Abstract
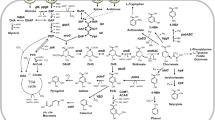
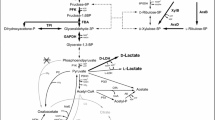
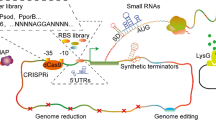
Bio-based production of industrially important chemicals provides an eco-friendly alternative to current petrochemical-based processes. Because of the limited supply of fossil fuel reserves, various technologies utilizing microbial host strains for the sustainable production of platform chemicals from renewable biomass have been developed. Corynebacterium glutamicum is a non-pathogenic industrial microbial species traditionally used for l-glutamate and l-lysine production. It is a promising species for industrial production of bio-based chemicals because of its flexible metabolism that allows the utilization of a broad spectrum of carbon sources and the production of various amino acids. Classical breeding, systems, synthetic biology, and metabolic engineering approaches have been used to improve its applications, ranging from traditional amino-acid production to modern biorefinery systems for production of value-added platform chemicals. This review describes recent advances in the development of genetic engineering tools and techniques for the establishment and optimization of metabolic pathways for bio-based production of major C2–C6 platform chemicals using recombinant C. glutamicum.






Similar content being viewed by others
References
Altaras NE, Cameron DC (2000) Enhanced production of (R)-1,2-propanediol by metabolically engineered Escherichia coli. Biotechnol Prog 16:940–946. https://doi.org/10.1021/bp000076z
Altaras NE, Etzel MR, Cameron DC (2001) Conversion of sugars to 1,2-propanediol by Thermoanaerobacterium thermosaccharolyticum HG-8. Biotechnol Prog 17:52–56. https://doi.org/10.1021/bp000130b
An SJ, Yim SS, Jeong KJ (2013) Development of a secretion system for the production of heterologous proteins in Corynebacterium glutamicum using the Porin B signal peptide. Protein Expr Purif 89(2):251–257. https://doi.org/10.1016/j.pep.2013.04.003
Anthony JR, Anthony LC, Nowroozi F, Kwon G, Newman JD, Keasling JD (2009) Optimization of the mevalonate-based isoprenoid biosynthetic pathway in Escherichia coli for production of the anti-malarial drug precursor amorpha-4,11-diene. Metab Eng 11(1):13–19. https://doi.org/10.1016/j.ymben.2008.07.007
Asakura Y, Kimura E, Usua Y, Kawahara Y, Matsui K, Osumi T, Nakamatsu T (2007) Altered metabolic flux due to deletion of odhA causes L-glutamate overproduction in Corynebacterium glutamicum. Appl Environ Microbiol 73(4):1308–1319. https://doi.org/10.1128/AEM.01867-06
Ashok S, Mohan Raj S, Ko Y, Sankaranarayanan M, Zhou S, Kumar V, Park S (2013a) Effect of puuC overexpression and nitrate addition on glycerol metabolism and anaerobic 3-hydroxypropionic acid production in recombinant Klebsiella pneumoniae ΔglpK ΔdhaT. Metab Eng 15:10–24. https://doi.org/10.1016/j.ymben.2012.09.004
Ashok S, Sankaranarayanan M, Ko Y, Jae KE, Ainala SK, Kumar V, Park S (2013b) Production of 3-hydroxypropionic acid from glycerol by recombinant Klebsiella pneumonia ΔdhaT ΔyqhD which can produce vitamin B12 naturally. Biotechnol Bioeng 110(2):511–524. https://doi.org/10.1002/bit.24726
Baez A, Cho KM, Liao JC (2011) High-flux isobutanol production using engineered Escherichia coli: a bioreactor study with in situ product removal. Appl Microbiol Biotechnol 90:1681–1690. https://doi.org/10.1007/s00253-011-3173-y
Barreiro C, Gonalez-Lavado E, Martin J (2001) Organization and transcriptional analysis of a six-gene cluster around the rplK-rplA operon of Corynebacterium glutamicum encoding the ribosomal proteins L11 and L1. Appl Environ Microbiol 67(5):2183–2190. https://doi.org/10.1128/AEM.67.5.2183-2190.2001
Bartek T, Makus P, Klein B, Lang S, Olidges (2008) Influence of L-isoleucine and pantothenate auxotrophy for L-valine formation in Corynebacterium glutamicum revisited by metabolome analyses. Bioprocess Biosyst Eng 31(3):217–225. https://doi.org/10.1007/s00449-008-0202-z
Becker J, Wittmann C (2012) Bio-based production of chemicals, materials and fuels—Corynebacterium glutamicum as versatile cell factory. Curr Opin Biotechnol 23:631–640. https://doi.org/10.1016/j.copbio.2011.11.012
Becker J, Wittmann C (2015) Advanced biotechnology: metabolically engineered cells for the bio-based production of chemicals and fuels, materials, and health-care products. Angew Chem Int Ed 54(11):3328–3350. https://doi.org/10.1002/anie.201409033
Becker J, Klopprogge C, Herold A, Zelder O, Bolten CJ, Wittmann C (2007) Metabolic flux of L-lysine production in Corynebacterium glutamicum over expression and modification of G6P dehydrogenase. J Biotechnol 132:99–109. https://doi.org/10.1016/j.jbiotec.2007.05.026
Becker J, Klopprogge C, Schroder H, Wittmann C (2009) Metabolic engineering of the tricarboxylic acid cycle for improved lysine production by Corynebacterium glutamicum. Appl Environ Microbiol 75(24):7866–7869. https://doi.org/10.1128/AEM.01942-09
Becker J, Buschke N, Bücker R, Wittmann C (2010) Systems level engineering of Corynebacterium glutamicum—reprogramming translational efficiency for superior production. Eng Life Sci 10(5):430–438. https://doi.org/10.1002/elsc.201000008
Becker J, Gießelmann G, Hoffmann SL, Wittmann C (2018) Corynebacterium glutamicum for sustainable bioproduction: from metabolic physiology to systems metabolic engineering. Adv Biochem Eng Biotechnol 162:217–263. https://doi.org/10.1007/10_2016_21
