Abstract
Background
Cavitating pulmonary tuberculosis (PTB) is generally known as a disease of adults, with children typically having features of primary PTB.
Objective
To group children with PTB and cavities according to possible pathogenesis by evaluating the clinical and radiological findings.
Materials and methods
The clinical and radiological findings in ten randomly selected children with PTB and cavitations on chest radiographs were retrospectively reviewed and evaluated.
Results
Three groups emerged: group 1 (four children) had cavities, usually single and unilateral in the classic upper lobe distribution of postprimary PTB; group 2 (three children) developed progressive primary spread of disease with extensive and bilateral pulmonary cavities; and group 3 (three children) developed cavities secondary to airway obstruction by mediastinal lymph nodes with consequent distal collapse and consolidation. Children in group 1 responded well to treatment and had unremarkable recoveries. Children in group 2 were all below 2 years of age with complicated recoveries. Children in group 3 had frequent complications resulting in one fatality.
Conclusion
Cavities in PTB in children may arise by one of three possible mechanisms with a relatively equal incidence. A study is underway to determine the incidence of cavity formation associated with mediastinal lymphadenopathy and airway obstruction.
Similar content being viewed by others
Introduction
While intrathoracic lymphadenopathy is the commonest radiographic feature of primary pulmonary tuberculosis (PTB) in children, parenchymal abnormalities do occur, the commonest findings being alveolar consolidation and linear interstitial opacification [1]. Primary PTB, the most common form in childhood, is radiologically distinct from postprimary TB, the most common form occurring in adults [2]. In primary pulmonary infection, a Ghon complex is formed with a primary parenchymal focus (Ghon focus) and lymphadenopathy occurs in the draining regional lymph nodes [3].
The reported incidence of lymphadenopathy seen on chest radiographs is very variable, ranging from 63% to 95% in different studies [4]. Lymphadenopathy occurs most frequently in the right hilar and right paratracheal positions, which represent the draining nodes of the lower and upper lobes of the right lung [4]. In 5–10% of children with primary PTB, the primary focus can enlarge and undergo caseous necrosis; this is called progressive primary PTB [5]. This can lead to endobronchial spread and miliary dissemination. The lymph nodes themselves may undergo disease progression, enlarging due to central caseation, and the result is lymphobronchial TB [3]. The airways may become obstructed with distal collapse/consolidation [3] and subsequent breakdown. They may also erode into the airways and cause intrabronchial spread with distal alveolar or bronchopneumonic consolidation [3].
In adults, cavitation results from reactivation of a dormant focus, the degree of tissue destruction being proportional to the severity of the hypersensitivity response [6].
Children have also been shown to develop adult-type postprimary PTB, with the characteristic distribution of cavities in the upper lobes and apices of the lower lobes of the lung [5]. This is rarely seen in prepubescent children [4]. The incidence of cavitation on chest radiography is between 5% and 16%; however it is frequently missed on plain radiographs alone, with CT demonstrating areas of cavitation or breakdown not seen on radiographs [4].
Understanding the pathological progression of the disease is essential in understanding the pathogenesis of the cavities, which may occur in a variety of clinical settings. We evaluated the clinical and radiological findings on plain chest radiographs of ten children found to have parenchymal cavitations as a result of PTB. In describing the patterns of cavitating lesions and associated chest radiograph findings and correlating with the clinical findings, we sought to group the lesions according to possible pathogenesis.
Materials and methods
Ten children with cavitating PTB were randomly chosen from children encountered in the radiology department of a tertiary hospital in a region where the incidence of TB is high. The children were all under the age of 16 years. The clinical picture and chest radiographic findings were documented and tabulated. This included initial and follow-up plain chest radiographs, if available, as well as contrast-enhanced CT chest scan if performed. Each image was evaluated for multiplicity, mural thickness and distribution of cavities, and the presence of associated findings, such as associated airspace disease, lymphadenopathy, with or without airway obstruction, and the presence of complications.
Results
The ten children included six girls and four boys; their ages ranged from 3 months to 15 years. Of the ten children, seven were culture-positive for Mycobacterium tuberculosis on gastric washings, sputum culture or lymph node aspiration; two were found to have multidrug-resistant PTB. In the other three children the diagnosis was based on a combination of the presence of constitutional symptoms, a positive tuberculin skin test (Mantoux or Tine test) and a positive TB contact according to National Tuberculosis Programme (NTP) guidelines [7]. Seven children had an HIV test, two of which were positive and five negative. The remaining three were not tested for HIV.
The children were divided into three groups on the basis of chest radiographic and CT findings. Their demographic data are summarized in Table 1, and their imaging findings are presented in Table 2.
Three children required ICU admission for respiratory complications and two subsequently died.
