Abstract
Background
Nivolumab, an anti-programmed death-1 (PD-1) antibody, is administered in patients with previously treated non-small cell lung cancer. However, little is known about the established biomarker predicting the efficacy of nivolumab. Here, we conducted a preliminary study to investigate whether 18F–FDG-PET/CT could predict the therapeutic response of nivolumab at the early phase.
Methods
Twenty-four patients were enrolled in this study. 18F–FDG-PET/CT was carried out before and 1 month after nivolumab therapy. SUVmax, metabolic tumour volume (MTV), and total lesion glycolysis (TLG) were calculated. Immunohistochemical analysis of PD-L1 expression and tumour-infiltrating lymphocytes was conducted.
Results
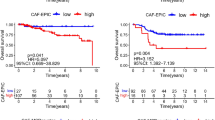
Among all patients, a partial metabolic response to nivolumab was observed in 29% on SUVmax, 25% on MTV, and 33% on TLG, whereas seven (29%) patients achieved a partial response (PR) based on RECIST v1.1. The predictive probability of PR (100% vs. 29%, p = 0.021) and progressive disease (100% vs. 22.2%, p = 0.002) at 1 month after nivolumab initiation was significantly higher in 18F–FDG on PET/CT than in CT scans. Multivariate analysis confirmed that 18F–FDG uptake after administration of nivolumab was an independent prognostic factor. PD-L1 expression and nivolumab plasma concentration could not precisely predict the early therapeutic efficacy of nivolumab.
Conclusion
Metabolic response by 18F–FDG was effective in predicting efficacy and survival at 1 month after nivolumab treatment.





Similar content being viewed by others
References
Mountain CF. Revisions in the international system for staging lung cancer. Chest. 1997;111:1719–7.
Pfister DG, Johnson DH, Azzoli CG, et al. American Society of Clinical Oncology treated of unresectable non-small-cell lung cancer guildeline: update 2003. J Clin Oncol. 2004;22:330–53.
Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus Docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015;373:123–35.
Borghaei H, Paz-Ares L, Hom L, et al. Nivolumab versus Docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373:1627–39.
Reck M, Rodriguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1 positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–33.
Pieterman RM, van Putten JW, Meuzelaar JJ, et al. Preoperative staging of non-small-cell lung cancer with positron-emission tomography. N Engl J Med. 2000;343:254–61.
Lardinois D, Weder W, Hany TF, et al. Staging of non-small-cell lung cancer with integrated positron- emission tomography and computed tomography. N Engl J Med. 2003;348:2500–7.
Moon SH, Cho SH, Park LC, et al. Metabolic response evaluated by 18F-FDG PET/CT as a potential screening tool in identifying a subgroup of patients with advanced non-small cell lung cancer for immediate maintenance therapy after first-line chemotherapy. Eur J Nucl Med Mol Imaging. 2013;40:1005–13.
Wahl RL, Jacene H, Kasamon Y, et al. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50(Suppl 1):122S–5S.
Sunaga N, Oriuchi N, Kaira K, et al. Usefulness of FDG-PET for early prediction of the response to gefitinib in non-small cell lung cancer. Lung Cancer. 2008;59:203–10.
Kaira K, Endo M, Abe M, et al. Biologic correlation of 2-[18F]-fluoro-2-deoxy-D-glucose uptake on positron emission tomography in thymic epithelial tumors. J Clin Oncol. 2010;28:3746–53.
Chang YL, Yang CY, Lin MW, et al. High co-expression of PD-L1 and HIF-1α correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur J Cancer. 2016;60:125–35.
Ruf M, Moch H, Schraml P. PD-L1 expression is regulated by hypoxia inducible factor in clear renal cell carcinoma. Int J Cancer. 2016;139:396–403.
Lopci E, Toschi L, Grizzi F, et al. Cprrelation of metabolic information on FDG-PET with tissue expression of immune markers in patients with non-small cell lung cancer (NSCLC) who are candidates for upfront surgery. Eur J Nucl Med Mol Imaging. 2016;43:1954–61.
Im HJ, Pak K, Cheon GJ, et al. Prognostic value of volumetric parameters of (18)F-FDG PET in non-small-cell lung cancer: a meta-analysis. Eur J Nucl Med Mol Imaging. 2015;42:241–51.
Chen J, Jiang CC, Jin L, et al. Regulation of PD-1:a novel role of pro-survival signalling in cancer. Ann Oncol. 2016;27:409–16.
Noman MZ, Desantis G, Janji B, et al. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J Exp Med. 2014;211:781–90.
Kaira K, Serizawa M, Koh Y, et al. Biological significance of 18F-FDG uptake on PET in patients with non-small-cell lung cancer. Lung Cancer. 2014;83:197–204.
Higuchi M, Owada Y, Inoue T, et al. FDG-PET in the evaluation of response to nivolumab in recurrent non-small-cell lung cancer. World J Surg Oncol. 2016;14:238.
Sachpekidis C, Hassel JC, Dimitrakopoulou-Strauss A. 18F-FDG PET/CT reveals disease remission in a patient with Ipilimumab-refractory advanced melanoma treated with Pembrolizumab. Clin Nucl Med. 2016;41:156–8.
England CG, Ehlerding EB, Hernandez R, et al. Preclinical pharmacokinetics and biodistribution studies of 89Zr-labeled Pembrolizumab. J Nucl Med. 2017;58:162–8.
Giglio BC, Fei H, Wang M, et al. Synthesis of 5-[18F]Fluoro-α-methyl tryptophan: new Trp based PET agents. Theranostics. 2017;7:1524–30.
Pauleit D, Stoffels G, Schaden W, et al. PET with O-(2-18F-fluoroethyl)-L-tyrosine in peripheral tumors: first clinical results. J Nucl Med. 2005;46:411–6.
Jager PL, Groen HJM, van der Leest A, et al. L-3-123I-iodo-a-methyl-L-tyrosine SPECT in non-small cell lung cancer: preliminary observations. J Nucl Med. 2001;42:579–85.
Kaira K, Oriuchi N, Otani Y, et al. Fluorine-18-α-methyltyrosine positron emission tomography for diagnosis and staging of lung cancer: a clinicopathologic study. Clin Cancer Res. 2007;13:6369–78.
Kaira K, Oriuchi N, Otani Y, et al. Diagnostic usefulness of fluorine-18-alpha-methyltyrosine positron emission tomography in combination with 18F-fluorodeoxyglucose in sarcoidosis patients. Chest. 2007;131:1019–27.
Acknowledgements
We thank Ms. Yuka Matsui for her technical assistance during the manuscript submission. We deeply appreciate the help provided by Ms. Yoko Tokumitsu of the Department of Outpatient Chemotherapy Center, Hidaka Hospital, Drs. Toshitaka Maeno, Kenichiro Hara, Yasuhiko Koga, and Akira Ono of the Department of Respiratory Medicine, and Drs. Toshiki Yajima and Takayuki Kosaka of the Department of Respiratory Surgery, Gunma University Hospital, for data collection and clinical advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
KK has received research grants and speaker honoraria from Ono Pharmaceutical Company and Bristol-Myers Company. All remaining authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Kaira, K., Higuchi, T., Naruse, I. et al. Metabolic activity by 18F–FDG-PET/CT is predictive of early response after nivolumab in previously treated NSCLC. Eur J Nucl Med Mol Imaging 45, 56–66 (2018). https://doi.org/10.1007/s00259-017-3806-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3806-1