Binder S, Siedler S, Marienhagen J, Bott M, Eggeling L (2013) Recombineering in Corynebacterium glutamicum combined with optical nanosensors: a general strategy for fast producer strain generation. Nucleic Acid Res 41(12):6360–6369. https://doi.org/10.1093/nar/gkt312
Blombach B, Eikmanns B (2011) Current knowledge on isobutanol production with Escherichia coli, Bacillus subtilis and Corynebacterium glutamicum. Bioeng Bugs 2(6):346–350. https://doi.org/10.4161/bbug.2.6.17845
Blombach B, Riester T, Wieschalka S, Ziert C, Youn JW, Wendisch VF, Eikmanns BJ (2011) Corynebacterium glutamicum tailored for efficient isobutanol production. Appl Environ Microbiol 77:3300–3310. https://doi.org/10.1128/AEM.02972-10
Buschke N, Schäfer R, Becker J, Wittmann C (2013) Metabolic engineering of industrial platform microorganisms for biorefinery applications—optimization of substrate spectrum and process robustness by rational and evolutive strategies. Bioresour Technol 135:544–554. https://doi.org/10.1016/j.biortech.2012.11.047
Cao L, Tang X, Zhang X, Zhang J, Tian X, Wang J, Xiao W (2014) Two-stage transcriptional reprogramming in Saccharomyces cerevisiae for optimizing ethanol production from xylose. Metab Eng 24:150–159. https://doi.org/10.1016/j.ymben.2014.05.001
Cèbe R, Geiser M (2006) Rapid and easy thermodynamic optimization of the 5′-end of mRNA dramatically increases the level of wild type protein expression in Escherichia coli. Protein Expr Purif 45(2):374–380. https://doi.org/10.1016/j.pep.2005.07.007
Celińska E (2010) Debottlenecking the 1,3-propanediol pathway by metabolic engineering. Biotechnol Adv 28:519–530. https://doi.org/10.1016/j.biotechadv.2010.03.003
Chae CG, Kim YJ, Lee SJ, Oh YH, Yang JE, Joo JC, Kang KH, Jang YA, Lee H, Park AR, Song BK, Lee SY, Park SJ (2016) Biosynthesis of poly(2-hydroxybutyrate-co-lactate) and in metabolically engineered Escherichia coli. Biotechnol Bioprocess Eng 21:169–174
Chen Z, Huang J, Wu Y, Liu D (2016a) Metabolic engineering of Corynebacterium glutamicum for the de novo production of ethylene glycol from glucose. Metab Eng 33:12–18. https://doi.org/10.1016/j.ymben.2015.10.013
Chen Z, Huang J, Wu Y, Wu W, Zhang Y, Liu D (2016b) Metabolic engineering of Corynebacterium glutamicum for the production of 3-hydroxypropionic from glucose and xylose. Metab Eng 39:151–158
Cho JS, Choi KR, Prabowo CPS, Shin JH, Yang D, Jang J, Lee SY (2017) CRISPR/Cas9-coupled recombineering for metabolic engineering of Corynebacterium glutamicum. Metab Eng 42:157–167. https://doi.org/10.1016/j.ymben.2017.06.010
Choi JW, Yim SS, Kim MJ, Jeong KJ (2015) Enhanced production of recombinant proteins with Corynebacterium glutamicum by deletion of insertion sequences (IS elements). Microb Cell Factories 14(207):207. https://doi.org/10.1186/s12934-015-0401-7
Choi SY, Park SJ, Kim WJ, Yang JE, Lee H, Shin J, Lee SY (2016) One-step fermentative production of poly(lactate-co-glycolate) from carbohydrates in Escherichia coli. Nat Biotechnol 34:435–440. https://doi.org/10.1038/nbt.3485
Choi JW, Yim SS, Jeong KJ (2017) Development of a high-copy-number plasmid via adaptive laboratory evolution of Corynebacterium glutamicum. Appl Microbiol Biotechnol 102:873–883. https://doi.org/10.1007/s00253-017-8653-2
Chu HS, Kim YS, Lee CM, Lee JH, Jung WS, Ahn JH, Song SH, Choi IS, Cho KM (2015) Metabolic engineering of 3-hydroxypropionic acid biosynthesis in Escherichia coli. Biotechnol Bioeng 112:356–364
Cleto S, Jensen JV, Wendisch VF, Lu TK (2016) Corynebacterium glutamicum metabolic engineering with CRISPR interference (CRISPRi). ACS Synth Biol 5:375–385. https://doi.org/10.1021/acssynbio.5b00216
David Y, Baylon MG, Sudheer PDVN, Baritugo KA, Chae CG, Kim YJ, Kim TW, Kim MS, Na JG, Park SJ (2017a) Screening of microorganisms able to degrade low-rank coal in aerobic condition as potential coal biosolubilization mediators from coal to biochemical. Biotechnol Bioprocess Eng 22:178–185. https://doi.org/10.1007/s12257-016-0263-9
David Y, Joo JC, Yang JE, Oh YH, Lee SY, Park SJ (2017b) Biosynthesis of 2-hydroxyacid-containing polyhydroxyalkanoates by employing butyryl-CoA transferases in metabolically engineered Escherichia coli. Biotechnol J 12:1700116. https://doi.org/10.1002/biot.201700116
Dickschat JS, Wickel S, Bolten CJ, Nawrath T, Schulz S, Wittmann C (2010) Pyrazine biosynthesis in Corynebacterium glutamicum. Eur J Org Chem 2010(14):2687–2695. https://doi.org/10.1002/ejoc.201000155
Dusch N, Pühler A, Kalinowski J (1999) Expression of the Corynebacterium glutamicum panD gene encoding L-aspartate-alpha-decarboxylase leads to pantothenate overproduction in Escherichia coli. Appl Environ Microbiol 65(4):1530–1539
Eggeling L, Bott M (2005) Handbook of Corynebacterium glutamicum. Taylor & Francis, Boca Raton
Ehira S, Teramoto H, Inui M, Yukawa H (2009) Regulation of Corynebacterium glutamicum heat shock response by the extracytoplasmic-function sigma factor SigH and transcriptional regulators HspR and HrcA. J Bacteriol 191:2964–2972. https://doi.org/10.1128/JB.00112-09
Eikmanns BJ, Kleinertz E, Liebl W, Sahm H (1991) A family of Corynebacterium glutamicum/Escherichia coli shuttle vectors for cloning, controlled gene expression and promoter probing. Gene 102:93–98. https://doi.org/10.1016/0378-1119(91)90545-M
Erickson B, Nelson JE, Winters P (2012) Perspective on opportunities in industrial biotechnology in renewable chemicals. Biotechnol J 7:176–185. https://doi.org/10.1002/biot.201100069
