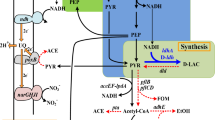
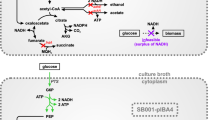
Abstract
Anaerobic fermentation is a favorable process for microbial production of bulk chemicals like ethanol and organic acids. Low productivity is the bottleneck of several anaerobic processes which has significant impact on the technique competitiveness of production strain. Improving growth rate of production strain can speed up the total production cycle and may finally increase productivity of anaerobic processes. In this work, evolutionary engineering of wild-type strain Escherichia coli W3110 was adopted to improve anaerobic growth in mineral medium. Significant increases in exponential growth rate and stationary cell density were achieved in evolved strain WE269, and a 96.5% increase in lactate productivity has also been observed in batch fermentation of this strain with M9 minimal medium. Then, an engineered strain for lactate production (BW100) was constructed by using WE269 as a platform and 98.3 g/L lactate (with an optical purity of D-lactate above 95%) was produced in a 5-L bioreactor after 48 h with a productivity of 2.05 g/(L·h). Finally, preliminary investigation demonstrated that mutation in sucD (sucD M245I) (encoding succinyl-CoA synthetase); ilvG (ilvG Δ1bp) (encoding acetolactate synthase 2 catalytic subunit), and rpoB (rpoB T1037P) (encoding RNA polymerase β subunit) significantly improved anaerobic growth of E. coli. Double-gene mutation in ilvG and sucD resumed most of the growth potential of evolved strain WE269. This work suggested that improving anaerobic growth of production host can increase productivity of organic acids like lactate, and specific mutation-enabled improved growth may also be applied to metabolic engineering for production of other bulk chemicals.








Similar content being viewed by others
References
Assavasirijinda N, Ge D, Yu B, Xue Y, Ma Y (2016) Efficient fermentative production of polymer-grade D-lactate by an engineered alkaliphilic Bacillus sp. strain under non-sterile conditions. Microb Cell Factories 15(1):3. https://doi.org/10.1186/s12934-015-0408-0
Baek SH, Kwon EY, Kim YH, Hahn JS (2016) Metabolic engineering and adaptive evolution for efficient production of D-lactic acid in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 100(6):2737–2748. https://doi.org/10.1007/s00253-015-7174-0
Barrick JE, Yu DS, Yoon SH, Jeong H, Oh TK, Schneider D, Lenski RE, Kim JF (2009) Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 461(7268):1243–1247. https://doi.org/10.1038/nature08480
Barrick JE, Colburn G, Deatherage DE, Traverse CC, Strand MD, Borges JJ, Knoester DB, Reba A, Meyer AG (2014) Identifying structural variation in haploid microbial genomes from short-read resequencing data using breseq. BMC Genomics 15:1039. https://doi.org/10.1186/1471-2164-15-1039
Clark DP (1989) The fermentation pathways of Escherichia coli. FEMS Microbiol Rev 5(3):223–234
Conrad TM, Joyce AR, Applebee MK, Barrett CL, Xie B, Gao Y, Palsson BO (2009) Whole-genome resequencing of Escherichia coli K-12 MG1655 undergoing short-term laboratory evolution in lactate minimal media reveals flexible selection of adaptive mutations. Genome Biol 10(10):R118. https://doi.org/10.1186/gb-2009-10-10-r118
Conrad TM, Frazier M, Joyce AR, Cho BK, Knight EM, Lewis NE, Landick R, Palsson BO (2010) RNA polymerase mutants found through adaptive evolution reprogram Escherichia coli for optimal growth in minimal media. Proc Natl Acad Sci U S A 107(47):20500–20505. https://doi.org/10.1073/pnas.0911253107
Deatherage DE, Barrick JE (2014) Identification of mutations in laboratory-evolved microbes from next-generation sequencing data using breseq. Methods Mol Biol (Clifton, NJ) 1151:165–188. https://doi.org/10.1007/978-1-4939-0554-6_12
