Abstract
Purpose
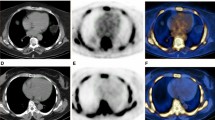
Our aim was to assess whether 18F-NaF PET/CT is able to predict progression of the CT calcium score.
Methods
Between August 2007 and November 2015, 34 patients (18 women, 16 men; age, mean ± standard deviation, 57.5 ± 13.9 years; age range 19–78 years) with malignancy or orthopaedic disease were enrolled in this study, with approximately 1-year follow-up data. Baseline and follow-up CT images were retrospectively evaluated for the presence of calcification sites in major vessel walls. The maximum and mean CT values (CTmax and CTmean, in Hounsfield units), calcification volumetric score (CVS, in cubic millimetres) and Agatston units score (AU) were evaluated for each site. Subsequent changes in CTmax, CTmean, CVS and AU were calculated and expressed as ΔCTmax, ΔCTmean, ΔCVS and ΔAU, respectively. We then evaluated the relationship between 18F-NaF uptake (using the maximum target-to-background ratio, TBRmax, and the maximum blood-subtracted 18F-NaF activity, bsNaFmax, which was obtained by subtracting the SUVmax of each calcified plaque lesion and NaF-avid site from the SUVmean in the right atrium blood pool) and the change in calcified plaque volume and characteristics obtained after 1 year.
Results
We detected and analysed 182 calcified plaque sites and 96 hot spots on major vessel walls. 18F-NaF uptake showed very weak correlations with CTmax, CTmean, CVS, CVS after 1 year, AU and AU after 1 year on both baseline and follow-up PET/CT scans for each site. 18F-NaF uptake showed no correlation with ΔCTmax or ΔCTmean. However, there was a significant correlation between the intensity of 18F-NaF uptake and ΔCVS and ΔAU.
Conclusion
18F-NaF uptake has a strong correlation with calcium score progression which was a predictor of future cardiovascular disease risk. PET/CT using 18F-NaF may be able to predict calcium score progression which is known to be the major characteristic of atherosclerosis.



Similar content being viewed by others
References
Chen W, Dilsizian V. Targeted PET/CT imaging of vulnerable atherosclerotic plaques: microcalcification with sodium fluoride and inflammation with fluorodeoxyglucose. Curr Cardiol Rep. 2013;15:364.
Hansson GK, Libby P, Tabas I. Inflammation and plaque vulnerability. J Intern Med. 2015;278:483–493.
Shah PK. Biomarkers of plaque instability. Curr Cardiol Rep. 2014;16:547.
Budoff MJ, Shaw LJ, Liu ST, Weinstein SR, Mosler TP, Tseng PH, et al. Long-term prognosis associated with coronary calcification: observations from a registry of 25,253 patients. J Am Coll Cardiol. 2007;49:1860–1870.
Forbang NI, McClelland RL, Remigio-Baker RA, Allison MA, Sandfort V, Michos ED, et al. Associations of cardiovascular disease risk factors with abdominal aortic calcium volume and density: the Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis. 2016;255:54–58.
Bastos Gonçalves F, Voûte MT, Hoeks SE, Chonchol MB, Boersma EE, Stolker RJ, et al. Calcification of the abdominal aorta as an independent predictor of cardiovascular events: a meta-analysis. Heart. 2012;98:988–994.
Huang H, Virmani R, Younis H, Burke AP, Kamm RD, Lee RT. The impact of calcification on the biomechanical stability of atherosclerotic plaques. Circulation. 2001;103:1051–1056.
Vengrenyuk Y, Carlier S, Xanthos S, Cardoso L, Ganatos P, Virmani R, et al. A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc Natl Acad Sci U S A. 2006;103:14678–14683.
Davaine JM, Quillard T, Chatelais M, Guilbaud F, Brion R, Guyomarch B, et al. Bone like arterial calcification in femoral atherosclerotic lesions: prevalence and role of osteoprotegerin and pericytes. Eur J Vasc Endovasc Surg. 2016;51:259–267.
New SE, Aikawa E. Molecular imaging insights into early inflammatory stages of arterial and aortic valve calcification. Circ Res. 2011;108:1381–1391.
Haust MD, More RH, Bencosme SA, Balis JU. Electron microscopic studies in human atherosclerosis extracellular elements in aortic dots and streaks. Exp Mol Pathol. 1967;6:300–313.
Zechariah A, ElAli A, Hagemann N, Jin F, Doeppner TR, Helfrich I, et al. Hyperlipidemia attenuates vascular endothelial growth factor-induced angiogenesis, impairs cerebral blood flow, and disturbs stroke recovery via decreased pericyte coverage of brain endothelial cells. Arterioscler Thromb Vasc Biol. 2013;33:1561–1567.
