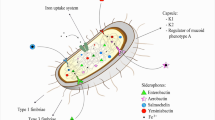
Abstract
Vibrio cholerae, the Gram-negative bacterium causing lethal diarrheal disease cholera, forms biofilm on solid surfaces to gain adaptive advantage for successful survival in aquatic reservoirs. Expression of exopolysaccharide (EPS), an extracellular matrix material, has been found critical for biofilm-based environmental persistence. In a subset of epidemic-causing V. cholerae, absence of flagellum but not motility was identified to induce elevated exopolysaccharide expression. Identification of the role played by quorum sensing autoinducer molecules, i.e., cholera autoinducer 1 (CAI-1) and autoinducer 2 (AI-2) as well as central regulator LuxO on EPS expression in the subset was explored. Deletion mutations were introduced in vital genes responsible for synthesizing CAI-1 (cqsA), AI-2 (luxS), flagellum (flaA), LuxO (luxO), flagellar motor (motX), and VpsR (vpsR) in the model strain MO10. Subsequent phenotypic alterations in terms of colony morphology, EPS expression, biofilm formation, and transcription level of relevant genes were analyzed. Autoinducer cross-feeding experiment confirmed the role of autoinducers in EPS signaling. Results reveal that autoinducers and flagellum are the two major EPS signaling units in this subset where one unit becomes predominant for EPS production in absence of the other. Moreover, either unit exerts negative influence on EPS induction by the other. Both the EPS signaling cascades are independent of LuxO contribution and essentially involve sodium-driven flagellar motor and VpsR. A cell density and flagellum-mediated, but LuxO-independent, EPS signaling mechanism is considered to be functional in these organisms that confers their survival fitness.





Similar content being viewed by others
References
Organization WH (2013) Monthly report on dracunculiasis cases, January-May 2013, Rapport mensuel des cas de dracunculose, janvier-mai 2013. Wkly Epidemiol. Rec. 88:335–336
Levine MM, Kaper JB, Black R, Clements ML (1983) New knowledge on pathogenesis of bacterial enteric infections as applied to vaccine development. Microbiol. Rev. 47:510
Taylor RK, Miller VL, Furlong DB, Mekalanos JJ (1987) Use of phoA gene fusions to identify a pilus colonization factor coordinately regulated with cholera toxin. Proc. Natl. Acad. Sci. U. S. A. 84:2833–2837
Skorupski K, Taylor RK (1997) Control of the ToxR virulence regulon in Vibrio cholerae by environmental stimuli. Mol. Microbiol. 25:1003–1009
Watnick PI, Lauriano CM, Klose KE, Croal L, Kolter R (2001) The absence of a flagellum leads to altered colony morphology, biofilm development and virulence in Vibrio cholerae O139. Mol. Microbiol. 39:223–235
Lutz C, Erken M, Noorian P, Sun S, McDougald D (2013) Environmental reservoirs and mechanisms of persistence of Vibrio cholerae. Front. Microbiol. 4:375
Yildiz FH, Visick KL (2009) Vibrio biofilms: so much the same yet so different. Trends Microbiol. 17:109–118
Yildiz FH, Schoolnik GK (1999) Vibrio cholerae O1 El Tor: identification of a gene cluster required for the rugose colony type, exopolysaccharide production, chlorine resistance, and biofilm formation. Proc. Natl. Acad. Sci. U. S. A. 96:4028–4033
White PB (1938) The rugose variant of Vibrios. J. Pathol. 46:1–6
Zhu J, Mekalanos JJ (2003) Quorum sensing-dependent biofilms enhance colonization in Vibrio cholerae. Dev. Cell 5:647–656
Miller MB, Skorupski K, Lenz DH, Taylor RK, Bassler BL (2002) Parallel quorum sensing systems converge to regulate virulence in Vibrio cholerae. Cell 110:303–314
Henke JM, Bassler BL (2004) Three parallel quorum-sensing systems regulate gene expression in Vibrio harveyi. J. Bacteriol. 186:6902–6914
Ng WL, Perez LJ, Wei Y, Kraml C, Semmelhack MF, Bassler BL (2011) Signal production and detection specificity in Vibrio CqsA/CqsS quorum-sensing systems. Mol. Microbiol. 79:1407–1417
Wei Y, Perez LJ, Ng W-L, Semmelhack MF, Bassler BL (2011) Mechanism of Vibrio cholerae autoinducer-1 biosynthesis. ACS Chem. Biol. 6:356–365
Schauder S, Shokat K, Surette MG, Bassler BL (2001) The LuxS family of bacterial autoinducers: biosynthesis of a novel quorum-sensing signal molecule. Mol. Microbiol. 41:463–476
Bassler BL (1999) How bacteria talk to each other: regulation of gene expression by quorum sensing. Curr. Opin. Microbiol. 2:582–587
Higgins DA, Pomianek ME, Kraml CM, Taylor RK, Semmelhack MF, Bassler BL (2007) The major Vibrio cholerae autoinducer and its role in virulence factor production. Nature 450:883–886
Jobling MG, Holmes RK (1997) Characterization of hapR, a positive regulator of the Vibrio cholerae HA/protease gene hap, and its identification as a functional homologue of the Vibrio harveyi luxR gene. Mol. Microbiol. 26:1023–1034