Feng X, Ding Y, Xian M, Xu X, Zhang R, Zhao G (2014) Production of optically pure d-lactate from glycerol by engineered Klebsiella pneumoniae strain. Bioresour Technol 172:269–275. https://doi.org/10.1016/j.biortech.2014.09.074
Finn TJ, Shewaramani S, Leahy SC, Janssen PH, Moon CD (2017) Dynamics and genetic diversification of Escherichia coli during experimental adaptation to an anaerobic environment. PeerJ 5:e3244. https://doi.org/10.7717/peerj.3244
Fong SS, Burgard AP, Herring CD, Knight EM, Blattner FR, Maranas CD, Palsson BO (2005) In silico design and adaptive evolution of Escherichia coli for production of lactic acid. Biotechnol Bioeng 91(5):643–648. https://doi.org/10.1002/bit.20542
Forster AH, Gescher J (2014) Metabolic engineering of Escherichia coli for production of mixed-acid fermentation end products. Front Bioeng Biotechnol 2:16. https://doi.org/10.3389/fbioe.2014.00016
Fu X, Wang Y, Wang J, Garza E, Manow R, Zhou S (2017) Semi-industrial scale (30 m(3)) fed-batch fermentation for the production of D-lactate by Escherichia coli strain HBUT-D15. J Ind Microbiol Biotechnol 44(2):221–228. https://doi.org/10.1007/s10295-016-1877-9
Grabar TB, Zhou S, Shanmugam KT, Yomano LP, Ingram LO (2006) Methylglyoxal bypass identified as source of chiral contamination in l(+) and d(−)-lactate fermentations by recombinant Escherichia coli. Biotechnol Lett 28(19):1527–1535. https://doi.org/10.1007/s10529-006-9122-7
Guzman GI, Utrilla J, Nurk S, Brunk E, Monk JM, Ebrahim A, Palsson BO, Feist AM (2015) Model-driven discovery of underground metabolic functions in Escherichia coli. Proc Natl Acad Sci U S A 112(3):929–934. https://doi.org/10.1073/pnas.1414218112
Harden MM, He A, Creamer K, Clark MW, Hamdallah I, Martinez KA 2nd, Kresslein RL, Bush SP, Slonczewski JL (2015) Acid-adapted strains of Escherichia coli K-12 obtained by experimental evolution. Appl Environ Microbiol 81(6):1932–1941. https://doi.org/10.1128/aem.03494-14
He A, Penix SR, Basting PJ, Griffith JM, Creamer KE, Camperchioli D, Clark MW, Gonzales AS, Chavez Erazo JS, George NS, Bhagwat AA, Slonczewski JL (2017) Acid evolution of Escherichia coli K-12 eliminates amino acid decarboxylases and reregulates catabolism. Appl Environ Microbiol 83(12). https://doi.org/10.1128/aem.00442-17
Herring CD, Raghunathan A, Honisch C, Patel T, Applebee MK, Joyce AR, Albert TJ, Blattner FR, van den Boom D, Cantor CR, Palsson BO (2006) Comparative genome sequencing of Escherichia coli allows observation of bacterial evolution on a laboratory timescale. Nat Genet 38(12):1406–1412. https://doi.org/10.1038/ng1906
Horinouchi T, Sakai A, Kotani H, Tanabe K, Furusawa C (2017) Improvement of isopropanol tolerance of Escherichia coli using adaptive laboratory evolution and omics technologies. J Biotechnol 255:47–56. https://doi.org/10.1016/j.jbiotec.2017.06.408
Hua Q, Joyce AR, Fong SS, Palsson BO (2006) Metabolic analysis of adaptive evolution for in silico-designed lactate-producing strains. Biotechnol Bioeng 95(5):992–1002. https://doi.org/10.1002/bit.21073
Ingram LO, Conway T, Clark DP, Sewell GW, Preston JF (1987) Genetic engineering of ethanol production in Escherichia coli. Appl Environ Microbiol 53(10):2420–2425
Kuhlman TE, Cox EC (2010) Site-specific chromosomal integration of large synthetic constructs. Nucleic Acids Res 38(6):e92–e92. https://doi.org/10.1093/nar/gkp1193
LaCroix RA, Sandberg TE, O'Brien EJ, Utrilla J, Ebrahim A, Guzman GI, Szubin R, Palsson BO, Feist AM (2015) Use of adaptive laboratory evolution to discover key mutations enabling rapid growth of Escherichia coli K-12 MG1655 on glucose minimal medium. Appl Environ Microbiol 81(1):17–30. https://doi.org/10.1128/aem.02246-14