Xu Y, Arai H, Murayama T, Kita T, Yokode M. Hypercholesterolemia contributes to the development of atherosclerosis and vascular remodeling by recruiting bone marrow-derived cells in cuff-induced vascular injury. Biochem Biophys Res Commun. 2007;363:782–787.
Insull Jr W. The pathology of atherosclerosis: plaque development and plaque responses to medical treatment. Am J Med. 2009;122:S3–S14.
Nadra I, Mason JC, Philippidis P, Florey O, Smythe CD, McCarthy GM, et al. Proinflammatory activation of macrophages by basic calcium phosphate crystals via protein kinase C and MAP kinase pathways: a vicious cycle of inflammation and arterial calcification? Circ Res. 2005;96:1248–1256.
Boström K. Proinflammatory vascular calcification. Circ Res. 2005;96:1219–20.
Ewence AE, Bootman M, Roderick HL, Skepper JN, McCarthy G, Epple M, et al. Calcium phosphate crystals induce cell death in human vascular smooth muscle cells: a potential mechanism in atherosclerotic plaque destabilization. Circ Res. 2008;103:e28–e34.
Hutcheson JD, Maldonado N, Aikawa E. Small entities with large impact: microcalcifications and atherosclerotic plaque vulnerability. Curr Opin Lipidol. 2014;25:327–332.
Aikawa E, Nahrendorf M, Figueiredo JL, Swirski FK, Shtatland T, Kohler RH, et al. Osteogenesis associates with inflammation in early-stage atherosclerosis evaluated by molecular imaging in vivo. Circulation. 2007;116:2841–50.
Shanahan CM. Inflammation ushers in calcification: a cycle of damage and protection? Circulation. 2007;116:2782–2785.
Chen W, Dilsizian V. (18)F-fluorodeoxyglucose PET imaging of coronary atherosclerosis and plaque inflammation. Curr Cardiol Rep. 2010;12:179–184.
Dilsizian V, Narula J. Putting the face to a name: concurrent assessment of vascular morphology and biology. JACC Cardiovasc Imaging. 2009;2:1243–1244.
Irkle A, Vesey AT, Lewis DY, Skepper JN, Bird JL, Dweck MR, et al. Identifying active vascular microcalcification by (18)F-sodium fluoride positron emission tomography. Nat Commun. 2015;6:7495.
Dweck MR, Jenkins WS, Vesey AT, Pringle MA, Chin CW, Malley TS, et al. 18F-sodium fluoride uptake is a marker of active calcification and disease progression in patients with aortic stenosis. Circ Cardiovasc Imaging. 2014;7:371–378.
Jenkins WS, Vesey AT, Shah AS, Pawade TA, Chin CW, White AC, et al. Valvular (18)F-fluoride and (18)F-fluorodeoxyglucose uptake predict disease progression and clinical outcome in patients with aortic stenosis. J Am Coll Cardiol. 2015;66:1200–1201.
Pawade TA, Cartlidge TR, Jenkins WS, Adamson PD, Robson P, Lucatelli C, et al. Optimization and reproducibility of aortic valve 18F-fluoride positron emission tomography in patients with aortic stenosis. Circ Cardiovasc Imaging. 2016;9, e005131.
Blomberg BA, Thomassen A, de Jong PA, Simonsen JA, Lam MG, Nielsen AL, et al. Impact of personal characteristics and technical factors on quantification of sodium 18F-fluoride uptake in human arteries: prospective evaluation of healthy subjects. J Nucl Med. 2015;56:1534–1540.
Callister TQ, Cooil B, Raya SP, Lippolis NJ, Russo DJ, Raggi P. Coronary artery disease: improved reproducibility of calcium scoring with an electron-beam CT volumetric method. Radiology. 1998;208:807–814.
Agatston A, Janowitz W, Hildner FJ, Zusmer NR, Viamonte Jr M, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15:827–832.
Forbang NI, Michos ED, McClelland RL, Remigio-Baker RA, Allison MA, Sandfort V, et al. Greater volume but not higher density of abdominal aortic calcium is associated with increased cardiovascular disease risk: MESA (Multi-Ethnic Study of Atherosclerosis). Circ Cardiovasc Imaging. 2016;9, e005138.
Acknowledgments
We thank Shintaro Nawata, M.D., and Tomohiro Kaneta, M.D., Ph.D., Department of Radiology, Yokohama City University Graduate School of Medicine, for their help in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Ishiwata, Y., Kaneta, T., Nawata, S. et al. Quantification of temporal changes in calcium score in active atherosclerotic plaque in major vessels by 18F-sodium fluoride PET/CT. Eur J Nucl Med Mol Imaging 44, 1529–1537 (2017). https://doi.org/10.1007/s00259-017-3680-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3680-x