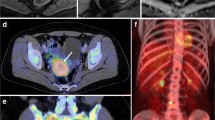
Abstract
Purpose
To determine whether the relative metabolic activity of pelvic or para-aortic LN compared with that of primary tumor measured by preoperative [18F]FDG PET/CT scan has prognostic value in patients with endometrioid endometrial carcinoma.
Methods
We retrospectively reviewed patients with endometrioid endometrial carcinoma who underwent preoperative [18F]FDG PET/CT scans. Prognostic values of PET/CT-derived metabolic variables such as maximum standardized uptake value (SUV) of the primary endometrial carcinoma (SUVTumor) and LN (SUVLN), and the LN-to-endometrial carcinoma SUV ratio (SUVLN / SUVTumor) were assessed.
Results
Clinico-pathological data, imaging data, and treatment results were reviewed for 107 eligible patients. Median post-surgical follow-up was 23 months (range, 6–60), and 7 (6.5%) patients experienced recurrence. Regression analysis showed that SUVLN / SUVTumor (P < 0.001), SUVLN (P = 0.003), International Federation of Gynecology and Obstetrics (FIGO) stage (P = 0.006), and tumor grade (P = 0.011) were risk factors of recurrence. Multivariate regression analysis revealed that FIGO stage (P = 0.034) was the independent risk factor of recurrence. SUVLN / SUVTumor showed significant correlation with FIGO stage (P < 0.001), LN metastasis (P < 0.001), lymphovascular space invasion (P < 0.001), recurrence (P = 0.001), tumor grade (P < 0.001), and deep myometrial invasion of tumor (P = 0.022). Patient groups categorized by SUVLN / SUVTumor showed significant difference in progression-free survival (Log-rank test, P = 0.001).
Conclusions
Preoperative SUVLN / SUVTumor measured by [18F]FDG PET/CT was significantly associated with recurrence, and may become a novel prognostic factor in patients with endometrioid endometrial carcinoma.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. https://doi.org/10.3322/caac.21332.
Morrow CP, Bundy BN, Kurman RJ, Creasman WT, Heller P, Homesley HD, et al. Relationship between surgical-pathological risk factors and outcome in clinical stage I and II carcinoma of the endometrium: a gynecologic oncology group study. Gynecol Oncol. 1991;40(1):55–65.
Salvesen HB, Haldorsen IS, Trovik J. Markers for individualised therapy in endometrial carcinoma. Lancet Oncol. 2012;13(8):e353–61. https://doi.org/10.1016/S1470-2045(12)70213-9.
Boronow RC, Morrow CP, Creasman WT, Disaia PJ, Silverberg SG, Miller A, et al. Surgical staging in endometrial cancer: clinical-pathologic findings of a prospective study. Obstet Gynecol. 1984;63(6):825–32.
Amant F, Moerman P, Neven P, Timmerman D, Van Limbergen E, Vergote I. Endometrial cancer. Lancet. 2005;366(9484):491–505. https://doi.org/10.1016/S0140-6736(05)67063-8.
Tejerizo-Garcia A, Jimenez-Lopez JS, Munoz-Gonzalez JL, Bartolome-Sotillos S, Marqueta-Marques L, Lopez-Gonzalez G, et al. Overall survival and disease-free survival in endometrial cancer: prognostic factors in 276 patients. OncoTargets Ther. 2013;9:1305–13. https://doi.org/10.2147/OTT.S51532.
Creasman WT, Odicino F, Maisonneuve P, Quinn MA, Beller U, Benedet JL, et al. Carcinoma of the corpus uteri. FIGO 26th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet. 2006;95(Suppl 1):S105–43. https://doi.org/10.1016/S0020-7292(06)60031-3.
Antonsen SL, Loft A, Fisker R, Nielsen AL, Andersen ES, Hogdall E, et al. SUVmax of 18FDG PET/CT as a predictor of high-risk endometrial cancer patients. Gynecol Oncol. 2013;129(2):298–303. https://doi.org/10.1016/j.ygyno.2013.01.019.
Kitajima K, Kita M, Suzuki K, Senda M, Nakamoto Y, Sugimura K. Prognostic significance of SUVmax (maximum standardized uptake value) measured by [(1)(8)F]FDG PET/CT in endometrial cancer. Eur J Nucl Med Mol Imaging. 2012;39(5):840–5. https://doi.org/10.1007/s00259-011-2057-9.
Amit A, Schink J, Reiss A, Lowenstein L. PET/CT in gynecologic cancer: present applications and future prospects--a clinician’s perspective. Obstet Gynecol Clin N Am. 2011;38(1):1–21, vii. https://doi.org/10.1016/j.ogc.2011.02.001.
Tsujikawa T, Tsuchida T, Yoshida Y, Kurokawa T, Kiyono Y, Okazawa H, et al. Role of PET/CT in gynecological tumors based on the revised FIGO staging classification. Clin Nucl Med. 2011;36(9):e114–8. https://doi.org/10.1097/RLU.0b013e31821c9a9a.
Horowitz NS, Dehdashti F, Herzog TJ, Rader JS, Powell MA, Gibb RK, et al. Prospective evaluation of FDG-PET for detecting pelvic and para-aortic lymph node metastasis in uterine corpus cancer. Gynecol Oncol. 2004;95(3):546–51. https://doi.org/10.1016/j.ygyno.2004.08.009.
Crivellaro C, Signorelli M, Guerra L, De Ponti E, Pirovano C, Fruscio R, et al. Tailoring systematic lymphadenectomy in high-risk clinical early stage endometrial cancer: the role of 18F-FDG PET/CT. Gynecol Oncol. 2013;130(2):306–11. https://doi.org/10.1016/j.ygyno.2013.05.011.
