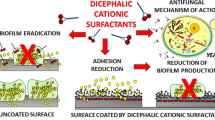
Abstract
Fungi—in being responsible for causing diseases in animals and humans as well as environmental contaminations in health and storage facilities—represent a serious concern to health security. Surfactants are a group of chemical compounds used in a broad spectrum of applications. The recently considered potential employment of cationic surfactants as antifungal or fungistatic agents has become a prominent issue in the development of antifungal strategies, especially if such surface-active agents can be synthesized in an eco-friendly manner. In this review, we describe the antifungal effect and the reported mechanisms of action of several types of cationic surfactants and also include a discussion of the contribution of these surfactants to the inhibition of yeast-based-biofilm formation. Furthermore, the putative mechanism of arginine-based tensioactive compounds as antifungal agents and their applications are also analyzed.



Similar content being viewed by others
References
Acmite Mark (2016) Global surfactant market. Market report. In: Acmite Mark. Intell. http://www.acmite.com/brochure/Brochure-Global-Surfactant-Market-Report.pdf. Accessed 6 Sept 2018
Aiad I, El-Sukkary MM, Soliman EA, El-Awady MY, Shaban SM (2014a) Inhibition of mild steel corrosion in acidic medium by some cationic surfactants. J Ind Eng Chem 20:3524–3535. https://doi.org/10.1016/j.jiec.2013.12.045
Aiad I, El-Sukkary MM, Soliman EA, El-Awady MY, Shaban SM (2014b) Characterization, surface properties and biological activity of new prepared cationic surfactants. J Ind Eng Chem 20:1633–1640. https://doi.org/10.1016/j.jiec.2013.08.010
Anderson SE, Shane H, Long C, Lukomska E, Meade BJ, Marshall NB (2016) Evaluation of the irritancy and hypersensitivity potential following topical application of didecyldimethylammonium chloride. J Immunotoxicol 13:557–566. https://doi.org/10.3109/1547691X.2016.1140854
Atanasov KE, Barboza-Barquero L, Tiburcio AF, Alcázar R (2016) Genome wide association mapping for the tolerance to the polyamine oxidase inhibitor guazatine in Arabidopsis thaliana. Front Plant Sci 7:1–11. https://doi.org/10.3389/fpls.2016.00401
Bahar AA, Ren D (2013) Antimicrobial peptides. Pharmaceuticals 6:1543–1575. https://doi.org/10.3390/ph6121543
Balba H (2007) Review of strobilurin fungicide chemicals. J Environ Sci Health B 42:441–451. https://doi.org/10.1080/03601230701316465
Baldridge JW, Michalow A (2004) Biofilm reduction in crossflow filtration systems. US Grant US6699391B2. 1–7
Bechara C, Sagan S (2013) Cell-penetrating peptides: 20 years later, where do we stand? FEBS Lett 587:1693–1702. https://doi.org/10.1016/j.febslet.2013.04.031
Benyagoub M, Bélanger RR (1995) Development of a mutant strain of Sporothrix flocculosa with resistance to dodemorph-acetate. Phytopathology 85:766–770. https://doi.org/10.1094/Phyto-85-766
Bordes R, Holmberg K (2015) Amino acid-based surfactants—do they deserve more attention? Adv Colloid Interf Sci 222:79–91. https://doi.org/10.1016/j.cis.2014.10.013
Brycki B, Dega-Szafran Z, Mirska I (2010) Synthesis and antimicrobial activities of some quaternary morpholinium chlorides. Polish J Microbiol 59:49–53
