22):2093–104. https://doi.org/10.1056/NEJMoa1801946.
Davison J, Mercier G, Russo G, Subramaniam RM. PET-based primary tumor volumetric parameters and survival of patients with non-small cell lung carcinoma. AJR Am J Roentgenol. 2013;200:635–40. https://doi.org/10.2214/ajr.12.9138.
Zhu A, Lee D, Shim H. Metabolic positron emission tomography imaging in cancer detection and therapy response. Semin Oncol. 2011;38:55–69. https://doi.org/10.1053/j.seminoncol.2010.11.012.
Chang CH, Qiu J, O’Sullivan D, Buck MD, Noguchi T, Curtis JD, et al. Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell. 2015;162(6):1229–41. https://doi.org/10.1016/j.cell.2015.08.016.
Steinbach JP, Wolburg H, Klumpp A, Probst H, Weller M. Hypoxia-induced cell death in human malignant glioma cells: energy deprivation promotes decoupling of mitochondrial cytochrome c release from caspase processing and necrotic cell death. Cell Death Differ. 2003;10:823–32. https://doi.org/10.1038/sj.cdd.4401252.
Soussan M, Cyrta J, Pouliquen C, Chouahnia K, Orlhac F, Martinod E, et al. Fluorine 18 fluorodeoxyglucose PET/CT volume-based indices in locally advanced non-small cell lung cancer: prediction of residual viable tumor after induction chemotherapy. Radiology. 2014;272:875–84. https://doi.org/10.1148/radiol.1413219.
Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017;18(3):e143–52. https://doi.org/10.1016/S1470-2045(17)30074-8.
Xie X, Willemink MJ, de Jong PA, van Ooijen PM, Oudkerk M, Vliegenthart R, et al. Small irregular pulmonary nodules in low-dose CT: observer detection sensitivity and volumetry accuracy. AJR Am J Roentgenol. 2014;202:W202–9. https://doi.org/10.2214/ajr.13.10830.
Mami-Chouaib F, Blanc C, Corgnac S, et al. Resident memory T cells, critical components in tumor immunology. J Immunother Cancer. 2018;6:87–97. https://doi.org/10.1186/s40425-018-0399-6.
Speiser DE, Ho PC, Verdeil G. Regulatory circuits of T cell function in cancer. Nat Rev Immunol. 2016;16(10):599–611. https://doi.org/10.1038/nri.2016.80.
Sheikhbahaei S, Mena E, Yanamadala A, Reddy S, Solnes LB, Wachsmann J, et al. The value of FDG PET/CT in treatment response assessment, follow-up, and surveillance of lung cancer. AJR Am J Roentgenol. 2017;208:420–33. https://doi.org/10.2214/ajr.16.16532.
Ilie M, Hofman V, Dietel M, Soria JC, Hofman P. Assessment of the PD-L1 status by immunohistochemistry: challenges and perspectives for therapeutic strategies in lung cancer patients. Virchows Arch. 2016;468:511–25. https://doi.org/10.1007/s00428-016-1910-4.
Passiglia F, Bronte G, Bazan V. PD-L1 expression as predictive biomarker in patients with NSCLC: a pooled analysis. Oncotarget. 2016;7(15):19738–47. https://doi.org/10.18632/oncotarget.7582.
Zhou ZJ, Zhan P, Song Y. PD-L1 over-expression and survival in patients with non-small cell lung cancer: a meta-analysis. Transl Lung Cancer Res. 2015;4:203–8. https://doi.org/10.3978/j.issn.2218-6751.2015.03.02.
Wang A, Wang HY, Liu Y, Zhao MC, Zhang HJ, Lu ZY, et al. The prognostic value of PD-L1 expression for non-small cell lung cancer patients: a meta-analysis. Eur J Surg Oncol. 2015;41:450–6. https://doi.org/10.1016/j.ejso.2015.01.020.
Gkogkou C, Frangia K, Saif MW, Trigidou R, Syrigos K. Necrosis and apoptotic index as prognostic factors in non-small cell lung carcinoma: a review. Springerplus. 2014;3:120. https://doi.org/10.1186/2193-1801-3-120.
Tormanen U, Eerola AK, Rainio P, Vahakangas K, Soini Y, Sormunen R, et al. Enhanced apoptosis predicts shortened survival in non-small cell lung carcinoma. Cancer Res. 1995;55:5595–602.