Li Y, Lin Z, Huang C, Zhang Y, Wang Z, Tang YJ, Chen T, Zhao X (2015) Metabolic engineering of Escherichia coli using CRISPR-Cas9 meditated genome editing. Metab Eng 31:13–21. https://doi.org/10.1016/j.ymben.2015.06.006
Liang S, Gao D, Liu H, Wang C, Wen J (2018) Metabolomic and proteomic analysis of D-lactate-producing Lactobacillus delbrueckii under various fermentation conditions. J Ind Microbiol Biotechnol https://doi.org/10.1007/s10295-018-2048-y
Liu M, Han X, Xian M, Ding Y, Liu H, Zhao G (2016) Development of a 3-hydroxypropionate resistant Escherichia coli strain. Bioengineered 7(1):21–27. https://doi.org/10.1080/21655979.2015.1122143
Lu H, Zhao X, Wang Y, Ding X, Wang J, Garza E, Manow R, Iverson A, Zhou S (2016) Enhancement of D-lactic acid production from a mixed glucose and xylose substrate by the Escherichia coli strain JH15 devoid of the glucose effect. BMC Biotechnol 16:19. https://doi.org/10.1186/s12896-016-0248-y
Maddamsetti R, Hatcher PJ, Green AG, Williams BL, Marks DS, Lenski RE (2017) Core genes evolve rapidly in the long-term evolution experiment with Escherichia coli. Genome Biol Evol 9:1072–1083. https://doi.org/10.1093/gbe/evx064
Morris JG (1983) Anaerobic fermentations—some new possibilities. Biochem Soc Symp 48:147–172
Niu D, Tian K, Prior BA, Wang M, Wang Z, Lu F, Singh S (2014) Highly efficient L-lactate production using engineered Escherichia coli with dissimilar temperature optima for L-lactate formation and cell growth. Microb Cell Factories 13:78. https://doi.org/10.1186/1475-2859-13-78
Okino S, Suda M, Fujikura K, Inui M, Yukawa H (2008) Production of D-lactic acid by Corynebacterium glutamicum under oxygen deprivation. Appl Microbiol Biotechnol 78(3):449–454. https://doi.org/10.1007/s00253-007-1336-7
Portnoy VA, Bezdan D, Zengler K (2011) Adaptive laboratory evolution—harnessing the power of biology for metabolic engineering. Curr Opin Biotechnol 22(4):590–594. https://doi.org/10.1016/j.copbio.2011.03.007
Qi X, Zha J, Liu GG, Zhang W, Li BZ, Yuan YJ (2015) Heterologous xylose isomerase pathway and evolutionary engineering improve xylose utilization in Saccharomyces cerevisiae. Front Microbiol 6:1165. https://doi.org/10.3389/fmicb.2015.01165
Sauer U (2001) Evolutionary engineering of industrially important microbial phenotypes. Adv Biochem Eng Biotechnol 73:129–169
Shui ZX, Qin H, Wu B, Ruan ZY, Wang LS, Tan FR, Wang JL, Tang XY, Dai LC, Hu GQ, He MX (2015) Adaptive laboratory evolution of ethanologenic Zymomonas mobilis strain tolerant to furfural and acetic acid inhibitors. Appl Microbiol Biotechnol 99(13):5739–5748. https://doi.org/10.1007/s00253-015-6616-z
Tan Z, Chen J, Zhang X (2016) Systematic engineering of pentose phosphate pathway improves Escherichia coli succinate production. Biotechnol Biofuels 9:262. https://doi.org/10.1186/s13068-016-0675-y
Thompson AD, Bernard SM, Skiniotis G, Gestwicki JE (2012) Visualization and functional analysis of the oligomeric states of Escherichia coli heat shock protein 70 (Hsp70/DnaK). Cell Stress Chaperones 17(3):313–327. https://doi.org/10.1007/s12192-011-0307-1
Tian K, Niu D, Liu X, Prior BA, Zhou L, Lu F, Singh S, Wang Z (2016) Limitation of thiamine pyrophosphate supply to growing Escherichia coli switches metabolism to efficient d-lactate formation. Biotechnol Bioeng 113(1):182–188. https://doi.org/10.1002/bit.25699
Valgepea K, Adamberg K, Seiman A, Vilu R (2013) Escherichia coli achieves faster growth by increasing catalytic and translation rates of proteins. Mol BioSyst 9(9):2344–2358. https://doi.org/10.1039/c3mb70119k
Wang Y, Tian T, Zhao J, Wang J, Yan T, Xu L, Liu Z, Garza E, Iverson A, Manow R, Finan C, Zhou S (2012) Homofermentative production of D-lactic acid from sucrose by a metabolically engineered Escherichia coli. Biotechnol Lett 34(11):2069–2075. https://doi.org/10.1007/s10529-012-1003-7