Bseiso E, Nasr M, Abd El Gawad N, Sammour O (2015) Recent advances in topical formulation carriers of antifungal agents. Indian J Dermatol Venereol Leprol 81:457. https://doi.org/10.4103/0378-6323.162328
Castillo JA (2006) Comparative study of the antimicrobial activity of bis(N-caproyl-L-arginine)-1,3-propanediamine dihydrochloride and chlorhexidine dihydrochloride against Staphylococcus aureus and Escherichia coli. J Antimicrob Chemother 57:691–698. https://doi.org/10.1093/jac/dkl012
Castillo JA, Infante MR, Manresa A, Vinardell MP, Mitjans M, Clapés P (2006) Chemoenzymatic synthesis and antimicrobial and haemolytic activities of amphiphilic bis(phenylacetylarginine) derivatives. ChemMedChem 1:1091–1098. https://doi.org/10.1002/cmdc.200600148
Chandra N, Tyagi VK (2013) Synthesis, properties, and applications of amino acids based surfactants: a review. J Dispers Sci Technol 34:800–808. https://doi.org/10.1080/01932691.2012.695967
Chen Y, Geurts M, Sjollema SB, Kramer NI, Hermens JLM, Droge STJ (2014) Acute toxicity of the cationic surfactant C12-benzalkonium in different bioassays: how test design affects bioavailability and effect concentrations. Environ Toxicol Chem 33:606–615. https://doi.org/10.1002/etc.2465
Codling CE, Maillard JY, Russell AD (2003) Aspects of the antimicrobial mechanisms of action of a polyquaternium and an amidoamine. J Antimicrob Chemother 51:1153–1158. https://doi.org/10.1093/jac/dkg228
Colomer A, Pinazo A, Manresa MA, Vinardell MP, Mitjans M, Infante MR, Pérez L (2011) Cationic surfactants derived from lysine: effects of their structure and charge type on antimicrobial and hemolytic activities. J Med Chem 54:989–1002. https://doi.org/10.1021/jm101315k
Dancer SJ (2009) The role of environmental cleaning in the control of hospital-acquired infection. J Hosp Infect 73:378–385. https://doi.org/10.1016/j.jhin.2009.03.030
Deepak SA, Basavaraju P, Chaluvaraju G, Shetty NP, Oros G, Shekar H (2006) Developmental stage response of pearl millet downy mildew (Sclerospora graminicola) to fungicides. Appl Ecol Environ Res 4:125–149. https://doi.org/10.15666/aeer/0402_125149
Desai JV, Mitchell AP, Andes DR (2014) Fungal biofilms, drug resistance, and recurrent infection. Cold Spring Harb Perspect Med 4:a019729. https://doi.org/10.1101/cshperspect.a019729
Di Nica V, Gallet J, Villa S, Mezzanotte V (2017) Toxicity of quaternary ammonium compounds (QACs) as single compounds and mixtures to aquatic non-target microorganisms: experimental data and predictive models. Ecotoxicol Environ Saf 142:567–577. https://doi.org/10.1016/j.ecoenv.2017.04.028
Dreassi E, Zizzari AT, D’Arezzo S, Visca P, Botta M (2007) Analysis of guazatine mixture by LC and LC–MS and antimycotic activity determination of principal components. J Pharm Biomed Anal 43:1499–1506. https://doi.org/10.1016/j.jpba.2006.10.029
Dupont S, Lemetais G, Ferreira T, Cayot P, Gervais P, Beney L (2012) Ergosterol biosynthesis: a fungal pathway for life on land? Evolution (N Y):1–8. https://doi.org/10.5061/dryad.pd28pm7n
Dusane DH, Dam S, Nancharaiah YV, Kumar A, Venugopalan VP, Zinjarde SS (2012) Disruption of Yarrowia lipolytica biofilms by rhamnolipid biosurfactant. Aquat Biosyst 8:17. https://doi.org/10.1186/2046-9063-8-17
Fait ME, Garrote GL, Clapés P, Tanco S, Lorenzo J, Morcelle SR (2015) Biocatalytic synthesis, antimicrobial properties and toxicity studies of arginine derivative surfactants. Amino Acids 47:1465–1477. https://doi.org/10.1007/s00726-015-1979-0