Chang MC, Chen JH, Liang JA, Yang KT, Cheng KY, Kao CH. 18F-FDG PET or PET/CT for detection of metastatic lymph nodes in patients with endometrial cancer: a systematic review and meta-analysis. Eur J Radiol. 2012;81(11):3511–7. https://doi.org/10.1016/j.ejrad.2012.01.024.
Ghooshkhanei H, Treglia G, Sabouri G, Davoodi R, Sadeghi R. Risk stratification and prognosis determination using (18)F-FDG PET imaging in endometrial cancer patients: a systematic review and meta-analysis. Gynecol Oncol. 2014;132(3):669–76. https://doi.org/10.1016/j.ygyno.2014.01.039.
Nakamura K, Kodama J, Okumura Y, Hongo A, Kanazawa S, Hiramatsu Y. The SUVmax of 18F-FDG PET correlates with histological grade in endometrial cancer. Int J Gynecol Cancer. 2010;20(1):110–5. https://doi.org/10.1111/IGC.0b013e3181c3a288.
Chung HH, Cheon GJ, Kim HS, Kim JW, Park NH, Song YS. Preoperative PET/CT standardized FDG uptake values of pelvic lymph nodes as a significant prognostic factor in patients with endometrial cancer. Eur J Nucl Med Mol Imaging. 2014;41(9):1793–9. https://doi.org/10.1007/s00259-014-2775-x.
Budiawan H, Cheon GJ, Im HJ, Lee SJ, Paeng JC, Kang KW, et al. Heterogeneity analysis of (18)F-FDG uptake in differentiating between metastatic and inflammatory lymph nodes in adenocarcinoma of the lung: comparison with other parameters and its application in a clinical setting. Nucl Med Mol Imaging. 2013;47(4):232–41. https://doi.org/10.1007/s13139-013-0216-6.
Nakamura K, Hongo A, Kodama J, Hiramatsu Y. The measurement of SUVmax of the primary tumor is predictive of prognosis for patients with endometrial cancer. Gynecol Oncol. 2011;123(1):82–7. https://doi.org/10.1016/j.ygyno.2011.06.026.
Kakhki VR, Shahriari S, Treglia G, Hasanzadeh M, Zakavi SR, Yousefi Z, et al. Diagnostic performance of fluorine 18 fluorodeoxyglucose positron emission tomography imaging for detection of primary lesion and staging of endometrial cancer patients: systematic review and meta-analysis of the literature. Int J Gynecol Cancer. 2013;23(9):1536–43. https://doi.org/10.1097/IGC.0000000000000003.
Haldorsen IS, Stefansson I, Gruner R, Husby JA, Magnussen IJ, Werner HM, et al. Increased microvascular proliferation is negatively correlated to tumour blood flow and is associated with unfavourable outcome in endometrial carcinomas. Br J Cancer. 2014;110(1):107–14. https://doi.org/10.1038/bjc.2013.694.
Walentowicz-Sadlecka M, Malkowski B, Walentowicz P, Sadlecki P, Marszalek A, Pietrzak T, et al. The preoperative maximum standardized uptake value measured by 18F-FDG PET/CT as an independent prognostic factor of overall survival in endometrial cancer patients. Biomed Res Int. 2014;2014:234813. https://doi.org/10.1155/2014/234813.
Haldorsen IS, Salvesen HB. What is the best preoperative imaging for endometrial cancer? Curr Oncol Rep. 2016;18(4):25. https://doi.org/10.1007/s11912-016-0506-0.
Kitajima K, Suenaga Y, Ueno Y, Maeda T, Ebina Y, Yamada H, et al. Preoperative risk stratification using metabolic parameters of (18)F-FDG PET/CT in patients with endometrial cancer. Eur J Nucl Med Mol Imaging. 2015;42(8):1268–75. https://doi.org/10.1007/s00259-015-3037-2.
Horree N, van Diest PJ, van der Groep P, Sie-Go DM, Heintz AP. Hypoxia and angiogenesis in endometrioid endometrial carcinogenesis. Cell Oncol. 2007;29(3):219–27.
Lee DW, Chong GO, Lee YH, Hong DG, Cho YL, Jeong SY, et al. Role of SUVmax and GLUT-1 expression in determining tumor aggressiveness in patients with clinical stage I endometrioid endometrial cancer. Int J Gynecol Cancer. 2015;25(5):843–9. https://doi.org/10.1097/IGC.0000000000000301.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. https://doi.org/10.1016/j.cell.2011.02.013.
Berg A, Fasmer KE, Mauland KK, Ytre-Hauge S, Hoivik EA, Husby JA, et al. Tissue and imaging biomarkers for hypoxia predict poor outcome in endometrial cancer. Oncotarget. 2016;7(43):69844–56. https://doi.org/10.18632/oncotarget.12004.
Tsujikawa T, Yoshida Y, Kudo T, Kiyono Y, Kurokawa T, Kobayashi M, et al. Functional images reflect aggressiveness of endometrial carcinoma: estrogen receptor expression combined with 18F-FDG PET. J Nucl Med. 2009;50(10):1598–604. https://doi.org/10.2967/jnumed.108.060145.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of retrospective study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Chung, H.H., Cheon, G.J., Kim, JW. et al. Prognostic value of lymph node-to-primary tumor standardized uptake value ratio in endometrioid endometrial carcinoma. Eur J Nucl Med Mol Imaging 45, 47–55 (2018). https://doi.org/10.1007/s00259-017-3805-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3805-2