Abstract
Background
At fetal MR, congenital lung lesions are usually T2 hyperintense with respect to normal lung parenchyma. Some lesions, however, demonstrate unusual patterns of T2 hypointensity, sometimes in a rosette-like pattern. These lesions usually present a diagnostic conundrum.
Objective
To evaluate the imaging findings and pathological characterization of fetal solid lung lesions with elements showing T2-hypointense signal with respect to lung.
Materials and methods
This is a retrospective study of lung lesions with elements showing T2 hypointensity treated prenatally and postnatally at our center and with available pathological evaluation. Prenatal imaging evaluation included US and MR; postnatal evaluation consisted of pathological examination of the lesion. We also performed prenatal and postnatal chart review.
Results
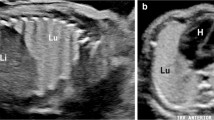
Six cases met study criteria. Areas of decreased echogenicity/T2-hypointense signal were more conspicuous at MR than US. At pathology, these areas correlated with immature parenchymal development and increased mesenchymal tissue. Five of these lesions were congenital pulmonary airway malformations (CPAM); one was a congenital peribronchial myofibroblastic tumor (CPMT). The lesions did not significantly change in size after steroid administration. They were all large in volume and were associated with increased amniotic fluid. All cases of CPAM underwent premature delivery (one of them weeks after fetal surgical resection of the lesion for worsening hydrops); the fetus with CPMT was delivered at term. The neonate with CPMT succumbed shortly after birth secondary to lung hypoplasia; the remaining five neonates survived.
Conclusion
The differential diagnoses of prenatal lung lesions that contain unusual T2-hypointense elements include CPAM and CPMT. The T2-hypointense areas appear to correlate with increasing degree of immaturity at histology. None of the lesions significantly changed in size after prenatal administration of steroids. All cases with CPAM lesions did well despite persistent polyhydramnios and premature birth. The single case of CPMT, however, resulted in neonatal demise shortly after birth secondary to pulmonary hypoplasia. It is important that fetal radiologists, obstetricians and fetal surgeons alike are aware of these lesions so that appropriate diagnosing and parental counseling can be reached.






Similar content being viewed by others
References
Langston C (2003) New concepts in the pathology of congenital lung malformations. Semin Pediatr Surg 12:17–37
Newman B (2006) Congenital bronchopulmonary foregut malformations: concepts and controversies. Pediatr Radiol 36:773–791
Pacharn P, Kline-Fath B, Calvo-Garcia M et al (2013) Congenital lung lesions: prenatal MRI and postnatal findings. Pediatr Radiol 43:1136–1143
Plunk MR, Chapman T (2014) The fundamentals of fetal magnetic resonance imaging: part 2. Curr Probl Diagn Radiol 43:347–355
Zucker EJ, Epelman M, Newman B (2015) Perinatal thoracic mass lesions: pre- and postnatal imaging. Semin Ultrasound CT MR 36:501–521
Calvo-Garcia MA, Lim FY, Stanek J et al (2014) Congenital peribronchial myofibroblastic tumor: prenatal imaging clues to differentiate from other fetal chest lesions. Pediatr Radiol 44:479–483
Lazar DA, Cass DL, Dishop MK et al (2011) Fetal lung interstitial tumor: a cause of late gestation fetal hydrops. J Pediatr Surg 46:1263–1266
Dishop MK, McKay EM, Kreiger PA et al (2010) Fetal lung interstitial tumor (FLIT): a proposed newly recognized lung tumor of infancy to be differentiated from cystic pleuropulmonary blastoma and other developmental pulmonary lesions. Am J Surg Pathol 34:1762–1772
Victoria T, Bebbington MW, Danzer E et al (2012) Use of magnetic resonance imaging in prenatal prognosis of the fetus with isolated left congenital diaphragmatic hernia. Prenat Diagn 32:715–723
Rypens F, Metens T, Rocourt N et al (2001) Fetal lung volume: estimation at MR imaging-initial results. Radiology 219:236–241
Crombleholme TM, Coleman B, Hedrick H et al (2002) Cystic adenomatoid malformation volume ratio predicts outcome in prenatally diagnosed cystic adenomatoid malformation of the lung. J Pediatr Surg 37:331–338
Rosado-de-Christenson ML, Stocker JT (1991) Congenital cystic adenomatoid malformation. Radiographics 11:865–886
Adzick NS, Harrison MR, Glick PL et al (1985) Fetal cystic adenomatoid malformation: prenatal diagnosis and natural history. J Pediatr Surg 20:483–488
Kreiger PA, Ruchelli ED, Mahboubi S et al (2006) Fetal pulmonary malformations: defining histopathology. Am J Surg Pathol 30:643–649
Cannie M, Jani J, Meersschaert J et al (2008) Prenatal prediction of survival in isolated diaphragmatic hernia using observed to expected total fetal lung volume determined by magnetic resonance imaging based on either gestational age or fetal body volume. Ultrasound Obstet Gynecol 32:633–639
Gorincour G, Bouvenot J, Mourot MG et al (2005) Prenatal prognosis of congenital diaphragmatic hernia using magnetic resonance imaging measurement of fetal lung volume. Ultrasound Obstet Gynecol 26:738–744
Kitagawa M, Hislop A, Boyden EA et al (1971) Lung hypoplasia in congenital diaphragmatic hernia. A quantitative study of airway, artery, and alveolar development. Br J Surg 58:342–346
Peranteau WH, Wilson RD, Liechty KW et al (2007) Effect of maternal betamethasone administration on prenatal congenital cystic adenomatoid malformation growth and fetal survival. Fetal Diagn Ther 22:365–371
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Victoria, T., Srinivasan, A.S., Pogoriler, J. et al. The rare solid fetal lung lesion with T2-hypointense components: prenatal imaging findings with postnatal pathological correlation. Pediatr Radiol 48, 1556–1566 (2018). https://doi.org/10.1007/s00247-018-4174-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-018-4174-0