Only five of the ten children had CT scans performed; these were those whose clinical course was complicated and for whom surgical intervention might be required. The rest had an unremarkable recovery or were too unwell to undergo scanning.
Group 1
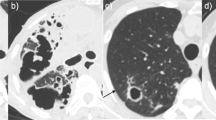
These four children were initially symptomatic, but responded rapidly to treatment. CT was performed in one child only. The cavities, visible on plain chest radiographs in all children, were thin-walled, mostly single and unilateral with half having associated airspace disease and half not. Their course was unremarkable and most were managed on an outpatient basis. There were no visible lymph nodes on plain radiographs (Fig. 1); however, on CT scans one child did have lymphadenopathy not visible on plain radiographs.
A 6-year-old girl (CP) with constitutional symptoms and a positive tuberculin skin test. a Chest radiograph shows a thin-walled cavity in the right mid-zone. b CT (lung windows) demonstrates cavitation of the lung parenchyma posteriorly at the level of the right hilum. Note also the bulky right pulmonary hilum in keeping with hilar lymphadenopathy
Group 2
These three children were below 2 years of age and were unwell. They presented with multiple, bilateral thin- or thick-walled cavities. Two of these children had CT scans; the third child died from respiratory failure before a scan could be performed (Fig. 2). On CT scans, lymph nodes were present in both children, but there was no associated obstruction. In two of the children there was also associated airspace disease (Fig. 3); in the third the lung destruction was so extensive that there was little remaining parenchyma.
A 4-month-old girl (LM) with worsening cough, dyspnoea and fever for 3 weeks was intubated and on intermittent positive pressure ventilation. The chest radiograph demonstrates bilateral, diffuse, air-filled cystic spaces of various sizes and large lung volumes. The child subsequently died and a post-mortem examination confirmed the diagnosis of PTB
A 2-year-old HIV-negative girl (CP) with severe adenopathy, clubbing and malnutrition and a positive TB contact. a The initial chest radiograph demonstrates bilateral airspace disease with areas of confluence and some early breakdown. b A follow-up chest radiograph shows progression of disease with bilateral cyst development; a large thin-walled cavity is identified in the right upper zone. c, d CT scans show multiple, bilateral thin-walled cavities of various sizes, some unicameral and others multicameral
Group 3
These three children were also unwell, and one child in this group died after a long and complicated course on a ventilator in intensive care. They developed cavitations as a consequence of lymphadenopathy causing airway obstruction with caseous liquefaction of the lung distal to the obstruction. Two of the three children had CT scans in view of the complications or for surgical planning (Fig. 4). In both these children, the adenopathy was visible on plain chest radiographs. One of the children was less symptomatic and while she had multiple bilateral cysts, no CT was done and lymph nodes were inferred from the presence of obvious bronchial obstruction on plain radiographs. She may well represent a child who lies somewhere between the first and the third group.
A 5-year-old HIV-positive child (TL) not receiving antiretroviral therapy. a The chest radiograph demonstrates dense airspace consolidation with breakdown in the left lower zone. b A decubitus view confirms an air-fluid level within a large thick-walled cavity. c CT demonstrates breakdown in an area of dense consolidation in the lingular region secondary to airway obstruction. Low-density rim-enhancing nodes (arrow) are seen at the left hilum
Discussion
In Cape Town, Western Cape, South Africa, a high TB burden area, children under 13 years of age contribute 13.7% to the total TB burden with the incidence of TB in children at 441/100,000/year [8]. The children in this study were all treated at the tertiary level after being referred from smaller clinics in this catchment area.
In children the chest radiograph is of major importance in early diagnosis as bacteriology is hampered by the difficulty in obtaining suitable sputum specimens [1]. Diagnosis is usually made and treatment instigated on the basis of high clinical suspicion especially in children who have been exposed to an adult with PTB. A tuberculin skin test and chest radiograph are routinely done and treatment started as per NTP guidelines [7]. Seven of the ten children in our series had the diagnosis of tuberculosis confirmed, with sputum or gastric washings either positive for acid-fast bacilli (AFB) or culture-positive for Mycobacterium tuberculosis.
HIV-positive patients appear to have an eight-times higher risk of infection by Mycobacterium tuberculosis. Such children acquire primary TB and then go on to develop a rapid and fulminating form of the disease [4]. They are also prone to develop other mimickers of PTB, namely lymphoid interstitial pneumonia and opportunistic infections such as Pneumocystis jiroveci pneumonia (PCP), viral and atypical Mycobacterium pneumonias. This has led to the over-diagnosis of PTB in HIV-positive children with 40% of HIV-positive children treated for the disease being incorrectly diagnosed [8].
