Abstract
Background
Pediatric optic pathway gliomas are typically indolent but have a variable clinical course. Treatment is dictated by symptoms and changes on contrast-enhanced MRI examinations. Gadolinium retention in children has motivated parsimonious use of gadolinium-based contrast agents.
Objectives
To determine surveillance MR factors that motivate changes in tumor-directed therapies and extrapolate cost-efficacy of a non-contrast follow-up protocol.
Materials and methods
Using an imaging database search we identified children with isolated optic pathway gliomas and ≥3 follow-up contrast-enhanced MRIs. We reviewed medical records and imaging for: (1) coincident changes on contrast-enhanced MRI and tumor-directed therapy, (2) demographics and duration of follow-up, (3) motivations for intervention, (4) assessment of gadolinium-based contrast agents’ utility and (5) health care utilization data. We assessed cost impact in terms of relative value unit (RVU) burden.
Results
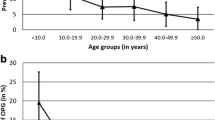
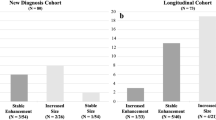
We included 17 neurofibromatosis type 1 (NF1) and 21 non-NF1 patients who underwent a median 16.9 and 24.3 cumulative contrast-enhanced MR exams over 7.7 years and 8.1 years of follow-up, respectively. Eight children (one with NF1) had intervention based on contrast-enhanced MR findings alone. For these eight, increased tumor size was the only common feature, and it was apparent on non-contrast T2 sequences. For the median patient, a non-contrast follow-up protocol could result in 15.9 (NF1) and 23.3 (non-NF1) fewer gadolinium-based contrast agent administrations, and a 39% lower yearly RVU burden.
Conclusion
Pediatric patients with isolated optic pathway gliomas undergo a large number of routine contrast-enhanced MR follow-up exams. Gadolinium might not be needed for these exams to inform management decisions. Secondary benefits of a non-contrast follow-up protocol include decreased cost and risk to the patient.




Similar content being viewed by others
References
Gale EM, Caravan P, Rao AG et al (2017) Gadolinium-based contrast agents in pediatric magnetic resonance imaging. Pediatr Radiol 47:507–521
McDonald JS, McDonald RJ, Jentoft ME et al (2017) Intracranial gadolinium deposition following gadodiamide-enhanced magnetic resonance imaging in pediatric patients: a case-control study. JAMA Pediatr 171:705–707
Kanda T, Fukusato T, Matsuda M et al (2015) Gadolinium-based contrast agent accumulates in the brain even in subjects without severe renal dysfunction: evaluation of autopsy brain specimens with inductively coupled plasma mass spectroscopy. Radiology 276:228–232
Miller JH, Hu HH, Pokorney A et al (2015) MRI brain signal intensity changes of a child during the course of 35 gadolinium contrast examinations. Pediatrics 136:e1637–e1640
Mithal LB, Patel PS, Mithal D et al (2017) Use of gadolinium-based magnetic resonance imaging contrast agents and awareness of brain gadolinium deposition among pediatric providers in North America. Pediatr Radiol 47:657–664
Roberts DR, Chatterjee AR, Yazdani M et al (2016) Pediatric patients demonstrate progressive T1-weighted hyperintensity in the dentate nucleus following multiple doses of gadolinium-based contrast agent. AJNR Am J Neuroradiol 37:2340–2347
Murata N, Gonzalez-Cuyar LF, Murata K et al (2016) Macrocyclic and other non-group 1 gadolinium contrast agents deposit low levels of gadolinium in brain and bone tissue: preliminary results from 9 patients with normal renal function. Investig Radiol 51:447–453
Roberts DR, Lindhorst SM, Welsh CT et al (2016) High levels of gadolinium deposition in the skin of a patient with normal renal function. Investig Radiol 51:280–289
Maximova N, Gregori M, Zennaro F et al (2016) Hepatic gadolinium deposition and reversibility after contrast agent-enhanced MR imaging of pediatric hematopoietic stem cell transplant recipients. Radiology 281:418–426