Fait ME, da Costa H PS, Freitas C DT, Bakás L, Morcelle SR (2018) Antifungal activity of arginine-based surfactants. Curr Bioact Compd 14:1–9. https://doi.org/10.2174/1573407214666180131161302
Florence AT, Atwood D (2006) Surfactants. In: Physicochemical principles of pharmacy, 4th edn. Pharmaceutical Press, London
Gaikwad KK, Lee SM, Lee JS, Lee YS (2017) Development of antimicrobial polyolefin films containing lauroyl arginate and their use in the packaging of strawberries. J Food Meas Charact 11:1706–1716. https://doi.org/10.1007/s11694-017-9551-0
Gecol H, Ergican E, Fuchs A (2004) Molecular level separation of arsenic (V) from water using cationic surfactant micelles and ultrafiltration membrane. J Memb Sci 241:105–119. https://doi.org/10.1016/j.memsci.2004.04.026
Gerez CL, Carbajo MS, Rollán G, Torres Leal G, Font de Valdez G (2010) Inhibition of citrus fungal pathogens by using lactic acid bacteria. J Food Sci 75:M354–M359. https://doi.org/10.1111/j.1750-3841.2010.01671.x
Gerova M, Rodrigues F, Lamère J-F, Dobrev A, Fery-Forgues S (2008) Self-assembly properties of some chiral N-palmitoyl amino acid surfactants in aqueous solution. J Colloid Interface Sci 319:526–533. https://doi.org/10.1016/j.jcis.2007.12.004
Gilbert P, Moore LE (2005) Cationic antiseptics: diversity of action under a common epithet. J Appl Microbiol 99:703–715. https://doi.org/10.1111/j.1365-2672.2005.02664.x
Gong T, Zhang X, Li Y, Xian Q (2016) Formation and toxicity of halogenated disinfection byproducts resulting from linear alkylbenzene sulfonates. Chemosphere 149:70–75. https://doi.org/10.1016/j.chemosphere.2016.01.067
González-Jaramillo LM, Aranda FJ, Teruel JA, Villegas-Escobar V, Ortiz A (2017) Antimycotic activity of fengycin C biosurfactant and its interaction with phosphatidylcholine model membranes. Colloids Surf B Biointerfaces 156:114–122. https://doi.org/10.1016/j.colsurfb.2017.05.021
Grand View Research (2016) Surfactants market revenue is expected to grow at CAGR of 5.4% owing to enhanced demand in personal care industry till 2022: Grand View Research, Inc. In: Gd. View Res. http://globenewswire.com/news-release/2016/02/11/809799/0/en/Surfactants-Market-Revenue-Is-Expected-To-Grow-At-CAGR-Of-5-4-Owing-To-Enhanced-Demand-In-Personal-Care-Industry-Till-2022-Grand-View-Research-Inc.html. Accessed 2 Sept 2018
Gupta AK, Ahmad I, Summerbell RC (2002) Fungicidal activities of commonly used disinfectants and antifungal pharmaceutical spray preparations against clinical strains of Aspergillus and Candida species. Med Mycol 40:201–208. https://doi.org/10.1080/mmy.40.2.201.208
Hanson JR (2008) Chemistry of fungi, RSC Publis. Royal Society of Chemistry, Cambridge
Hawser SP, Douglas LJ (1994) Biofilm formation by Candida species on the surface of catheter materials in vitro. Infect Immun 62:915–921
Hay R (2017) Superficial fungal infections. Med (United Kingdom) 45:707–710. https://doi.org/10.1016/j.mpmed.2017.08.006
Hayes DG (2009) Surfactants overview and industrial state-of-the-art. In: Hayes DG, Kitamoto D, Solaiman D, Ashby RD (eds) Biobased surfactants and detergents: synthesis, properties, and applications. AOCS Press, Illinois, pp 3–28