Fu J, Wang Z, Chen T, Liu W, Shi T, Wang G, Tang Y, Zhao X (2014) NADH plays the vital role for chiral pure D-(−)-2,3-butanediol production in Bacillus subtilis under limited oxygen conditions. Biotechnol Bioeng 111:2126–2131. https://doi.org/10.1002/bit.25265
Gaspar P, Neves AR, Gasson MJ, Shearman CA (2011) Santos H (2011) High yields of 2,3-butanediol and mannitol in Lactococcus lactis through engineering of NAD+ cofactor recycling. Appl Environ Microbiol 77:6826–6835. https://doi.org/10.1128/AEM.05544-11
Gerstmeir R, Wendisch VF, Schnicke S, Ruan H, Farwick M, Reinscheid D, Eikmanns BJ (2003) Acetate metabolism and its regulation in Corynebacterium glutamicum. J Biotechnol 104:99–122. https://doi.org/10.1016/S0168-1656(03)00167-6
Gonzalez R, Murarka A, Dharmadi Y, Yazdani SS (2008) A new model for the anaerobic fermentation of glycerol in enteric bacteria: trunk and auxiliary pathways in Escherichia coli. Metab Eng 10:234–245. https://doi.org/10.1016/j.ymben.2008.05.001
Gopinath V, Meiswinkel TM, Wendisch VF, Nampoothri KM (2011) Amino acid production from rice straw and wheat bran hydrolysates by recombinant pentose-utilizing Corynebacterium glutamicum. Appl Microbiol Biotechnol 92(5):985–996. https://doi.org/10.1007/s00253-011-3478-x
Gopinath V, Murali A, Dhar KS, Nampoothiri KM (2012) Corynebacterium glutamicum as a potent biocatalyst for the bioconversion of pentose sugars to value-added products. Appl Microbiol Biotechnol 93(1):95–106. https://doi.org/10.1007/s00253-011-3686-4
Guillouet S, Rodal AA, An G, Lessard PA, Sinskey AJ (1999) Expression of the Escherichia coli catabolic threonine dehydratase in Corynebacterium glutamicum and its effect on isoleucine production. Appl Environ Microbiol 65:3100–3107
Harth G, Maslesa-Galić S, Horwitz MA (2004) A two-plasmid system for stable, selective-pressure-independent expression of multiple extracellular proteins in mycobacteria. Microbiology 150(7):2143–2151. https://doi.org/10.1099/mic.0.27113-0
He YC, Xu JH, Su JH, Zhou L (2010) Bioproduction of glycolic acid from glycolonitrile with a new bacterial isolate of Alcaligenes sp. ECU0401. Appl Biochem Biotechnol 160:1428–1440. https://doi.org/10.1007/s12010-009-8607-y
Heider SAE, Wendisch VF (2015) Engineering microbial cell factories: metabolic engineering of Corynebacterium glutamicum with a focus on non-natural products. Biotechnol J 10(8):1170–1184. https://doi.org/10.1002/biot.201400590
Hermann T (2003) Industrial production of amino acids by coryneform bacteria. J Biotechnol 104:155–172. https://doi.org/10.1016/S0168-1656(03)00149-4
Hermann T, Pfefferle W, Baumann C, Busker E, Schaffer S, Bott M, Sahm H, Dusch N, Kalinowski J, Puhler A, Bendt AK, Kramer R, Burkovski A (2001) Proteome analysis of Corynebacterium glutamicum. Electrophoresis 22(9):1712–1723. https://doi.org/10.1002/1522-2683(200105)22:9%3C1712::AID-ELPS1712%3E3.0.CO;2-G
Huang Y, Li Z, Ye Q (2016) Transcriptional regulation of genes involved in 3-hydroxypropionic acid production in response to aeration of recombinant Klebsiella pneumoniae. Appl Biochem Biotechnol 178(6):1129–1140. https://doi.org/10.1007/s12010-015-1933-3
Huang J, Wu Y, Wu W, Zhang Y, Liu CZ (2017) Cofactor recycling for co-production of 1,3-propanediol and glutamate by metabolically engineered Corynebacterium glutamicum. Sci Rep 7:42246. https://doi.org/10.1038/srep42246
Hüser AT, Chassagnole C, Lindley ND, Merkamm M, Guyonvarch A, Elisáková V, Pátek M, Kalinowski J, Brune I, Pühler A, Tauch A (2005) Rational design of a Corynebacterium glutamicum pantothenate production strain and its characterization by metabolic flux analysis and genome-wide transcriptional profiling. Appl Environ Microbiol 71(6):3255–3268. https://doi.org/10.1128/AEM.71.6.3255-3268.2005
Ikeda M, Nakagawa S (2003) The Corynebacterium glutamicum genome: features and impacts on biotechnological processes. Appl Microbiol Biotechnol 62:99–109. https://doi.org/10.1007/s00253-003-1328-1
Ikeda M, Okamoto K, Katsumata R (1999) Cloning of the transketolase gene and the effect of its dosage on aromatic amino acid production in Corynebacterium glutamicum. Appl Microbiol Biotechnol 51:201–206. https://doi.org/10.1007/s002530051382
Ikeda M, Mizuno Y, Awane S, Hayashi M, Mitsuhashi S, Takeno S (2011) Identification and application of a different glucose uptake system that functions as an alternative to the phosphotransferase system in Corynebacterium glutamicum. Appl Microbiol Biotechnol 90:1443–1451. https://doi.org/10.1007/s00253-011-3210-x
Inui M, Kawaguchi H, Murakami S, Vertes AA, Yukawa H (2005) Metabolic Engineering of Corynebacterium glutamicum for Fuel Ethanol Production under Oxygen-Deprivation Conditions. J Mol Microbiol Biotechnol 8 (4):243-254. https://doi.org/10.1159/000086705
Inui M, Suda M, Okino S, Nonaka H, Puskas LG, Vertes AA, Yukawa H (2007) Transcriptional profiling of Corynebacterium glutamicum metabolism during organic acid production under oxygen deprivation conditions. Microbiology 153:2491–2504. https://doi.org/10.1099/mic.0.2006/005587-0
Isern NG, Xue J, Rao JV, Cort JR, Ahring BK (2013) Novel monosaccharide fermentation productsin Caldicellulosiruptor saccharolyticus identified using NMR spectroscopy. Biotechnol Biofuels 6:47. https://doi.org/10.1186/1754-6834-6-47
Jakoby M, Ngouoto-Nkili CE, Burkovski A (1999) Construction and application of new Corynebacterium glutamicum vectors. Biotechnol Tech 13:437–441. https://doi.org/10.1023/A:1008968419217
Jang YS, Kim B, Shin JH, Choi YJ, Choi S, Song CW, Lee J, Par HG, Lee SY (2012) Bio-based production of C2-C6 platform chemicals. Biotechnol Bioeng 109(10):2437–2459. https://doi.org/10.1002/bit.24599