Rogosnitzky M, Branch S (2016) Gadolinium-based contrast agent toxicity: a review of known and proposed mechanisms. Biometals 29:365–376
Semelka RC, Ramalho M, AlObaidy M, Ramalho J (2016) Gadolinium in humans: a family of disorders. AJR Am J Roentgenol 207:229–233
U. S. Food and Drug Administration (2017) Drug safety communications. FDA evaluating the risk of brain deposits with repeated use of gadolinium-based contrast agents for magnetic resonance imaging. https://www.fda.gov/downloads/Drugs/DrugSafety/UCM455390.pdf. Accessed 17 April 2017
European Medicines Agency (2017) PRAC concludes assessment of gadolinium agents used in body scans and recommends regulatory actions, including suspension for some marketing authorisations. http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2017/03/WC500223209.pdf. Accessed 17 April 2017
American College of Radiology (2017) ACR manual on contrast media version 10.3 (78). https://www.acr.org/-/media/ACR/Files/Clinical-Resources/Contrast_Media.pdf. Accessed 12 April 2018
American College of Radiology (2017) News releases: ACR response to the European PRAC recommendations (April 4, 2017). http://www.publicnow.com/view/DA714236788989CE92F38E18EA3CCB6FCC9DFDE3?2017-04-04-17:31:30+01:00-xxx935. Accessed 12 April 2018
Goischke HK (2017) Safety assessment of gadolinium-based contrast agents (GBCAs) requires consideration of long-term adverse effects in all human tissues. Mult Scler J Exp Transl Clin 3:2055217317704450
Gupta A, Al-Dasuqi K, Xia F et al (2017) The use of noncontrast quantitative MRI to detect gadolinium-enhancing multiple sclerosis brain lesions: a systematic review and meta-analysis. AJNR Am J Neuroradiol 38:1317–1322
Heideman RL (1993) Tumors of the central nervous system. In: Pizzo PA, Poplack DG (eds) Principles and practice of pediatric oncology. Lippincott-Raven, Philadelphia, pp 633–681
Ostrom QT, Gittleman H, Fulop J et al (2015) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008-2012. Neuro Oncol 17:iv1–iv62
Albers AC, Gutmann DH (2009) Gliomas in patients with neurofibromatosis type 1. Expert Rev Neurother 9:535–539
Recht LD (2017) Optic pathway glioma. In: UpToDate. https://www.uptodate.com/contents/optic-pathway-glioma. Accessed 12 April 2018
Wan MJ, Ullrich NJ, Manley PE et al (2016) Long-term visual outcomes of optic pathway gliomas in pediatric patients without neurofibromatosis type 1. J Neuro Oncol 129:173–178
Gaudino S, Martucci M, Russo R et al (2017) MR imaging of brain pilocytic astrocytoma: beyond the stereotype of benign astrocytoma. Childs Nerv Syst 33:35–54
Hernaiz Driever P, von Hornstein S, Pietsch T et al (2010) Natural history and management of low-grade glioma in NF-1 children. J Neuro Oncol 100:199–207
Kornreich L, Blaser S, Schwarz M et al (2001) Optic pathway glioma: correlation of imaging findings with the presence of neurofibromatosis. AJNR Am J Neuroradiol 22:1963–1969
Kerrison JB (2005) Chapter 38: phacomatoses. In: Miller NR (ed) Walsh & Hoyt's clinical neuro-ophthalmology, 6th edn. Lippincott Williams & Wilkins, Philadelphia, pp 1823–1898
Chateil JF, Soussotte C, Pedespan JM et al (2001) MRI and clinical differences between optic pathway tumours in children with and without neurofibromatosis. Br J Radiol 74:24–31
Prada CE, Hufnagel RB, Hummel TR et al (2015) The use of magnetic resonance imaging screening for optic pathway gliomas in children with neurofibromatosis type 1. J Pediatr 167:851–856
Listernick R, Ferner RE, Liu GT, Gutmann DH (2007) Optic pathway gliomas in neurofibromatosis-1: controversies and recommendations. Ann Neurol 61:189–198
Balcer LJ, Liu GT, Heller G et al (2001) Visual loss in children with neurofibromatosis type 1 and optic pathway gliomas. Relation to tumor location by magnetic resonance imaging. Am J Ophthalmol 131:442–445
Listernick R, Louis DN, Packer RJ, Gutmann DH (1997) Optic pathway gliomas in children with neurofibromatosis 1: consensus statement from the NF1 optic pathway glioma task force. Neuro Oncol 41:143–149