He K, Xu L (2017) Unique mixtures of anionic/cationic surfactants: a new approach to enhance surfactant performance in liquids-rich shale reservoirs. SPE Prod Oper 33:1–8. https://doi.org/10.2118/184515-PA
Hegazy MA, El-Etre AY, El-Shafaie M, Berry KM (2016) Novel cationic surfactants for corrosion inhibition of carbon steel pipelines in oil and gas wells applications. J Mol Liq 214:347–356. https://doi.org/10.1016/j.molliq.2015.11.047
Hegstad K, Langsrud S, Lunestad BT, Scheie AA, Sunde M, Yazdankhah SP (2010) Does the wide use of quaternary ammonium compounds enhance the selection and spread of antimicrobial resistance and thus threaten our health? Microb Drug Resist 16:91–104. https://doi.org/10.1089/mdr.2009.0120
Hollis CG, Terry JP, Jaquess PA (1995) Methods for removing biofilm from or preventing buildup thereof on surfaces in industrial water systems. 1–10
Holtappels M, Swinnen E, De Groef L, Wuyts J, Moons L, Lagrou K, Van Dijck P, Kucharíková S (2017) Antifungal activity of oleylphosphocholine on in vitro and in vitro Candida albicans biofilms. Antimicrob Agents Chemother 62:e01767–e01717. https://doi.org/10.1128/AAC.01767-17
Infante MR, Pérez L, Morán C, Pons R, Pinazo A (2009) Synthesis, aggregation properties, and applications of biosurfactants derived from arginine. In: Hayes DG, Kitamoto D, Solaiman D, Ashby R (eds) Biobased surfactants and detergents: synthesis, properties, and applications. AOCS Press, Illinois, pp 351–387
Isaac J, Scheinman PL (2017) Benzalkonium chloride. Dermatitis 28:346–352. https://doi.org/10.1097/DER.0000000000000316
Jabra-Rizk MA, Falkler WA, Meiller TF (2004) Fungal biofilms and drug resistance. Emerg Infect Dis 10:14–19. https://doi.org/10.3201/eid1001.030119
Jennings MC, Minbiole KPC, Wuest WM (2015) Quaternary ammonium compounds: an antimicrobial mainstay and platform for innovation to address bacterial resistance. ACS Infect Dis 1:288–303. https://doi.org/10.1021/acsinfecdis.5b00047
Jessop PG, Ahmadpour F, Buczynski MA, Burns TJ, Green II NB, Korwin R, Long D, Massad SK, Manley JB, Omidbakhsh N, Pearl R, Pereira S, Predale RA, Sliva PG, VanderBilt H, Weller S, Wolf MH (2015) Opportunities for greener alternatives in chemical formulations. Green Chem 17:2664–2678. https://doi.org/10.1039/C4GC02261K
Jiang Q, Yue D, Nie Y, Xu X, He Y, Zhang S, Wagner E, Gu Z (2016) Specially-made lipid-based assemblies for improving transmembrane gene delivery: comparison of basic amino acid residue rich periphery. Mol Pharm 13:1809–1821. https://doi.org/10.1021/acs.molpharmaceut.5b00967
Johansson J, Somasundarau P (eds) (2007) Handbook for cleaning/decontamination of surfaces. Elsevier, Amsterdam
Kanazawa A, Ikeda T, Endo T (1995) A novel approach to mode of action of cationic biocides: morphological effect on antibacterial activity. J Appl Bacteriol 78:55–60. https://doi.org/10.1111/j.1365-2672.1995.tb01673.x
Labib ME, Lai C-Y (2000) Cleaning method for removing biofilm and debris from lines and tubing. 1–10
Lavorgna M, Russo C, D’Abrosca B, Parrella A, Isidori M (2016) Toxicity and genotoxicity of the quaternary ammonium compound benzalkonium chloride (BAC) using Daphnia magna and Ceriodaphnia dubia as model systems. Environ Pollut 210:34–39. https://doi.org/10.1016/j.envpol.2015.11.042