Jiang Y, Chen B, Duan C, Sun B, Yang J, Yang S (2015) Multigene editing in the Escherichia coli genome via the CRISPR- Cas9 system. Appl Environ Microbiol 81:2506–2514. https://doi.org/10.1128/AEM.04023-14
Jo S, Yoon J, Lee SM, Um Y, Han SO, Woo HM (2017) Modular pathway engineering of Corynebacterium glutamicum to improve xylose utilization and succinate production. J Biotechnol 258:69–78. https://doi.org/10.1016/j.jbiotec.2017.01.015
Jojima T, Igari T, Gunji W, Suda M, Inui M, Yukawa H (2012) Identification of a HAD superfamily phosphatase, HdpA, involved in 1,3-dihydroxyacetone production during sugar catabolism in Corynebacterium glutamicum. FEBS Lett 586(23):4228–4232. https://doi.org/10.1016/j.febslet.2012.10.028
Jojima T, Noburyu R, Sasaki M, Tajima T, Suda M, Yukawa H, Inui M (2015) Metabolic engineering for improved production of ethanol by Corynebacterium glutamicum. Appl Microbiol Biotechnol 99:1165–1172. https://doi.org/10.1007/s00253-014-6223-4
Joo JC, Khusnutdinova AN, Flick R, Kim T, Bornscheuer UT, Yakunin AF, Mahadevan R (2017a) Alkene hydrogenation activity of enoate reductases for an environmentally benign biosynthesis of adipic acid. Chem Sci 8:1406–1413. https://doi.org/10.1039/C6SC02842J
Joo JC, Oh YH, Yu JH, Hyun SM, Khang TU, Kang KH, Park SJ (2017b) Production of 5-aminovaleric acid in recombinant Corynebacterium glutamicum strains from a Miscanthus hydrolysate solution prepared by a newly developed Miscanthus hydrolysis process. Bioresour Technol 245:1692–1700. https://doi.org/10.1016/j.biortech.2017.05.131
Jung JY, Choi ES, Oh MK (2008) Enhanced production of 1,2-propanediol by tpi1 deletion in Saccharomyces cerevisiae. J Microbiol Biotechnol 18:1797–1802. https://doi.org/10.4014/jmb.0800.010
Jorge JMP, Pérez-García F, Wendisch VF (2017) A new metabolic route for the fermentative production of 5-aminovalerate from glucose and alternative carbon sources. Bioresour Technol 245:1701–1709. https://doi.org/10.1016/J.BIORTECH.2017.04.108
Kabisch J, Pratzka I, Meyer H, Albrecht D, Lalk M, Ehrenreich A, Schweder T (2013) Metabolic engineering of Bacillus subtilis for growth on overflow metabolites. Microb Cell Factories 12:72. https://doi.org/10.1186/1475-2859-12-72
Kalinowski J, Bathe B, Bartels D, Bischoff N, Bott M, Burkovski A, Tauch A (2003) The complete Corynebacterium glutamicum ATCC 13032 genome sequence and its impact on the production of L-aspartate-derived amino acids and vitamins. J Biotechnol 104:5–25
Kang MK, Lee J, Um Y, Lee T, Bott M, Park SJ, Woo HM (2014) Synthetic biology platform of CoryneBrick vectors for gene expression in Corynebacterium glutamicum and its application to xylose utilization. Appl Microbiol Biotechnol 98:5991–6002. https://doi.org/10.1007/s00253-014-5714-7
Kaur G, Srivastava AK, Chand S (2012) Advances in biotechnological production of 1,3-propanediol. Biochem Eng J 64:106–118. https://doi.org/10.1016/j.bej.2012.03.002
Kawaguchi H, Vertes AA, Okino S, Inui M, Yukawa H (2006) Engineering of a xylose metabolic pathway in Corynebacterium glutamicum. Appl Environ Microbiol 72:3418–3428. https://doi.org/10.1128/AEM.72.5.3418-3428.2006
Kim J, Hirasawa T, Sato Y, Nagahisa K, Furusawa C, Shimizu H (2009) Effect of odhA overexpression and odhA antisense RNA expression on Tween-40-triggered glutamate production by Corynebacterium glutamicum. Appl Microbiol Biotechnol 81(6):1097–1106. https://doi.org/10.1007/s00253-008-1743-4
Kim BR, Lee SJ, Jeong DU, Yang JM, Oh MK, Lee JW (2014) Redistribution of carbon flux toward 2,3-butanediol production in Klebsiella pneumoniae by metabolic engineering. PLoS One 9(10):e105322. https://doi.org/10.1371/journal.pone.0105322
Kind S, Jeong WK, Schröder H, Wittmann C (2010) Systems-wide metabolic pathway engineering in Corynebacterium glutamicum for bio-based production of diaminopentane. Metab Eng 12(4):341–351. https://doi.org/10.1016/j.ymben.2010.03.005
Kind S, Neubauer S, Becker J, Yamamoto M, Volkert M, Von Abendroth G, Zelder O, Wittmann C (2014) From zero to hero—production of bio-based nylon from renewable resources using engineered Corynebacterium glutamicum. Metab Eng 25:113–123. https://doi.org/10.1016/j.ymben.2014.05.007
Kirchner O, Tauch A (2003) Tools for genetic engineering in the amino acid-producing bacterium Corynebacterium glutamicum. J Biotechnol 104:287–299. https://doi.org/10.1016/S0168-1656(03)00148-2
Koivistoinen OM, Kuivanen J, Barth D, Turkia H, Pitkanen JP, Penttila M, Richard P (2013) Glycolic acid production in the engineered yeasts Saccharomyces cerevisiae and Kluyveromyces lactis. Microb Cell Factories 12:82. https://doi.org/10.1186/1475-2859-12-82
Kortmann M, Kuhl V, Klaffl S, Bott M (2015) A chromosomally encoded T7 RNA polymerase-dependent gene expression system for Corynebacterium glutamicum: construction and comparative evaluation at the single-cell level. Microb Biotechnol 8(2):253–365. https://doi.org/10.1111/1751-7915.12236
Kotrba P, Inui M, Yukawa H (2001) The ptsI gene encoding enzyme I of the phosphotransferase system of Corynebacterium glutamicum. Biochem Biophys Res Commun 289:1307–1313. https://doi.org/10.1006/bbrc.2001.6116
Krause FS, Blombach B, Eikmanns BJ (2010) Metabolic engineering of Corynebacterium glutamicum for 2-Ketoisovalerate production. Appl Environ Microbiol 76:8053–8061. https://doi.org/10.1128/AEM.01710-10