Liu GT (2006) Optic gliomas of the anterior visual pathway. Curr Opin Ophthalmol 17:427–431
Thiagalingam S, Flaherty M, Billson F, North K (2004) Neurofibromatosis type 1 and optic pathway gliomas: follow-up of 54 patients. Ophthalmology 111:568–577
Fisher MJ, Loguidice M, Gutmann DH et al (2012) Visual outcomes in children with neurofibromatosis type 1-associated optic pathway glioma following chemotherapy: a multicenter retrospective analysis. Neuro-Oncology 14:790–797
Ater JL, Zhou T, Holmes E et al (2012) Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children's oncology group. J Clin Oncol 30:2641–2647
Kelly JP, Leary S, Khanna P, Weiss AH (2012) Longitudinal measures of visual function, tumor volume, and prediction of visual outcomes after treatment of optic pathway gliomas. Ophthalmology 119:1231–1237
Shofty B, Mauda-Havakuk M, Weizman L et al (2015) The effect of chemotherapy on optic pathway gliomas and their sub-components: a volumetric MR analysis study. Pediatr Blood Cancer 62:1353–1359
van den Bent MJ, Wefel JS, Schiff D et al (2011) Response assessment in neuro-oncology (a report of the RANO group): assessment of outcome in trials of diffuse low-grade gliomas. Lancet Oncol 12:583–593
Wen PY, Chang SM, Van den Bent MJ et al (2017) Response assessment in neuro-oncology clinical trials. J Clin Oncol 35:2439–2449
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org. Accessed 14 March 2018
Centers for Medicare and Medicaid Services (2017) Searchable Medicare physician fee schedule. http://www.cms.gov/apps/physician-fee-schedule/overview.aspx. Accessed 12 Sept 2017
Clemens J, Gottlieb JD (2017) In the shadow of a giant: Medicare's influence on private physician payments. J Polit Econ 125:1–39
The Henry J. Kaiser Family Foundation (2016) State health facts — Medicaid-to-Medicare fee index (2016). http://www.kff.org/state-category/medicaid-chip/medicaid-physician-fees/. Accessed 15 Sept 2017
Taylor T, Jaspan T, Milano G et al (2008) Radiological classification of optic pathway gliomas: experience of a modified functional classification system. Br J Radiol 81:761–766
Dillman JR, Ellis JH, Cohan RH et al (2007) Frequency and severity of acute allergic-like reactions to gadolinium-containing i.v. contrast media in children and adults. AJR Am J Roentgenol 189:1533–1538
Astrup J (2003) Natural history and clinical management of optic pathway glioma. Br J Neurosurg 17:327–335
Dodge HW Jr, Love JG, Craig WM et al (1958) Gliomas of the optic nerves. AMA Arch Neurol Psychiatry 79:607–621
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Pollack IF, Hoffman HJ, Humphreys RP, Becker L (1993) The long-term outcome after surgical treatment of dorsally exophytic brain-stem gliomas. J Neurosurg 78:859–863
Lee AG (2007) Neuroophthalmological management of optic pathway gliomas. Neurosurg Focus 23:E1
Lambron J, Rakotonjanahary J, Loisel D et al (2016) Can we improve accuracy and reliability of MRI interpretation in children with optic pathway glioma? Proposal for a reproducible imaging classification. Neuroradiology 58:197–208
Weizman L, Sira LB, Joskowicz L et al (2014) Semiautomatic segmentation and follow-up of multicomponent low-grade tumors in longitudinal brain MRI studies. Med Phys 41:052303
Weizman L, Ben Sira L, Joskowicz L et al (2012) Automatic segmentation, internal classification, and follow-up of optic pathway gliomas in MRI. Med Image Anal 16:177–188
Shofty B, Weizman L, Joskowicz L et al (2011) MRI internal segmentation of optic pathway gliomas: clinical implementation of a novel algorithm. Childs Nerv Syst 27:1265–1272
Gaudino S, Quaglio F, Schiarelli C et al (2012) Spontaneous modifications of contrast enhancement in childhood non-cerebellar pilocytic astrocytomas. Neuroradiology 54:989–995
Beni-Adani L, Gomori M, Spektor S, Constantini S (2000) Cyst wall enhancement in pilocytic astrocytoma: neoplastic or reactive phenomena. Pediatr Neurosurg 32:234–239
