Abstract
Purpose
Current guidelines recommend thyroid hormone withdrawal (THW) of 3–4 weeks before radioiodine remnant ablation (RRA) of differentiated thyroid carcinoma (DTC). We aimed to evaluate (1) the reliability of a shorter THW (i.e., 14 days) to achieve adequate TSH levels (i.e., 30 mU/l), (2) the association between length of THW and response to therapy, and (3) the potential association between pre-ablation TSH levels and patients’ outcome.
Methods
After thyroidectomy, all patients started LT4 therapy, which was subsequently discontinued in order to perform RRA. Patients were broken down into two groups according to the length of THW: group A, 2 weeks of THW, and group B, 3–4 weeks of THW. We used clinical, biochemical, and imaging data to evaluate patients’ outcome. By means of univariate and multivariate analysis, including main DTC prognostic factors, we assessed the impact of THW length and TSH levels on patients’ outcome.
Results
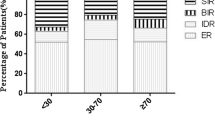
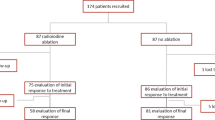
We evaluated 222 patients, 85 of whom were treated with RRA after a THW period of 2 weeks (group A). All other 137 patients underwent RRA after 3–4 weeks THW (group B). At the time of RRA all patients presented TSH levels ≥30 mU/l. After a median follow-up time of 3.4 years, we found 183 patients (82%) with excellent response to treatment and 39 patients (18%) showing incomplete response. Kaplan–Meier response to therapy curves showed that ablation-Tg, tumor size, and lymph node status were significantly associated with prognosis; no associations were found between THW length, TSH levels, and prognosis. Multivariate Cox model showed that only ablation-Tg was significantly associated with treatment response.
Conclusions
Prior to RRA, a short 2-week THW is an effective method to stimulate TSH levels. No difference in terms of incomplete response to treatment was observed between DTC patients prepared for RRA with a short THW and those with the long THW.

Similar content being viewed by others
References
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133.
Zhao T, Liang J, Guo Z, Li T, Lin Y. In patients with low- to intermediate-risk thyroid cancer, a preablative thyrotropin level of 30 μIU/ml is not adequate to achieve better response to 131I therapy. Clin Nucl Med. 2016;41:454–8.
Edmonds CJ, Hayes S, Kermode JC, Thompson BD. Measurement of serum TSH and thyroid hormones in the management of treatment of thyroid carcinoma with radioiodine. Br J Radiol. 1977;50:799–807.
Vrachimis A, Riemann B, Mäder U, Reiners C, Verburg FA. Endogenous TSH levels at the time of (131)I ablation do not influence ablation success, recurrence-free survival or differentiated thyroid cancer-related mortality. Eur J Nucl Med Mol Imaging. 2016;43:224–31.
Golger A, Fridman TR, Eski S, Witterick IJ, Freeman JL, Walfish PG. Three-week thyroxine withdrawal thyroglobulin stimulation screening test to detect low-risk residual/recurrent well differentiated thyroid carcinoma. J Endocrinol Investig. 2003;26:1023–31.
Liel Y. Preparation for radioactive iodine administration in differentiated thyroid cancer patients. Clin Endocrinol. 2002;57(4):523–7.
Luster M, Clarke SE, Dietlein M, Lassmann M, Lind P, Oyen WJ, et al. Guidelines for radioiodine therapy of differentiated thyroid cancer. Eur J Nucl Med Mol Imaging. 2008;35:1941–59.
Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JW, Wiersinga W. European thyroid cancer taskforce. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol. 2006;154:787–803.
Nixon IJ, Wang LY, Migliacci JC, Eskander A, Campbell MJ, Aniss A, et al. An international multi-institutional validation of age 55 years as a cutoff for risk stratification in the AJCC/UICC staging system for well-differentiated thyroid cancer. Thyroid. 2016;26:373–80.
Baloch Z, LiVolsi VA, Tondon R. Aggressive variants of follicular cell-derived thyroid carcinoma; the so-called ‘real thyroid carcinomas’. J Clin Pathol. 2013;66:733–43.
Piccardo A, Arecco F, Morbelli S, Bianchi P, Barbera F, Finessi M, et al. Low thyroglobulin concentrations after thyroidectomy increase the prognostic value of undetectable thyroglobulin levels on levo-thyroxine suppressive treatment in low-risk differentiated thyroid cancer. J Endocrinol Investig. 2010;33:83–7.
Piccardo A, Arecco F, Puntoni M, Foppiani L, Cabria M, Corvisieri S, et al. F focus on high-risk DTC patients: high postoperative serum thyroglobulin level is a strong predictor of disease persistence and is associated to progression-free survival and overall survival. Clin Nucl Med. 2013;38:18–24.
Tuttle RM, Tala H, Shah J, Leboeuf R, Ghossein R, Gonen M, et al. Estimating risk of recurrence in differentiated thyroid cancer after total thyroidectomy and radioactive iodine remnant ablation: using response to therapy variables to modify the initial risk estimates predicted by the new American Thyroid Association staging system. Thyroid. 2010;20:1341–9.
Tuttle RM, Leboeuf R. Follow up approaches in thyroid cancer: a risk adapted paradigm. Endocrinol Metab Clin N Am. 2007;37:419–35. ix–x
Piccardo A, Puntoni M, Bottoni G, Treglia G, Foppiani L, Bertoli M, et al. Differentiated thyroid cancer lymph-node relapse. Role of adjuvant radioactive iodine therapy after lymphadenectomy. Eur J Nucl Med Mol Imaging. 2017;44:926–34.
Giovanella L, Piccardo A. A "new/old method" for TSH stimulation: could a third way to prepare DTC patients for (131)I remnant ablation possibly exist? Eur J Nucl Med Mol Imaging. 2016;43:221–3.
American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer, Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214.
Funding
This research did not receive any specific grants from any funding agency in the public, commercial, or non-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Piccardo, A., Puntoni, M., Ferrarazzo, G. et al. Could short thyroid hormone withdrawal be an effective strategy for radioiodine remnant ablation in differentiated thyroid cancer patients?. Eur J Nucl Med Mol Imaging 45, 1218–1223 (2018). https://doi.org/10.1007/s00259-018-3955-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-3955-x