Li Z, Alessi D, Zhang P, Bowman RS (2002) Organo-illite as a low permeability sorbent to retard migration of anionic contaminants. J Environ Eng 128:583–587. https://doi.org/10.1061/(ASCE)0733-9372(2002)128:7(583)
Li Z, Willms CA, Kniola K (2003) Removal of anionic contaminants using surfactant-modified palygorskite and sepiolite. Clay Clay Miner 51:445–451. https://doi.org/10.1346/CCMN.2003.0510411
Li Y, Zhang W, Kong B, Puerto M, Bao X, Sha O, Shen Z, Yang Y, Liu Y, Gu S, Miller C, Hirasaki GJ (2014) Mixtures of anionic-cationic surfactants: a new approach for enhanced oil recovery in low-salinity, high-temperature sandstone reservoir. In: SPE Improved Oil Recovery Symposium. Society of Petroleum Engineers, pp 1164–1177
Löffler J, Einsele H, Hebart H, Schumacher U, Hrastnik C, Daum G (2000) Phospholipid and sterol analysis of plasma membranes of azole-resistant Candida albicans strains. FEMS Microbiol Lett 185:59–63. https://doi.org/10.1111/j.1574-6968.2000.tb09040.x
Lozano N, Pérez L, Pons R, Pinazo A (2011) Diacyl glycerol arginine-based surfactants: biological and physicochemical properties of catanionic formulations. Amino Acids 40:721–729. https://doi.org/10.1007/s00726-010-0710-4
Luz C, Netto MCB, Rocha LFN (2007) In vitro susceptibility to fungicides by invertebrate-pathogenic and saprobic fungi. Mycopathologia 164:39–47. https://doi.org/10.1007/s11046-007-9020-0
Mahmoud YA (2016) Fungal keratitis efficient treatments using surface active agents (cetrimide): an overview. J Bacteriol Mycol Open Access 2:3–7. https://doi.org/10.15406/jbmoa.2016.02.00026
Maiale SJ, Marina M, Sánchez DH, Pieckenstain FL, Ruiz OA (2008) In vitro and in vivo inhibition of plant polyamine oxidase activity by polyamine analogues. Phytochemistry 69:2552–2558. https://doi.org/10.1016/j.phytochem.2008.07.003
Maier C, Zeeb B, Weiss J (2014) Investigations into aggregate formation with oppositely charged oil-in-water emulsions at different pH values. Colloids Surfaces B Biointerfaces 117:368–375. https://doi.org/10.1016/j.colsurfb.2014.03.012
Maldonado MC, Santa Runco R, Navarro AR (2005) Isolation, identification and antifungal susceptibility of lemon pathogenic and non pathogenic fungi. Rev Iberoam Micol 22:57–59. https://doi.org/10.1016/S1130-1406(05)70009-7
Mallo AC, Nitiu DS, Elíades LA, Saparrat MCN (2017) Fungal degradation of cellulosic materials used as support for cultural heritage. Int J Conserv Sci 8:619–632
Manrique Y, Gibis M, Schmidt H, Weiss J (2017) Influence of application sequence and timing of eugenol and lauric arginate (LAE) on survival of spoilage organisms. Food Microbiol 64:210–218. https://doi.org/10.1016/j.fm.2017.01.002
Mao X, Jiang R, Xiao W, Yu J (2015) Use of surfactants for the remediation of contaminated soils: a review. J Hazard Mater 285:419–435. https://doi.org/10.1016/j.jhazmat.2014.12.009
Messier C, Epifano F, Genovese S, Grenier D (2011) Inhibition of Candida albicans biofilm formation and yeast-hyphal transition by 4-hydroxycordoin. Phytomedicine 18:380–383. https://doi.org/10.1016/j.phymed.2011.01.013
Morán C, Clapés P, Comelles F, García T, Pérez L, Vinardell P, Mitjans M, Infante MR (2001) Chemical structure/property relationship in single-chain arginine surfactants. Langmuir 17:5071–5075. https://doi.org/10.1021/la010375d