Wang ZW, Saini M, Lin LJ, Chiang CJ, Chao YP (2015) Systematic engineering of Escherichia coli for d-lactate production from crude glycerol. J Agric Food Chem 63(43):9583–9589. https://doi.org/10.1021/acs.jafc.5b04162
Wang J, Lin M, Xu M, Yang ST (2016) Anaerobic fermentation for production of carboxylic acids as bulk chemicals from renewable biomass. Adv Biochem Eng Biotechnol 156:323–361. https://doi.org/10.1007/10_2015_5009
Weusthuis RA, Lamot I, van der Oost J, Sanders JP (2011) Microbial production of bulk chemicals: development of anaerobic processes. Trends Biotechnol 29(4):153–158. https://doi.org/10.1016/j.tibtech.2010.12.007
Winkler JD, Kao KC (2014) Recent advances in the evolutionary engineering of industrial biocatalysts. Genomics 104(6 Pt A):406–411. https://doi.org/10.1016/j.ygeno.2014.09.006
Wu SG, He L, Wang Q, Tang YJ (2015) An ancient Chinese wisdom for metabolic engineering: Yin-Yang. Microb Cell Factories 14(1):39. https://doi.org/10.1186/s12934-015-0219-3
Xu K, Lv B, Huo YX, Li C (2018) Toward the lowest energy consumption and emission in biofuel production: combination of ideal reactors and robust hosts. Curr Opin Biotechnol 50:19–24. https://doi.org/10.1016/j.copbio.2017.08.011
Zeikus JG (1980) Chemical and fuel production by anaerobic bacteria. Annu Rev Microbiol 34:423–464. https://doi.org/10.1146/annurev.mi.34.100180.002231
Zhang X, Jantama K, Moore JC, Jarboe LR, Shanmugam KT, Ingram LO (2009) Metabolic evolution of energy-conserving pathways for succinate production in Escherichia coli. Proc Natl Acad Sci U S A 106(48):20180–20185. https://doi.org/10.1073/pnas.0905396106
Zhang H, Yang J, Wu S, Gong W, Chen C, Perrett S (2016) Glutathionylation of the bacterial Hsp70 chaperone DnaK provides a link between oxidative stress and the heat shock response. J Biol Chem 291(13):6967–6981. https://doi.org/10.1074/jbc.M115.673608
Zhao T, Liu D, Ren H, Shi X, Zhao N, Chen Y, Ying H (2014) D-lactic acid production by Sporolactobacillus inulinus Y2-8 immobilized in fibrous bed bioreactor using corn flour hydrolyzate. J Microbiol Biotechnol 24(12):1664–1672
Zhou S, Causey TB, Hasona A, Shanmugam KT, Ingram LO (2003) Production of optically pure d-lactic acid in mineral salts medium by metabolically engineered Escherichia coli W3110. Appl Environ Microbiol 69(1):399–407. https://doi.org/10.1128/aem.69.1.399-407.2003
Zhou S, Grabar TB, Shanmugam KT, Ingram LO (2006) Betaine tripled the volumetric productivity of D(−)-lactate by Escherichia coli strain SZ132 in mineral salts medium. Biotechnol Lett 28(9):671–676. https://doi.org/10.1007/s10529-006-0033-4
Zhu J, Shimizu K (2004) The effect of pfl gene knockout on the metabolism for optically pure D-lactate production by Escherichia coli. Appl Microbiol Biotechnol 64(3):367–375. https://doi.org/10.1007/s00253-003-1499-9
Zhu J, Shimizu K (2005) Effect of a single-gene knockout on the metabolic regulation in Escherichia coli for D-lactate production under microaerobic condition. Metab Eng 7(2):104–115. https://doi.org/10.1016/j.ymben.2004.10.004
Acknowledgements
We thank Prof. Qinghong Wang (TIB, CAS) for his kind gift of wild-type strain of this study. We also thank Dr. Zhubo Dai (TIB, CAS) and Yufeng Mao (TJU, China) for discussion on whole genome resequencing analysis.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC-21776208, NSFC- 21621004, NSFC-21776209 and NSFC-21390201).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 800 kb)
Rights and permissions
About this article
Cite this article
Wang, B., Zhang, X., Yu, X. et al. Evolutionary engineering of Escherichia coli for improved anaerobic growth in minimal medium accelerated lactate production. Appl Microbiol Biotechnol 103, 2155–2170 (2019). https://doi.org/10.1007/s00253-018-09588-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-09588-9