Römling U, Amikam D (2006) Cyclic di-GMP as a second messenger. Curr. Opin. Microbiol. 9:218–228
Hammer BK, Bassler BL (2009) Distinct sensory pathways in Vibrio cholerae El Tor and classical biotypes modulate cyclic dimeric GMP levels to control biofilm formation. J. Bacteriol. 191:169–177
Jung SA, Chapman CA, Ng W-L (2015) Quadruple quorum-sensing inputs control Vibrio cholerae virulence and maintain system robustness. PLoS Pathog. 11:e1004837
Papenfort K, Bassler BL (2016) Quorum sensing signal-response systems in gram-negative bacteria. Nat Rev Microbiol 14:576–588
Lauriano CM, Ghosh C, Correa NE, Klose KE (2004) The sodium-driven flagellar motor controls exopolysaccharide expression in Vibrio cholerae. J. Bacteriol. 186:4864–4874
Shao Y, Bassler BL (2014) Quorum regulatory small RNAs repress type VI secretion in Vibrio cholerae. Mol. Microbiol. 92:921–930
Joelsson A, Liu Z, Zhu J (2006) Genetic and phenotypic diversity of quorum-sensing systems in clinical and environmental isolates of Vibrio cholerae. Infect. Immun. 74:1141–1147
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J. Mol. Biol. 166:557–580
Miller VL, Mekalanos JJ (1988) A novel suicide vector and its use in construction of insertion mutations: osmoregulation of outer membrane proteins and virulence determinants in Vibrio cholerae requires toxR. J. Bacteriol. 170:2575–2583
Correa NE, Lauriano CM, McGee R, Klose KE (2000) Phosphorylation of the flagellar regulatory protein FlrC is necessary for Vibrio cholerae motility and enhanced colonization. Mol. Microbiol. 35:743–755
Simons RW, Houman F, Kleckner N (1987) Improved single and multicopy lac-based cloning vectors for protein and operon fusions. Gene 53:85–96
Sambrook J, Russel DW, Russel DW (2001) Molecular cloning: a laboratory manual (Ed. 3). Cold Spring Harbor Laboratory Press, NY, USA
Heidelberg JF, Eisen JA, Nelson WC, Clayton RA, Gwinn ML, Dodson RJ, Haft DH, Hickey EK, Peterson JD, Umayam L (2000) DNA sequence of both chromosomes of the cholera pathogen Vibrio cholerae. Nature 406:477–483
Miller JH (1992) A short course in bacterial genetics : a laboratory manual and handbook for Escherichia coli and related bacteria. Cold Spring Harbor laboratory press. New York (N.Y.)
Sun M-L, Zhao F, Shi M, Zhang X-Y, Zhou B-C, Zhang Y-Z, Chen X-L (2015) Characterization and biotechnological potential analysis of a new exopolysaccharide from the Arctic marine bacterium Polaribacter sp. SM1127. Sci. Rep. 5:18435
Dubois M, Gilles KA, Hamilton JK, Pt R, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal. Chem. 28:350–356
Fong JC, Yildiz FH (2007) The rbmBCDEF gene cluster modulates development of rugose colony morphology and biofilm formation in Vibrio cholerae. J. Bacteriol. 189:2319–2330
Mok KC, Wingreen NS, Bassler BL (2003) Vibrio harveyi quorum sensing: a coincidence detector for two autoinducers controls gene expression. EMBO J. 22:870–881
Antonova ES, Hammer BK (2011) Quorum-sensing autoinducer molecules produced by members of a multispecies biofilm promote horizontal gene transfer to Vibrio cholerae. FEMS Microbiol. Lett. 322:68–76
Bassler BL, Wright M, Silverman MR (1994) Multiple signalling systems controlling expression of luminescence in Vibrio harveyi: sequence and function of genes encoding a second sensory pathway. Mol. Microbiol. 13:273–286
Kawagishi I, Imagawa M, Imae Y, McCarter L, Homma M (1996) The sodium-driven polar flagellar motor of marine Vibrio as the mechanosensor that regulates lateral flagellar expression. Mol. Microbiol. 20:693–699
Yildiz FH, Dolganov NA, Schoolnik GK (2001) VpsR, a member of the response regulators of the two-component regulatory systems, is required for expression of vps biosynthesis genes and EPSETr-associated phenotypes in Vibrio cholerae O1 El Tor. J. Bacteriol. 183:1716–1726
Acknowledgements
Authors thank Prof. Karl E. Klose (Dept. of Biology, UTSA, USA) for gifts of strains and plasmid vectors and Prof. Sunando Bandyopadhyay, Department of Geography, University of Calcutta, for technical support.
Funding
This study is financially supported by the Science and Engineering Research Council, Department of Science and Technology (DST), Government of India, in the form of a research grant (SR/SO/HS-43/2006), and the Department of Biotechnology, Govt. of India (Project Number BT/PR3802/BRB/10/981/2011).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Biswas, S., Mukherjee, P., Manna, T. et al. Quorum Sensing Autoinducer(s) and Flagellum Independently Mediate EPS Signaling in Vibrio cholerae Through LuxO-Independent Mechanism. Microb Ecol 77, 616–630 (2019). https://doi.org/10.1007/s00248-018-1262-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-018-1262-5