MycoBank (2018) Fungal databases, nomenclature & species banks. International Mycological Association. http://www.mycobank.org/. Accessed 15 Sept 2018
Negm NA, Mohamed AS (2008) Synthesis, characterization and biological activity of sugar-based gemini cationic amphiphiles. J Surfactant Deterg 11:215–221. https://doi.org/10.1007/s11743-008-1071-9
Nogueira DR, Scheeren LE, Macedo LB, Marcolino AIP, Pilar Vinardell M, Mitjans M, Rosa Infante M, Farooqi AA, Rolim CMB (2015) Inclusion of a pH-responsive amino acid-based amphiphile in methotrexate-loaded chitosan nanoparticles as a delivery strategy in cancer therapy. Amino Acids 48:157–168. https://doi.org/10.1007/s00726-015-2075-1
Obłąk E, Piecuch A, Krasowska A, Łuczyński J (2013) Antifungal activity of gemini quaternary ammonium salts. Microbiol Res 168:630–638. https://doi.org/10.1016/j.micres.2013.06.001
Obłąk E, Piecuch A, Dworniczek E, Olejniczak T (2015) The influence of biodegradable gemini surfactants, N,N’-bis(1-decyloxy-1-oxopronan-2-yl)-N,N,N’,N’ tetramethylpropane-1,3-diammonium dibromide and N,N’-bis(1-dodecyloxy-1-oxopronan-2-yl) N,N,N’,N’-tetramethylethane-1,2-diammonium dibromide. Fungal Biof J Oleo Sci 64:527–537. https://doi.org/10.5650/jos.ess14195
Occams Business Research & Consulting (2017) Global surfactants market insights, opportunity analysis, market shares and forecast 2017-2023. In: Data, Mark Anal http://www.occamsresearch.com/surfactants-market. Accessed 2 Oct 2017
Palermo EF, Kuroda K (2010) Structural determinants of antimicrobial activity in polymers which mimic host defense peptides. Appl Microbiol Biotechnol 87:1605–1615. https://doi.org/10.1007/s00253-010-2687-z
Pang X, Chu C-C (2010) Synthesis, characterization and biodegradation of functionalized amino acid-based poly(ester amide)s. Biomaterials 31:3745–3754. https://doi.org/10.1016/j.biomaterials.2010.01.027
Peña LC, Argarañá MF, De Zan MM, Giorello A, Antuña S, Prieto CC, Veaute CMI, Müller DM (2017) New amphiphilic amino acid derivatives for efficient DNA transfection in vitro. Adv Chem Eng Sci 07:191–205. https://doi.org/10.4236/aces.2017.72014
Pérez L, Torres JL, Manresa A, Solans C, Infante MR (1996) Synthesis, aggregation, and biological properties of a new class of gemini cationic amphiphilic compounds from arginine, bis(Args) †. Langmuir 12:5296–5301. https://doi.org/10.1021/la960301f
Pérez L, Pinazo A, Teresa García M, Lozano M, Manresa A, Angelet M, Pilar Vinardell M, Mitjans M, Pons R, Rosa Infante M (2009) Cationic surfactants from lysine: synthesis, micellization and biological evaluation. Eur J Med Chem 44:1884–1892. https://doi.org/10.1016/j.ejmech.2008.11.003
Perfect JR (2017) The antifungal pipeline: a reality check. Nat Rev Drug Discov 16:603–616. https://doi.org/10.1038/nrd.2017.46
Pinazo A, Pons R, Pérez L, Infante MR (2011) Amino acids as raw material for biocompatible surfactants. Ind Eng Chem Res 50:4805–4817. https://doi.org/10.1021/ie1014348
Pinazo A, Manresa MA, Marques AM, Bustelo M, Espuny MJ, Pérez L (2016) Amino acid–based surfactants: new antimicrobial agents. Adv Colloid Interf Sci 228:17–39. https://doi.org/10.1016/j.cis.2015.11.007
Posadas JB, Comerio RM, Mini JI, Nussenbaum AL, Lecuona RE (2012) A novel dodine-free selective medium based on the use of cetyl trimethyl ammonium bromide (CTAB) to isolate Beauveria bassiana, Metarhizium anisopliae sensu lato and Paecilomyces lilacinus from soil. Mycologia 104:974–980. https://doi.org/10.3852/11-234
