Abstract
Background
Glomerular hyperfiltration has recently been reported in children with malignancies and has been attributed to increased solute from breakdown of tumor tissues.
Objective
To evaluate the prevalence of hyperfiltration in the pediatric oncology population and explore its pathophysiological mechanism.
Materials and methods
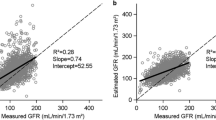
Tc-99 m diethylenetriaminepentaacetic acid (DTPA) glomerular filtration rate (GFR) examinations (437 studies) and medical records of 177 patients <21 years of age diagnosed with a malignancy between January 2005 and October 2013 were retrospectively reviewed. Hyperfiltration was defined as a GFR ≥ 160 ml/min/1.73 m2.
Results
Seventy-seven (43.5%) patients had hyperfiltration in at least one GFR exam. A significantly higher percentage of patients with central nervous system (CNS) tumors (63.6%) had hyperfiltration when compared to other tumor types (27.3%, P < 0.001). No association was found between hyperfiltration and age, gender, race or bone marrow involvement. There was a significant trend toward decreasing hyperfiltration after the second cycle of chemotherapy (P = 0.006) and a significant increase in subjects with low GFR (<100 ml/min/1.73 m2) with increasing number of cycles of chemotherapy (P = 0.005).
Conclusion
Glomerular hyperfiltration is common in children with malignancies at diagnosis and during initial cycles of chemotherapy. It is particularly prevalent in patients with central nervous tumors, which are frequently smaller in volume. Therefore, the pathophysiological mechanism of hyperfiltration cannot be explained solely on the basis of large tumor volume and subsequent cell breakdown. We hypothesize that host hypermetabolic state plays an important role in pathophysiology of hyperfiltration.



Similar content being viewed by others
References
Hjorth L, Wiebe T, Karpman D (2011) Hyperfiltration evaluated by glomerular filtration rate at diagnosis in children with cancer. Pediatr Blood Cancer 56:762–766
Fleming JS, Zivanovic MA, Blake GM et al (2004) Guidelines for the measurement of glomerular filtration rate using plasma sampling. Nucl Med Commun 25:759–769
Piepsz A, Pintelon H, Ham HR (1994) Estimation of normal chromium-51 ethylene diamine tetra-acetic acid clearance in children. Eur J Nucl Med 21:12–16
Brandt JR, Wong C, Jones DR et al (2003) Glomerular filtration rate in children with solid tumors: normative values and a new method for estimation. Pediatr Hematol Oncol 20:309–318
Blake GM, Gardiner N, Gnanasegaran G et al (2005) Reference ranges for 51Cr-EDTA measurements of glomerular filtration rate in children. Nucl Med Commun 26:983–987
Piepsz A, Tondeur M, Ham H (2006) Revisiting normal (51)Cr-ethylenediaminetetraacetic acid clearance values in children. Eur J Nucl Med Mol Imaging 33:1477–1482
Bhatt MK, Bartlett ML, Mallitt K-A et al (2011) Correlation of various published radionuclide glomerular filtration rate estimation techniques and proposed paediatric normative data. Nucl Med Commun 32:1088–1094
Thomas DM, Coles GA, Williams JD (1994) What does the renal reserve mean? Kidney Int 45:411–416
Aygun B, Mortier NA, Smeltzer MP et al (2013) Hydroxyurea treatment decreases glomerular hyperfiltration in children with sickle cell anemia. Am J Hematol 88:116–119
Helal I, Fick-Brosnahan GM, Reed-Gitomer B et al (2012) Glomerular hyperfiltration: definitions, mechanisms and clinical implications. Nat Rev Nephrol 8:293–300
Douglas RG, Shaw JH (1990) Metabolic effects of cancer. Br J Surg 77:246–254
Jeevanandam M, Horowitz GD, Lowry SF et al (1984) Cancer cachexia and protein metabolism. Lancet 1:1423–1426
Donohoe CL, Ryan AM, Reynolds JV (2011) Cancer cachexia: mechanisms and clinical implications. Gastroenterol Res Pract 2011:601434
Fearon KCH, Glass DJ, Guttridge DC (2012) Cancer cachexia: mediators, signaling, and metabolic pathways. Cell Metab 16:153–166
Li Z, Zhang H (2016) Reprogramming of glucose, fatty acid and amino acid metabolism for cancer progression. Cell Mol Life Sci 73:377–392
Stallings VA, Vaisman N, Chan HS et al (1989) Energy metabolism in children with newly diagnosed acute lymphoblastic leukemia. Pediatr Res 26:154–157
den Broeder E, Oeseburg B, Lippens RJ et al (2001) Basal metabolic rate in children with a solid tumour. Eur J Clin Nutr 55:673–681
Picton SV (1998) Aspects of altered metabolism in children with cancer. Int J Cancer Suppl 11:62–64
Green GJ, Weitzman SS, Pencharz PB (2008) Resting energy expenditure in children newly diagnosed with stage IV neuroblastoma. Pediatr Res 63:332–336
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Kwatra, N.S., Meany, H.J., Ghelani, S.J. et al. Glomerular hyperfiltration in children with cancer: prevalence and a hypothesis. Pediatr Radiol 47, 221–226 (2017). https://doi.org/10.1007/s00247-016-3733-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-016-3733-5