Takeuchi H, Kubota T, Sato K, Arishima H (2004) Ultrastructure of capillary endothelium in pilocytic astrocytomas. Brain Tumor Pathol 21:23–26
Strong JA, Hatten HP Jr, Brown MT et al (1993) Pilocytic astrocytoma: correlation between the initial imaging features and clinical aggressiveness. AJR Am J Roentgenol 161:369–372
Jittapiromsak N, Hou P, Liu HL et al (2017) Prognostic role of conventional and dynamic contrast-enhanced MRI in optic pathway gliomas. J Neuroimaging 27:594–601
Jost SC, Ackerman JW, Garbow JR et al (2008) Diffusion-weighted and dynamic contrast-enhanced imaging as markers of clinical behavior in children with optic pathway glioma. Pediatr Radiol 38:1293–1299
Singhal S, Birch JM, Kerr B et al (2002) Neurofibromatosis type 1 and sporadic optic gliomas. Arch Dis Child 87:65–70
Campian J, Gutmann DH (2017) CNS tumors in neurofibromatosis. J Clin Oncol 35:2378–2385
Guillamo JS, Creange A, Kalifa C et al (2003) Prognostic factors of CNS tumours in neurofibromatosis 1 (NF1): a retrospective study of 104 patients. Brain 126:152–160
Mentzel HJ, Seidel J, Fitzek C et al (2005) Pediatric brain MRI in neurofibromatosis type I. Eur Radiol 15:814–822
Bonawitz C, Castillo M, Chin CT et al (1998) Usefulness of contrast material in MR of patients with neurofibromatosis type 1. AJNR Am J Neuroradiol 19:541–546
Jost G, Lenhard DC, Sieber MA et al (2016) Signal increase on unenhanced T1-weighted images in the rat brain after repeated, extended doses of gadolinium-based contrast agents: comparison of linear and macrocyclic agents. Investig Radiol 51:83–89
Sieber MA, Steger-Hartmann T, Lengsfeld P, Pietsch H (2009) Gadolinium-based contrast agents and NSF: evidence from animal experience. J Magn Reson Imaging 30:1268–1276
Radbruch A, Haase R, Kickingereder P et al (2017) Pediatric brain: no increased signal intensity in the dentate nucleus on unenhanced T1-weighted MR images after consecutive exposure to a macrocyclic gadolinium-based contrast agent. Radiology 283:828–836
Flood TF, Stence NV, Maloney JA, Mirsky DM (2017) Pediatric brain: repeated exposure to linear gadolinium-based contrast material is associated with increased signal intensity at unenhanced T1-weighted MR imaging. Radiology 282:222–228
Abraham JL, Thakral C (2008) Tissue distribution and kinetics of gadolinium and nephrogenic systemic fibrosis. Eur J Radiol 66:200–207
Costello JR, Kalb B, Martin DR (2016) Incidence and risk factors for gadolinium-based contrast agent immediate reactions. Top Magn Reson Imaging 25:257–263
Jung JW, Kang HR, Kim MH et al (2012) Immediate hypersensitivity reaction to gadolinium-based MR contrast media. Radiology 264:414–422
Stecker EC, Schroeder SA (2013) Adding value to relative-value units. N Engl J Med 369:2176–2179
The Henry J. Kaiser Family Foundation (2017) Next steps for CHIP: what is at stake for children? http://files.kff.org/attachment/Fact-Sheet-Next-Steps-for-CHIP-What-is-at-Stake-for-Children. Accessed 14 Sept 2017
Patton DD, Woolfenden JM (1989) A utility-based model for comparing the cost-effectiveness of diagnostic studies. Investig Radiol 24:263–271
Hollingworth W, Jarvik JG (2007) Technology assessment in radiology: putting the evidence in evidence-based radiology. Radiology 244:31–38
Acknowledgment
Dr. Maloney is supported, in part, through a National Institutes of Health, National Research Service Award training grant (grant number 5 T32 DK0072).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
This article was awarded the John Kirkpatrick Young Investigator Award at the Society for Pediatric Radiology 2017 meeting.
Rights and permissions
About this article
Cite this article
Maloney, E., Stanescu, A.L., Perez, F.A. et al. Surveillance magnetic resonance imaging for isolated optic pathway gliomas: is gadolinium necessary?. Pediatr Radiol 48, 1472–1484 (2018). https://doi.org/10.1007/s00247-018-4154-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-018-4154-4