Raicu V (1998) Effects of cetyltrimethylammonium bromide (CTAB) surfactant upon the dielectric properties of yeast cells. Biochim Biophys Acta - Gen Subj 1379:7–15. https://doi.org/10.1016/S0304-4165(97)00056-1
Ramage G, Saville SP, Wickes BL, Lopez-Ribot JL (2002) Inhibition of Candida albicans biofilm formation by farnesol, a quorum-sensing molecule. Appl Environ Microbiol 68:5459–5463. https://doi.org/10.1128/AEM.68.11.5459-5463.2002
Rosa M, del Carmen Morán M, da Graça Miguel M, Lindman B (2007) The association of DNA and stable catanionic amino acid-based vesicles. Colloids Surfaces A Physicochem Eng Asp 301:361–375. https://doi.org/10.1016/j.colsurfa.2006.12.082
Sandle T, Vijayakumar R, Saleh Al Aboody M, Saravanakumar S (2014) In vitro fungicidal activity of biocides against pharmaceutical environmental fungal isolates. J Appl Microbiol 117:1267–1273. https://doi.org/10.1111/jam.12628
Sant DG, Tupe SG, Ramana CV, Deshpande MV (2016) Fungal cell membrane-promising drug target for antifungal therapy. J Appl Microbiol 121:1498–1510. https://doi.org/10.1111/jam.13301
Shaban SM, Aiad I, El-Sukkary MM, Soliman EA, El-Awady MY (2014) Synthesis, surface, thermodynamic properties and biological activity of dimethylaminopropylamine surfactants. J Ind Eng Chem 20:4194–4201. https://doi.org/10.1016/j.jiec.2014.01.020
Shaban SM, Aiad I, El-Sukkary MM, Soliman EA, El-Awady MY (2015a) Evaluation of some cationic surfactants based on dimethylaminopropylamine as corrosion inhibitors. J Ind Eng Chem 21:1029–1038. https://doi.org/10.1016/j.jiec.2014.05.012
Shaban SM, Aiad I, Fetouh HA, Maher A (2015b) Amidoamine double tailed cationic surfactant based on dimethylaminopropylamine: synthesis, characterization and evaluation as biocide. J Mol Liq 212:699–707. https://doi.org/10.1016/j.molliq.2015.10.024
Shirai A, Sumitomo T, Kurimoto M, Maseda H, Kourai H (2009) The mode of the antifungal activity of gemini-pyridinium salt against yeast. Biocontrol Sci 14:13–20. https://doi.org/10.4265/bio.14.13
Singare PU, Mhatre JD (2012) Cationic surfactants from arginine: synthesis and physicochemical properties. Am J Chem 2:186–190. https://doi.org/10.5923/j.chemistry.20120204.02
Singh A, Tyagi VK (2014) Arginine based novel cationic surfactants: a review. Tenside Surfactants Deterg 51:202–214. https://doi.org/10.3139/113.110299
Stenbæk J, Löf D, Falkman P, Jensen B, Cárdenas M (2017) An alternative anionic bio-sustainable anti-fungal agent: investigation of its mode of action on the fungal cell membrane. J Colloid Interface Sci 497:242–248. https://doi.org/10.1016/j.jcis.2017.03.018
Szkolnik M, Gilpatrick JD (1969) Apparent resistance of Venturia inaequalis to dodine in New York apple orchards. Plant Dis Report 53:861–864
Tavano L, Pinazo A, Abo-Riya M, Infante MRR, Manresa MAA, Muzzalupo R, Pérez L (2014) Cationic vesicles based on biocompatible diacyl glycerol-arginine surfactants: physicochemical properties, antimicrobial activity, encapsulation efficiency and drug release. Colloids Surf B Biointerfaces 120:160–167. https://doi.org/10.1016/j.colsurfb.2014.04.009
