Abstract
Background
Granulocyte colony-stimulating factors (G-CSF) speed recovery from chemotherapy-induced myelosuppression but the marrow stimulation they cause can interfere with interpretation of F-18 fluorodeoxyglucose positron emission tomography (F-18 FDG PET) exams.
Objective
To assess the frequency of interfering G-CSF-induced bone marrow activity on FDG PET imaging in children and young adults with Ewing sarcoma and rhabdomyosarcoma and to define an interval between G-CSF administration and FDG PET imaging that limits marrow interference.
Materials and methods
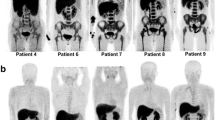
Blinded, retrospective review of FDG PET exams performed in patients treated with long-acting G-CSF as part of their chemotherapeutic regimen. Exams were subjectively scored by two reviewers (R1 and R2) who assessed the level of marrow uptake of FDG and measured standardized uptake values in the marrow, liver, spleen and blood pool. FDG PET findings were correlated with time since G-CSF administration and with blood cell counts.
Results
Thirty-eight FDG PET exams performed in 17 patients were reviewed with 47.4% (18/38) of exams having marrow uptake of FDG sufficient to interfere with image interpretation. Primary predictors of marrow uptake of FDG were patient age (P = 0.0037) and time since G-CSF exposure (P = 0.0028 for subjective marrow uptake of FDG, P = 0.008 [R1] and P = 0.004 [R2] for measured maximum standardized uptake value (SUVmax)). The median interval between G-CSF administration and PET imaging in cases with marrow activity considered normal or not likely to interfere was 19.5 days (range: 7–55 days).
Conclusion
In pediatric and young adult patients with Ewing sarcoma and rhabdomyosarcoma, an interval of 20 days between administration of the long-acting form of G-CSF and FDG PET imaging should limit interference by stimulated marrow.



Similar content being viewed by others
References
Hollingshead LM, Goa KL (1991) Recombinant granulocyte colony-stimulating factor (rG-CSF). A review of its pharmacological properties and prospective role in neutropenic conditions. Drugs 42:300–330
Whelan J, Khan A, Sharma A et al (2012) Interval compressed vincristine, doxorubicin, cyclophosphamide alternating with ifosfamide, etoposide in patients with advanced Ewing’s and other small round cell sarcomas. Clin Sarcoma Res 2:12
Womer RB, West DC, Krailo MD et al (2012) Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: a report from the Children’s Oncology Group. J Clin Oncol 30:4148–4154
Yao WJ, Hoh CK, Hawkins RA et al (1995) Quantitative PET imaging of bone marrow glucose metabolic response to hematopoietic cytokines. J Nucl Med 36:794–799
Fan C, Hernandez-Pampaloni M, Houseni M et al (2007) Age-related changes in the metabolic activity and distribution of the red marrow as demonstrated by 2-deoxy-2-[F-18] fluoro-D-glucose-positron emission tomography. Mol Imaging Biol 9:300–307
Taccone A, Oddone M, Dell’Acqua AD et al (1995) MRI “road-map” of normal age-related bone marrow. II. Thorax, pelvis and extremities. Pediatr Radiol 25:596–606
Taccone A, Oddone M, Occhi M et al (1995) MRI “road-map” of normal age-related bone marrow. I. Cranial bone and spine. Pediatr Radiol 25:588–595
Johnston KL, Farnen JP, Manske BR et al (2005) Abnormal positron emission tomography (PET) scan secondary to the use of hematopoietic growth factors. Haematologica 90:EIM03
Hollinger EF, Alibazoglu H, Ali A et al (1998) Hematopoietic cytokine-mediated FDG uptake simulates the appearance of diffuse metastatic disease on whole-body PET imaging. Clin Nucl Med 23:93–98
Jacene HA, Ishimori T, Engles JM et al (2006) Effects of pegfilgrastim on normal biodistribution of 18 F-FDG: preclinical and clinical studies. J Nucl Med 47:950–956
Filgrastim prescribing information. Amgen Inc., Thousand Oaks, CA. Available via http://www.neupogen.com. Accessed 06 Nov 2014
Pegfilgrastim prescribing information. Amgen Inc., Thousand Oaks, CA. Available via http://www.neulastahcp.com/characteristics/dosing-and-administration. Accessed 06 Nov 2014
Sugawara Y, Zasadny KR, Kison PV et al (1999) Splenic fluorodeoxyglucose uptake increased by granulocyte colony-stimulating factor therapy: PET imaging results. J Nucl Med 40:1456–1462
Hanaoka K, Hosono M, Usami K et al (2011) Fluorodeoxyglucose uptake in the bone marrow after granulocyte colony-stimulating factor administration in patients with non-Hodgkin’s lymphoma. Nucl Med Commun 32:678–683
Sugawara Y, Fisher SJ, Zasadny KR et al (1998) Preclinical and clinical studies of bone marrow uptake of fluorine-1-fluorodeoxyglucose with or without granulocyte colony-stimulating factor during chemotherapy. J Clin Oncol 16:173–180
Kazama T, Swanston N, Podoloff DA et al (2005) Effect of colony-stimulating factor and conventional- or high-dose chemotherapy on FDG uptake in bone marrow. Eur J Nucl Med Mol Imaging 32:1406–1411
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trout, A.T., Sharp, S.E., Turpin, B.K. et al. Optimizing the interval between G-CSF therapy and F-18 FDG PET imaging in children and young adults receiving chemotherapy for sarcoma. Pediatr Radiol 45, 1001–1006 (2015). https://doi.org/10.1007/s00247-014-3273-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-014-3273-9