Lange J, Muller F, Takors R, Bombach B (2018) Harnessing novel chromosomal integration loci to utilize an organosolv-derived hemicellulose fraction for isobutanol production with engineered Corynebacterium glutamicum. Microb Biotechnol 11(1):257–263. https://doi.org/10.1111/1751-7915.12879
Lee J (2014) Development and characterization of expression vectors for Corynebacterium glutamicum. J Microbiol Biotechnol 24:70–79. https://doi.org/10.4014/jmb.1310.10032
Lee SY, Kim HU (2015) Systems strategies for developing industrial microbial strains. Nat Biotechnol 33:1061–1072. https://doi.org/10.1038/nbt.3365
Lee JW, Kim TY, Jang YS, Choi S, Lee SY (2011) Systems metabolic engineering for chemicals and materials. Trends Biotechnol 29(8):370–378. https://doi.org/10.1016/j.tibtech.2011.04.001
Lee J, Sim SJ, Bott M, Um Y, Oh M, Woo HM (2014a) Succinate production from CO2-grown microalgal biomass as carbon source using engineered Corynebacterium glutamicum through consolidated bioprocessing. Sci Rep 4:5819. https://doi.org/10.1038/srep05819
Lee SM, Jellison T, Alper HS (2014b) Systematic and evolutionary engineering of a xylose isomerase-based pathway in Saccharomyces cerevisiae for efficient conversion yields. Biotechnol Biofuels 7(1):122. https://doi.org/10.1186/s13068-014-0122-x
Leßmeier L, Wendisch VF (2015) Identification of two mutations increasing the methanol tolerance of Corynebacterium glutamicum. BMC Microbiol 15(216):216. https://doi.org/10.1186/s12866-015-0558-6
Letek M, Valbuena N, Ramos A, Ordonez E, Gil JA, Mateos LM (2006) Characterization and use of catabolite-repressed promoters from gluconate genes in Corynebacterium glutamicum. J Bacteriol 188:409–423. https://doi.org/10.1128/JB.188.2.409-423.2006
Li L, Wada M, Yokota A (2007) Cytoplasmic proteome reference map for a glutamic acid-producing Corynebacterium glutamicum ATCC 14067. Proteomics 7(23):4317–4322. https://doi.org/10.1002/pmic.200700269
Lim HG, Noh MH, Jeong JH, Park S, Jung GY (2016) Optimum rebalancing of the 3-hydroxypropionic acid production pathway from glycerol in Esherichia coli. ACS Synth Biol 5(11):1247–1255. https://doi.org/10.1021/acssynbio.5b00303
Litsanov B, Brocker M, Bott M (2013) Glycerol as a substrate for aerobic succinate production in minimal medium with Corynebacterium glutamicum. Microb Biotechnol 6(2):189–195. https://doi.org/10.1111/j.1751-7915.2012.00347.x
Liu Q, Ouyang SP, Kim J, Chen GQ (2007) The impact of PHB accumulation on L-glutamate production by recombinant Corynebacterium glutamicum. J Biotechnol 132(3):273–279. https://doi.org/10.1016/j.jbiotec.2007.03.014
Liu Z, Tyo KEJ, Martínez JL, Petranovic D, Nielsen J (2012) Different expression systems for production of recombinant proteins in Saccharomyces cerevisiae. Biotechnol Bioeng 109:1259–1268. https://doi.org/10.1002/bit.24409
Liu H, Ramos KRM, Valdehuesa KNG, Nisola GM, Lee W, Chung W (2013) Biosynthesis of ethylene glycol in Escherichia coli. Appl Microbiol Biotechnol 97:3409–3417. https://doi.org/10.1007/s00253-012-4618-7
Liu X, Yang Y, Zhang W, Sun Y, Peng F, Jeffrey L, Harvey L, McNeil B, Bai Z (2015) Expression of recombinant protein using Corynebacterium glutamicum: progress, challenges and applications. Crit Rev Biotechnol 36:1–13. https://doi.org/10.3109/07388551.2015.1004519
Liu M, Han X, Xian M, Ding Y, Liu H, Zhao G (2016) Development of a 3-hydroxypropionate resistant Escherichia coli strain. Bioengineered 7(1):21–27. https://doi.org/10.1080/21655979.2015.1122143
Liu X, Zhang W, Zhao Z, Dai X, Yang Y, Bai Z (2017) Protein secretion in Corynebacterium glutamicum. Crit Rev Biotechnol 37:541–551. https://doi.org/10.1080/07388551.2016.1206059
Maervoet VET, Maeseneire SL, De Avci FG, Beauprez J, Soetaert WK (2016) High yield 1,3-propanediol production by rational engineering of the 3 -hydroxypropionaldehyde bottleneck in Citrobacter werkmanii. Microb Cell Factories 15(23):23. https://doi.org/10.1186/s12934-016-0421-y
Martin CH, Dhamankar H, Tseng HC, Sheppard MJ, Reisch CR, Prather KL (2013) A platform pathway for production of 3-hydroxyacids provides a biosynthetic route to 3-hydroxy-gamma-butyrolactone. Nat Commun 4:1414. https://doi.org/10.1038/ncomms2418
Mateos L, Pisabarro A, Patek M, Malumbres M, Guerrero C, Eikmanns B, Sahm H, Martin J (1994) Transcriptional analysis and regulatory signals of the hom-thrB cluster of Brevibacterium lactofermentum. J Bacteriol 176(23):7362–7371. https://doi.org/10.1128/jb.176.23.7362-7371.1994
Meiswinkel TM, Rittmann D, Lindner SN, Wendisch VF (2013) Crude glycerol-based production of amino acids and putrescine by Corynebacterium glutamicum. Bioresour Technol 145:254–258. https://doi.org/10.1016/j.biortech.2013.02.053
Mentz A, Neshat A, Pfeifer-Sancar K, Pühler A, Rückert C, Kalinowski J (2013) Comprehensive discovery and characterization of small RNAs in Corynebacterium glutamicum ATCC 13032. BMC Genomics 14(1):714. https://doi.org/10.1186/1471-2164-14-714
Merkamm M, Guyonvarch A (2001) Cloning of the sodA gene from Corynebacterium melassecola and role of superoxide dismutase in cellular viability. J Bacteriol 183(4):1284–1295. https://doi.org/10.1128/JB.2001.183.4.1284-1295.2001
Misra K, Banerjee AB, Ray S, Ray M (1996) Reduction of methylglyoxal in Escherichia coli K12 by an aldehyde reductase and alcohol dehydrogenase. Mol Cell Biochem 156:117–124. https://doi.org/10.1007/BF00426333
Nakamura Y, Nishio Y, Ikeo K, Gojobori T (2003) The genome stability in Corynebacterium species due to lack of the recombinational repair system. Gene 317:149–155. https://doi.org/10.1016/S0378-1119(03)00653-X