Cho S, Sung SW, Jheon S, Chung JH. Risk of recurrence in surgically resected stage I adenocarcinoma of the lung: histopathologic and immunohistochemical analysis. Lung. 2008;186:411–9. https://doi.org/10.1007/s00408-008-9116-4.
Kilicgun A, Turna A, Sayar A, Solak O, Urer N, Gurses A. Very important histopathological factors in patients with resected non-small cell lung cancer: necrosis and perineural invasion. Thorac Cardiovasc Surg. 2010;58:93–7. https://doi.org/10.1055/s-0029-1186240.
Park SY, Lee HS, Jang HJ, Lee GK, Chung KY, Zo JI. Tumor necrosis as a prognostic factor for stage IA non-small cell lung cancer. Ann Thorac Surg. 2011;91:1668–73. https://doi.org/10.1016/j.athoracsur.2010.12.028.
Pataer A, Kalhor N, Correa AM, Raso MG, Erasmus JJ, Kim ES, et al. Histopathologic response criteria predict survival of patients with resected lung cancer after neoadjuvant chemotherapy. J Thorac Oncol. 2012;7:825–32. https://doi.org/10.1097/JTO.0b013e318247504a.
Tantraworasin A, Saeteng S, Lertprasertsuke N, Arreyakajohn N, Kasemsarn C, Patumanond J. Prognostic factors of tumor recurrence in completely resected non-small cell lung cancer. Cancer Manag Res. 2013;5:77–84. https://doi.org/10.2147/cmar.s45642.
Swinson DE, Jones JL, Richardson D, Cox G, Edwards JG, O’Byrne KJ. Tumour necrosis is an independent prognostic marker in non-small cell lung cancer: correlation with biological variables. Lung Cancer. 2002;37:235–40.
Langendijk H, Thunnissen E, Arends JW, de Jong J, ten Velde G, Lamers R, et al. Cell proliferation and apoptosis in stage III inoperable non-small cell lung carcinoma treated by radiotherapy. Radiother Oncol. 2000;56:197–207.
Dworakowska D, Jassem E, Jassem J, Karmolinski A, Lapinski M, Tomaszewski D, et al. Prognostic value of the apoptotic index analysed jointly with selected cell cycle regulators and proliferation markers in non-small cell lung cancer. Lung Cancer. 2009;66:127–33. https://doi.org/10.1016/j.lungcan.2009.01.008.
Garg AD, Agostinis P. Cell death and immunity in cancer: from danger signals to mimicry of pathogen defense responses. Immunol Rev. 2017;280(1):126–48. https://doi.org/10.1111/imr.12574.
Chang YL, Yang CY, Lin MW, Wu CT, Yang PC. High co-expression of PD-L1 and HIF-1α correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur J Cancer. 2016;60:125–35. https://doi.org/10.1016/j.ejca.2016.03.012.
Wang V, Yang L, Huang F, Zhang Q, Liu S, Ma L, et al. Inflammatory cytokines IL-17 and TNF-α up-regulate PD-L1 expression in human prostate and colon cancer cells. Immunol Lett. 2017;184:7–14. https://doi.org/10.1016/j.imlet.2017.02.006.
Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39(1):1–10. https://doi.org/10.1016/j.immuni.2013.07.012.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was waived due to the retrospective nature of this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – Chest
Electronic supplementary material
Supplementary Fig. 1
Multispectral Immunofluorescence images at x10 magnification in a patient with tumour-associated necrosis (a) and a patient with no tumour-associated necrosis (b). (PPTX 600 kb)
Supplementary Fig. 2
High-resolution multispectral images at x20 magnification in a patient with tumour-associated necrosis (a) and a patient with no tumour-associated necrosis (b). pink cytokeratin (CK), red CD8, green PD1, cyan granzyme B (GrzB), yellow NFATC2. (PPTX 2746 kb)
Rights and permissions
About this article
Cite this article
Jreige, M., Letovanec, I., Chaba, K. et al. 18F-FDG PET metabolic-to-morphological volume ratio predicts PD-L1 tumour expression and response to PD-1 blockade in non-small-cell lung cancer. Eur J Nucl Med Mol Imaging 46, 1859–1868 (2019). https://doi.org/10.1007/s00259-019-04348-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04348-x