Tedersoo L, Sánchez-Ramírez S, Kõljalg U, Bahram M, Döring M, Schigel D, May T, Ryberg M, Abarenkov K (2018) High-level classification of the fungi and a tool for evolutionary ecological analyses. Fungal Divers 90:135–159. https://doi.org/10.1007/s13225-018-0401-0
Terjung N, Loeffler M, Gibis M, Salminen H, Hinrichs J, Weiss J (2014) Impact of lauric arginate application form on its antimicrobial activity in meat emulsions. Food Biophys 9:88–98. https://doi.org/10.1007/s11483-013-9321-4
Tezel U, Pavlostathis SG (2015) Quaternary ammonium disinfectants: microbial adaptation, degradation and ecology. Curr Opin Biotechnol 33:296–304. https://doi.org/10.1016/j.copbio.2015.03.018
Tóth A, Petróczy M, Hegedűs M, Nagy G, Lovász C, Ágoston J, Palkovics L (2012) Development of plant protection technology against sour cherry anthracnose. In: György J, Kövics GJ, Dávid I (eds) 6th International Plant Protection Symposium at University of Debrecen. Journal of agricultural sciences. Acta agraria debreceniensis, Debrecen, Hungary, pp 54–59
Tripathy DB, Mishra A, Clark J, Farmer T (2018) Synthesis, chemistry, physicochemical properties and industrial applications of amino acid surfactants: a review. Comptes Rendus Chim 21:112–130. https://doi.org/10.1016/j.crci.2017.11.005
Tsui C, Kong EF, Jabra-Rizk MA (2016) Pathogenesis of Candida albicans biofilm. Pathog Dis 74:ftw018. https://doi.org/10.1093/femspd/ftw018
Tyagi S, Tyagi VK (2014) Novel cationic gemini surfactants and methods for determination of their antimicrobial activity—review. Tenside Surfactants Deterg 51:379–386. https://doi.org/10.3139/113.110319
van der Rest ME, Kamminga AH, Nakano A, Anraku Y, Poolman B, Konings WN (1995) The plasma membrane of Saccharomyces cerevisiae: structure, function, and biogenesis. Microbiol Rev 59:304–322
Vieira DB, Carmona-Ribeiro AM (2006) Cationic lipids and surfactants as antifungal agents: mode of action. J Antimicrob Chemother 58:760–767. https://doi.org/10.1093/jac/dkl312
Wang L-F, He D-Q, Tong Z-H, Li W-W, Yu H-Q (2014) Characterization of dewatering process of activated sludge assisted by cationic surfactants. Biochem Eng J 91:174–178. https://doi.org/10.1016/j.bej.2014.08.008
Weber DJ, Rutala WA (2013) Self-disinfecting surfaces: review of current methodologies and future prospects. Am J Infect Control 41:S31–S35. https://doi.org/10.1016/j.ajic.2012.12.005
Yagura Y, Kirinuki T, Matsunaka S (1984) Mode of action of the fungicide guazatine in Alternaria kikuchiana. J Pestic Sci 9:425–431. https://doi.org/10.1584/jpestics.9.425
Yoder KS, Klos EJ (1976) Tolerance to dodine in Venturia inaequalis. Phytopathology 66:918–923
Acknowledgments
MEF was awarded a CONICET fellowship. SRM, MCNS, and GLG are members of CONICET. LB is member of the CICPBA as a career investigator. Dr. Donald F. Haggerty, a retired academic career investigator and native English speaker, edited the final version of the manuscript.
Funding
This study was funded by MINCyT (PICT 2013-2531 and PICT 2015-1620), CAPES-MINCyT (017/2014), and UNLP (X11-682).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Fait, M.E., Bakas, L., Garrote, G.L. et al. Cationic surfactants as antifungal agents. Appl Microbiol Biotechnol 103, 97–112 (2019). https://doi.org/10.1007/s00253-018-9467-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9467-6