Nešvera J, Pátek M (2011) Tools for genetic manipulations in Corynebacterium glutamicum and their applications. Appl Microbiol Biotechnol 90(5):1641–1654. https://doi.org/10.1007/s00253-011-3272-9
Niimi S, Suzuki N, Inui M, Yukawa H (2011) Metabolic engineering of 1,2-propanediol pathways in Corynebacterium glutamicum. Appl Microbiol Biotechnol 90(5):1721–1729. https://doi.org/10.1007/s00253-011-3190-x
Oh YH, Eom IY, Joo JC, Yu JH, Song BK, Lee SH, Hong SH, Park SJ (2015a) Recent advances in development of biomass pretreatment technologies used in biorefinery for the production of bio-based fuels, chemicals and polymers. Korean J Chem Eng 32:1945–1959. https://doi.org/10.1007/s11814-015-0191-y
Oh YH, Choi JW, Kim EY, Song BK, Jeong KJ, Park K, Kim IK, Woo HM, Lee SH, Park SJ (2015b) Construction of synthetic promoter-based expression cassettes for the production of cadaverine in recombinant Corynebacterium glutamicum. Appl Biochem Biotechnol 176:2065–2075. https://doi.org/10.1007/s12010-015-1701-4
Ohse M, Takahashi K, Kadowaki Y, Kusaoke H (1995) Effects of plasmid DNA sizes and several other factors on transformation of Bacillus subtilis ISW1214 with plasmid DNA by electroporation. Biosci Biotechnol Biochem 59(8):1433–1437. https://doi.org/10.1271/bbb.59.1433
Oreb M, Dietz H, Farwick A, Boles E (2012) Novel strategies to improve co-fermentation of pentoses with D-glucose by recombinant yeast strains in lignocellulosic hydrolysates. Bioengineered 3(6):347–351. https://doi.org/10.4161/bioe.21444
Panova A, Mersingera LI, Liu Q, Foo T, Roe DC, Spillan WL, Sigmund AE, Ben-Bassat A, Wagner LW, DP O‘K, Wu S, Petrillo KL, Payne MS, Breske ST, Gallagher FG, Di Cosimo R (2007) Chemoenzymatic synthesis of glycolic acid. Adv Synth Catal 349:1462–1474. https://doi.org/10.1002/adsc.200700061
Papagianni M (2012) Recent advances in engineering the central carbon metabolism of industrially important bacteria. Microb Cell Factories 11:50. https://doi.org/10.1186/1475-2859-11-50
Park SD, Lee SN, Park IH, Choi JS, Jeong WK, Kim Y, Lee HS (2004) Isolation and characterization of transcriptional elements from Corynebacterium glutamicum. J Microbiol Biotechnol 14:789–795
Park JU, Jo JH, Kim YJ, Chung SS, Lee JH, Lee HH (2008) Construction of heat-inducible expression vector of Corynebacterium glutamicum and C. ammoniagenes: fusion of lambda operator with promoters isolated from C. ammoniagenes. J Microbiol Biotechnol 18:639–647
Park SH, Kim HU, Kim TY, Park JS, Kim SS, Lee SY (2014) Metabolic engineering of Corynebacterium glutamicum for L-arginine production. Nat Commun 5:4618. https://doi.org/10.1038/ncomms5618
Pátek M, Nešvera J (2013) Promoters and plasmid vectors of Corynebacterium glutamicum. In: Tatsumi N, Inui M (eds) Corynebacterium glutamicum: biology and biotechnology. Springer, Berlin, pp 51–88. https://doi.org/10.1007/978-3-642-29857-8_2
Peters-Wendisch PG, Schiel B, Wendisch VF, Katsoulidis E, Möckel B, Sahm H, Eikmanns BJ (2001) Pyruvate carboxylase is a major bottleneck for glutamate and lysine production by Corynebacterium glutamicum. J Mol Microbiol Biotechnol 3:295–300
Rados D, Carvahalo AL, Wieschalka S, Neves AR, Blombach B, Eikmanns BJ, Santos H (2015) Engineering Corynebacterium glutamicum for the production of 2,3-butanediol. Microb Cell Factories 14:171. https://doi.org/10.1186/s12934-015-0362-x
Ravasi P, Peiru S, Gramajo H, Menzella HG (2012) Design and testing of a synthetic biology framework for genetic engineering of Corynebacterium glutamicum. Microb Cell Factories 11:1–11. https://doi.org/10.1186/1475-2859-11-147
Ren C, Wen Z, Xu Y, Jiang W, Gu Y (2016) Clostridia: a flexible microbial platform for the production of alcohols. Curr Opin Chem Biol 35:65–72. https://doi.org/10.1016/j.cbpa.2016.08.024
Rittmann D, Lindner SN, Wendisch VF (2008) Engineering of a glycerol utilization pathway for amino acid production by Corynebacterium glutamicum. Appl Environ Microbiol 74(20):6216–6222. https://doi.org/10.1128/AEM.00963-08
Sakai S, Tsuchida Y, Nakamoto H, Okino S, Ichihashi O, Kawaguchi H, Yukawa H (2007) Effect of lignocellulose-derived inhibitors on growth of and ethanol production by growth-arrested Corynebacterium glutamicum R. Appl Environ Microbiol 73:2349–2353. https://doi.org/10.1128/AEM.02880-06
Sanchez-Rivera F, Cameron DC, Cooney CL (1987) Influence of environmental factors in the production of R-(−)-1,2-propanediol by Clostridium thermosacchrolyticum. Biotechnol Lett 9:449–454. https://doi.org/10.1007/BF01027450
Saxena RK, Anand P, Saran S, Isar J (2009) Microbial production of 1,3-propanediol: recent developments and emerging opportunities. Biotechnol Adv 27:895–913. https://doi.org/10.1016/j.biotechadv.2009.07.003
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145(1):69–73. https://doi.org/10.1016/0378-1119(94)90324-7
Schneider J, Niermann K, Wendisch VF (2011) Production of the amino acids L-glutamate, L-lysine, L-ornithine and L-arginine from arabinose by recombinant Corynebacterium glutamicum. J Biotechnol 154:191–198. https://doi.org/10.1016/j.jbiotec.2010.07.009
Schneider J, Eberhardt D, Wendisch VF (2012) Improving putrescine production by Corynebacterium glutamicum by fine-tuning ornithine transcarbamoylase activity using a plasmid addiction system. Appl Microbiol Biotechnol 95(1):169–178. https://doi.org/10.1007/s00253-012-3956-9
Schwarzer A, Pühler A (1991) Manipulation of Corynebacterium glutamicum by gene disruption and replacement. Nat Biotechnol 9(1):84–87. https://doi.org/10.1038/nbt0191-84
