Abstract
Background
Multivisceral transplantation represents an important treatment option for children with intestinal failure. The attendant immunosuppression can lead to a spectrum of cellular proliferations including benign and malignant smooth muscle tumors and lymphoproliferative disorders, many related to cellular dysregulation from Epstein-Barr virus infection.
Objective
The purpose of this study is to investigate the rates of post-transplantation proliferative disorders among children with multivisceral transplantation and to characterize the imaging and pathological features of these disorders.
Materials and methods
We identified all consecutive children who underwent multivisceral transplant from August 2004 to October 2011 with at least 27 months of clinical and imaging follow-up. We reviewed medical records to determine the underlying causes of the multivisceral transplant, age at transplantation, onset of neoplasm development, and outcome. Two pediatric radiologists reviewed all imaging studies independently and diagnosis of disease was made by consensus interpretation. Pathological specimens were reviewed for histopathological findings of post-transplantation neoplasm in this pediatric patient population.
Results
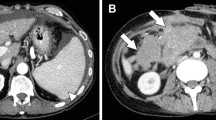
The study population consisted of 14 consecutive pediatric patients (7 boys and 7 girls; mean age 26 months, range 4–113 months). Of these 14 children, 4 (29%) developed histologically confirmed post-transplant neoplasms at a mean time of 2.4 years after multivisceral transplantation. Types of neoplasms included post-transplant lymphoproliferative disorder (PTLD) in three (21%) and Epstein-Barr-virus-associated smooth muscle tumor in two (14%). (One child developed both neoplasms following transplantation). Both children with smooth muscle tumor associated with Epstein-Barr virus presented with characteristic hypointense solid masses with peripheral rim enhancement on cross-sectional imaging studies. The mortality rate of children who developed post-transplant neoplasms was higher than that of those who did not develop post-transplant neoplasm (50% vs. 10%, P = 0.17), suggesting a possible risk factor for death.
Conclusion
Post-transplant neoplasm in children with multivisceral transplantation occurs with high frequency, often presents as Epstein-Barr-virus-associated smooth muscle tumor showing characteristic peripheral rim enhancement on cross-sectional imaging studies.



Similar content being viewed by others
References
LaRosa C, Baluarte HJ, Meyers KE (2011) Outcomes in pediatric solid-organ transplantation. Pediatr Transplant 15:128–141
Gross TG, Savoldo B, Punnett A (2010) Posttransplant lymphoproliferative diseases. Pediatr Clin N Am 57:481–503
Buell JF, Gross TG, Woodle ES (2005) Malignancy after transplantation. Transplantation 80:S254–S264
Green M, Michaels MG (2013) Epstein–Barr virus infection and posttransplant lymphoproliferative disorder. Am J Transplant 13:41–54, quiz 54
Swerdlow S, Campo E, Harris NL et al (2008) WHO classification of tumours of haematopoietic and lymphoid tissues, 4th edn. World Health Organization, Geneva
Mucha K, Foroncewicz B, Ziarkiewicz-Wroblewska B et al (2010) Post-transplant lymphoproliferative disorder in view of the new WHO classification: a more rational approach to a protean disease? Nephrol Dial Transplant 25:2089–2098
Penn I (1998) De novo malignances in pediatric organ transplant recipients. Pediatr Transplant 2:56–63
Cockfield SM (2001) Identifying the patient at risk for post-transplant lymphoproliferative disorder. Transpl Infect Dis 3:70–78
Tzakis AG, Kato T, Levi DM et al (2005) 100 multivisceral transplants at a single center. Ann Surg 242:480–490, discussion 491–483
Kato T, Tzakis AG, Selvaggi G et al (2006) Intestinal and multivisceral transplantation in children. Ann Surg 243:756–764, discussion 764–756
Siegel MJ, Lee EY, Sweet SC et al (2003) CT of posttransplantation lymphoproliferative disorder in pediatric recipients of lung allograft. AJR Am J Roentgenol 181:1125–1131
Lim GY, Newman B, Kurland G et al (2002) Posttransplantation lymphoproliferative disorder: manifestations in pediatric thoracic organ recipients. Radiology 222:699–708
Wilde GE, Moore DJ, Bellah RD (2005) Posttransplantation lymphoproliferative disorder in pediatric recipients of solid organ transplants: timing and location of disease. AJR Am J Roentgenol 185:1335–1341
Hansell DM, Bankier AA, MacMahon H et al (2008) Fleischner society: glossary of terms for thoracic imaging. Radiology 246:697–722
Siegel MJ (2008) Pediatric Body CT, 2nd edn. Lippincott, Williams & Wilkins, Philadelphia
Grant D (1999) Intestinal transplantation: 1997 report of the international registry. Intestinal transplant registry. Transplantation 67:1061–1064
Abu-Elmagd K, Reyes J, Todo S et al (1998) Clinical intestinal transplantation: new perspectives and immunologic considerations. J Am Coll Surg 186:512–525, discussion 525–527
Abu-Elmagd KM, Mazariegos G, Costa G et al (2009) Lymphoproliferative disorders and de novo malignancies in intestinal and multivisceral recipients: improved outcomes with new outlooks. Transplantation 88:926–934
Purgina B, Rao UN, Miettinen M et al (2011) AIDS-related EBV-associated smooth muscle tumors: a review of 64 published cases. Pathol Res Int 2011:561548
Shaw RK, Issekutz AC, Fraser R et al (2012) Bilateral adrenal EBV-associated smooth muscle tumors in a child with a natural killer cell deficiency. Blood 119:4009–4012
Tan CS, Loh HL, Foo MW et al (2013) Epstein–Barr virus-associated smooth muscle tumors after kidney transplantation: treatment and outcomes in a single center. Clin Transpl 27:E462–E468
Salamanca J, Massa DS (2009) EBV-associated hepatic smooth muscle tumor after lung transplantation: report of a case and review of the literature. J Heart Lung Transplant 28:1217–1220
Brichard B, Smets F, Sokal E et al (2001) Unusual evolution of an Epstein–Barr virus-associated leiomyosarcoma occurring after liver transplantation. Pediatr Transplant 5:365–369
Boudjemaa S, Boman F, Guigonis V et al (2004) Brain involvement in multicentric Epstein–Barr virus-associated smooth muscle tumours in a child after kidney transplantation. Virchows Arch 444:387–391
Bianchi E, Pascual M, Nicod M et al (2008) Clinical usefulness of FDG-PET/CT scan imaging in the management of posttransplant lymphoproliferative disease. Transplantation 85:707–712
Takehana CS, Twist CJ, Mosci C et al (2014) (18)F-FDG PET/CT in the management of patients with post-transplant lymphoproliferative disorder. Nucl Med Commun 35:276–281
Von Falck C, Maecker B, Schirg E et al (2007) Post transplant lymphoproliferative disease in pediatric solid organ transplant patients: a possible role for [18 F]-FDG-PET(/CT) in initial staging and therapy monitoring. Eur J Radiol 63:427–435
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hryhorczuk, A.L., Kim, H.B., Harris, M.H. et al. Imaging findings in children with proliferative disorders following multivisceral transplantation. Pediatr Radiol 45, 1138–1145 (2015). https://doi.org/10.1007/s00247-015-3303-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-015-3303-2