Shin JH, Park SH, Oh YH, Choi JW, Lee MH, Cho JS, Jeong KJ, Joo JC, Yu J, Park SJ, Lee SY (2016) Metabolic engineering of Corynebacterium glutamicum for enhanced production of 5-aminovaleric acid. Microb Cell Factories 15:174. https://doi.org/10.1186/s12934-016-0566-8
Siebert D, Wendisch VF (2015) Metabolic pathway engineering for production of 1,2-propanediol and 1-propanol by Corynebacterium glutamicum. Biotechnol Biofuels 8:91. https://doi.org/10.1186/s13068-015-0269-0
Smith KM, Cho KM, Liao JC (2010) Engineering Corynebacterium glutamicum for isobutanol production. Appl Microbiol Biotechnol 87:1045–1055. https://doi.org/10.1007/s00253-010-2522-6
Srivastava P, Deb JK (2002) Construction of fusion vectors of Corynebacteria: expression of glutathione-S-transferase fusion protein in Corynebacterium acetoacidophilum ATCC 21476. FEMS Microbiol Lett 212:209–216. https://doi.org/10.1111/j.1574-6968.2002.tb11268.x
Stansen C, Uy D, Delaunay S, Eggeling L, Goergen JL, Wendisch VF (2005) Characterization of a Corynebacterium glutamicum lactate utilization operon induced during temperature-triggered glutamate production. Appl Environ Microbiol 71:5920–5928. https://doi.org/10.1128/AEM.71.10.5920-5928.2005
Stolz M, Peters-Wendisch P, Etterich H, Gerharz T, Faurie R, Sahm H, Fersterra H, Eggeling L (2007) Reduced folate supply as a key to enhanced L-serine production by Corynebacterium glutamicum. Appl Environ Microbiol 73:750–755. https://doi.org/10.1128/AEM.02208-06
Sudheer PDVN, David Y, Chae CG, Kim YJ, Baylon MG, Baritugo KA, Kim TW, Kim MS, Na JG, Park SJ (2016) Advances in biological treatment of coal for synthetic natural gas and chemicals. Korean J Chem Eng 33:2788–2801. https://doi.org/10.1007/s11814-016-0225-0
Sun Y, Guo W, Wang F, Zhan C, Yang Y, Liu X, Bai Z (2017) Transcriptome analysis of Corynebacterium glutamicum in the process of recombinant protein expression in bioreactors. PLoS One 12(4):e0174824. https://doi.org/10.1371/journal.pone.0174824
Suzuki N, Inui M (2013) Genome engineering of Corynebacterium glutamicum. In: Tatsumi N, Inui M (eds) Corynebacterium glutamicum: biology and biotechnology. Springer, Berlin, pp 89–105. https://doi.org/10.1007/978-3-642-29857-8_3
Suzuki N, Nonaka H, Tsuge Y, Inui M, Yukawa H (2005) New multiple-deletion method for the Corynebacterium glutamicum genome, using a mutant lox sequence. Appl Environ Microbiol 71:8472–8480. https://doi.org/10.1128/AEM.71.12.8472-8480.2005
Tateno T, Okada Y, Tsuchidate T, Tanaka T, Fukuda H, Kondo A (2009) Direct production of cadaverine from soluble starch using Corynebacterium glutamicum coexpressing alpha-amylase and lysine decarboxylase. Appl Microbiol Biotechnol 82:115–121. https://doi.org/10.1007/s00253-008-1751-4
Teramoto H, Watanabe K, Suzuki N, Inui M, Yukawa H (2011) High yield secretion of heterologous proteins in Corynebacterium glutamicum using its own tat-type signal sequence. Appl Microbiol Biotechnol 91(3):677–687. https://doi.org/10.1007/s00253-011-3281-8
Tran-Din K, Gottschalk G (1985) Formation of D(−)-1,2-propanedioland D(−)-lactate from glucose by Clostridium sphenoides underphosphate limitation. Arch Microbiol 142:87–92. https://doi.org/10.1007/s00253-011-3281-8
Tsuchiya M, Morinaga Y (1988) Genetic control systems of Escherichia coli can confer inducible expression of cloned genes in coryneform bacteria. Nat Biotechnol 6:428–430. https://doi.org/10.1038/nbt0488-428
Tsuge Y, Suzuki N, Inui M, Yukawa H (2007) Random segment deletion based on IS31831 and Cre/loxP excision system in Corynebacterium glutamicum. Appl Microbiol Biotechnol 74:1333–1341. https://doi.org/10.1007/s00253-006-0788-5
Venkova-Canova T, Pátek M, Nesvera J (2004) Characterization of the cryptic plasmid pCC1 from Corynebacterium callunae and its use for vector construction. Plasmid 51(1):54–60. https://doi.org/10.1016/j.plasmid.2003.09.002
Vertès AA, Inui M, Kobayashi M, Kurusu Y, Yukawa H (1993) Presence of mrr- and mcr-like restriction systems in coryneform bacteria. Res Microbiol 144:181–185. https://doi.org/10.1016/0923-2508(93)90043-2
Wei G, Yang X, Gan T, Zhou W, Lin J, Wei D (2009) High cell density fermentation of Gluconobacter oxydans DSM 2003 for glycolic acid production. J Ind Microbiol Biotechnol 36:1029–1034. https://doi.org/10.1007/s10295-009-0584-1
Wendisch VF (2003) Genome-wide expression analysis in Corynebacterium glutamicum using DNA microarrays. J Biotechnol 104:273–285. https://doi.org/10.1016/S0168-1656(03)00147-0
Wendisch VF, Bott M, Eikmanns BJ (2006a) Metabolic engineering of Escherichia coli and Corynebacterium glutamicum for biotechnological production of organic acids and amino acids. Curr Opin Microbiol 9:268–274. https://doi.org/10.1016/j.mib.2006.03.001
Wendisch VF, Bott M, Kalinowski J, Oldiges M, Wiechert W (2006b) Emerging Corynebacterium glutamicum systems biology. J Biotechnol 124:74–92. https://doi.org/10.1016/j.jbiotec.2005.12.002
Wieschalka S, Blombach B, Bott M, Eikmanns BJ (2013) Bio-based production of organic acids with Corynebacterium glutamicum. Microb Biotechnol 6:87–102. https://doi.org/10.1111/1751-7915.12013
Woo HM, Park JB (2014) Recent progress in development of synthetic biology platforms and metabolic engineering of Corynebacterium glutamicum. J Biotechnol 180:43–51. https://doi.org/10.1016/j.jbiotec.2014.03.003
Woo HM, Noack S, Seibold G, Willbold S, Eikmanns B, Bott M (2010) Link between phosphate starvation and glycogen metabolism in Corynebacterium glutamicum, revealed by metabolomics. Appl Environ Microbiol 76:6910–6919. https://doi.org/10.1128/AEM.01375-10
Xu Y, Chu H, Gao C, Tao F, Zhou Z, Li K, Li L, Ma C, Xu P (2014) Systematic metabolic engineering of Escherichia coli for high-yield production of fuel biochemical 2,3-butanediol. Metab Eng 23:22–33. https://doi.org/10.1016/j.ymben.2014.02.004
Yamamoto S, Suda M, Niimi S, Inui M, Yukawa H (2013) Strain optimization for efficient isobutanol production using Corynebacterium glutamicum under oxygen deprivation. Biotechnol Bioeng 110(11):2938–2948. https://doi.org/10.1002/bit.24961
Yang J, Yang S (2017) Comparative analysis of Corynebacterium glutamicum genomes: a new perspective for the industrial production of amino acids. BMC Genomics 18:940. https://doi.org/10.1186/s12864-016-3255-4
Yang J, Kim B, Kim H, Kweon Y, Lee S, Lee J (2015) Industrial production of 2,3-butanediol from the engineered Corynebacterium glutamicum. Appl Biochem Biotechnol 176(8):2303–2313. https://doi.org/10.1007/s12010-015-1719-7
Yang JE, Kim JW, Oh YH, Choi SY, Lee H, Park AR, Shin J, Park SJ, Lee SY (2016) Biosynthesis of poly(2-hydroxyisovalerate-co-lactate) by metabolically engineered Escherichia coli. Biotechnol J 11:1572–1585. https://doi.org/10.1002/biot.201600420
Yang JE, Park SJ, Kim WJ, Kim HJ, Kim BJ, Lee H, Shin J, Lee SY (2018) One-step fermentative production of aromatic polyesters from glucose by metabolically engineered Escherichia coli strains. Nat Commun 9(79):79. https://doi.org/10.1038/s41467-017-02498-w
Yim SS, An SJ, Kang M, Lee J, Jeong KJ (2013) Isolation of fully synthetic promoters for high-level gene expression in Corynebacterium glutamicum. Biotechnol Bioeng 110(11):2959–2969. https://doi.org/10.1002/bit.24954
Yim SS, An SJ, Choi JW, Ryu AJ, Jeong KJ (2014) High-level secretory production of recombinant single-chain variable fragment (scFv) in Corynebacterium glutamicum. Appl Microbiol Biotechnol 98:273–284
Yim SS, Choi JW, Lee SH, Jeong KJ (2016a) Modular optimization of a hemicellulose-utilizing pathway in Corynebacterium glutamicum for consolidated bioprocessing of hemicellulosic biomass. ACS Synth Biol 5:334–343. https://doi.org/10.1021/acssynbio.5b00228
Yim SS, Choi JW, Lee RJ, Lee YJ, Lee SH, Kim SY, Jeong KJ (2016b) Development of a new platform for secretory production of recombinant proteins in Corynebacterium glutamicum. Biotechnol Bioeng 113(1):163–172. https://doi.org/10.1002/bit.25692
Yim SS, Choi JW, Lee SH, Jeon EJ, Chung WJ, Jeong KJ (2017) Engineering of Corynebacterium glutamicum for consolidated conversion of hemicellulosic biomass into xylonic acid. Biotechnol J 12(11):1700040. https://doi.org/10.1002/biot.201700040
Yukawa H, Omumasaba CA, Nonaka H, Kos P, Okai N, Suzuki N, Suda M, Tsuge Y, Watanabe J, Ikeda Y, Vertes AA, Inui M (2007) Comparative analysis of the Corynebacterium glutamicum group and complete genome sequence of strain R. Microbiology 153:1042–1058. https://doi.org/10.1099/mic.0.2006/003657-0
Zahoor A, Lindner SN, Wendisch VF (2012) Metabolic engineering of Corynebacterium glutamicum aimed at alternative carbon sources and new products. Comput Struct Biotechnol J 3:e201210004. https://doi.org/10.5936/csbj.201210004
Zahoor A, Otten A, Wendisch VF (2014) Metabolic engineering of Corynebacterium glutamicum for glycolate production. J Biotechnol 192:366–375. https://doi.org/10.1016/j.jbiotec.2013.12.020
Zhang Q, Xiu Z (2009) Metabolic pathway analysis of glycerol metabolism in Klebsiella pneumoniae incorporating oxygen regulatory system. Biotechnol Prog 25:103–115. https://doi.org/10.1002/btpr.70
Zhang Y, Shang X, Lai S, Zhang G, Liang Y, Wen T (2012) Development and application of an arabinose-inducible expression system by facilitating inducer uptake in Corynebacterium glutamicum. Appl Environ Microbiol 78:5831–5838. https://doi.org/10.1128/AEM.01147-12
Zhang B, Yu M, Zhou Y, Li Y, Ye BC (2017) Systematic pathway engineering of Corynebacterium glutamicum S9114 for L-ornithine production. Microb Cell Factories 16(158):158. https://doi.org/10.1186/s12934-017-0776-8
Zhou LB, Zeng AP (2015) Engineering a lysine-ON riboswitch for metabolic control of lysine production in Corynebacterium glutamicum. ACS Synth Biol 4(12):1335–1340. https://doi.org/10.1021/acssynbio.5b00075
Zhu Q, Zhang X, Luo Y, Guo W, Xu G, Shi J, Xu Z (2014) L-serine over-production with minimization of by-product synthesis by engineered Corynebacterium glutamicum. Appl Microbiol Biotechnol 99:1665–1673. https://doi.org/10.1007/s00253-014-6243-0
Acknowledgements
This work was supported by the Mid-career Researcher Program through the National Research Foundation (NRF) of Korea funded by the Ministry of Science and ICT (MSIT) (NRF-2016R1A2B4008707), a basic research grant from the KRIBB, and the Lignin Biorefinery from MSIT through the NRF of Korea (NRF-2017M1A2A2087634).
Availability of data and material
Please contact corresponding author for any data requests.
Funding
Funding sources are declared in acknowledgement section.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable. This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable. Our manuscript does not contain any individual person’s data in any form.
Rights and permissions
About this article
Cite this article
Baritugo, KA., Kim, H.T., David, Y. et al. Metabolic engineering of Corynebacterium glutamicum for fermentative production of chemicals in biorefinery. Appl Microbiol Biotechnol 102, 3915–3937 (2018). https://doi.org/10.1007/s00253-018-8896-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-8896-6