Lymphadenopathy is the commonest single manifestation of PTB in children [1] and, with or without concomitant parenchymal abnormality, is the radiological hallmark of primary TB in children [2]. Cavitations are reported in children; however, the aetiology and pathogenesis is somewhat more complex than in adults. In a study done of 80 children with PTB, cavitation was found in 7 (9%) on initial chest radiographs [9]. CT is more sensitive in demonstrating any cavitation that may occur [10]; our children were recruited on the basis of cavitation on plain radiographs.
There are three groups into which children with cavitation due to PTB can be placed. These relate to the clinical presentation, radiological characteristics and distribution of the cavities, and associated findings, for example mediastinal lymph nodes.
Group 1
The adult form of PTB does occur in children, but prior to 14 years of age, primary PTB is more likely to be seen, and thereafter cavitation becomes progressively more common [11]. In our study, three children in this group were older than 10 years of age. Cavitations were single or multiple, almost always unilateral and often associated with airspace disease (three out of four).
Postprimary PTB results from reactivation of a dormant focus with cavitation being the radiographic hallmark [5]. Evidence of previous primary PTB is frequently seen on chest radiographs in children [5]. The cavities usually already exist on presentation, surrounded by alveolar consolidation and fibrosis [3]. The common sites are apical and posterior segments of the upper lobe and apical segments of the lower lobe [3].
Group 2
Why some children go on to develop progressive primary PTB may be related to the number and virulence of the organisms in the setting of a child with poor resistance to infection [12]. Poor containment of the primary infection is more common in children under the age of 2 years who are immunocompromised [3]. These children are usually very ill. The cavities develop as a result of enlargement of the Ghon focus with eventual caseous liquefaction at the centre of the focus. When this focus ruptures into an airway, cavitation occurs and endobronchial spread of disease results. Distal bronchopneumonic consolidation occurs.
Group 3
Newborn children up to 3 years of age have a higher prevalence of adenopathy and a lower prevalence of parenchymal abnormalities [2]. This, along with a small airway size makes young children most vulnerable to develop lymphobronchial TB [3]. Airway compromise due to extraluminal compression of an airway is a recognized complication of adenopathy in primary PTB. If the airway is only partially obstructed, a ball-valve effect may occur giving distal hyperinflation. However, should the airway be completely obstructed, distal air becomes resorbed and collapse occurs [3]. Destructive caseating pneumonia may result with bulging fissures and cavitation.
Lymph nodes are a frequent cause of complications as they erode into adjacent structures, for example, causing oesophageal perforation or bronchopleural fistula [4]. The cavities are thick-walled and often difficult to separate from surrounding consolidated lung on plain radiographs. CT confirmed the airway compromise and the associated thick-walled cavities were seen to enhance and contain air-fluid levels.
It is often technically difficult to see lymphadenopathy on plain radiographs and especially AP chest radiographs [3]. Lateral chest radiographs were not always available in our patients making the detection of nodes more difficult. The use of high-kVp radiography to demonstrate airway compression, or better still contrast-enhanced CT, is of great benefit in confirming the presence of adenopathy. CT scanning was only performed in two of the three children in the obstruction group. In both these children, however, the nodes were visible on plain radiographs.
Among children who present with expansile pneumonias caused by Mycobacterium tuberculosis, 83% have significant airway obstruction (more than 75% occlusion) on bronchoscopy and all have enhancing mediastinal lymph nodes on CT. Cavities are present in 63% of children with expansile pneumonia [13].
Conclusion
Cavitation is a frequent finding on initial chest radiographs in children with PTB. There are three possible mechanisms involved in the formation of cavities: ‘adult-type’ post-primary PTB, progressive primary spread of disease, or cavities arising as a consequence of bronchial obstruction by lymph nodes. In our small sample there were equal numbers of children in all three groups. Some children do not quite fit the described groups and these children are probably those who present in the first group, i.e. well children who developed cavities as result of nodal obstruction or progressive primary disease and who subsequently recovered. Many variables, including the dose and the virulence of the organism, the age of the patient and, therefore, the predilection for certain sites of progression, and lastly the immune competence of the host all determine the presentation on chest radiographs. It was not possible to determine the role of immune status in cavity formation in this study as not all children were tested and the resistance of the organisms, whether they were multidrug-resistant (MDR) tuberculous bacilli, was not explored.
A larger study involving all children with PTB who have undergone CT scanning is underway and will better determine the true incidence of parenchymal cavitation and the association with the presence of mediastinal lymphadenopathy and airway obstruction.
References
Lamont AC, Cremin BJ, Pelteret RM (1986) Radiological patterns of pulmonary tuberculosis in the paediatric age group. Pediatr Radiol 16:2–7
Leung AN, Muller NL, Pineda PR et al (1992) Primary tuberculosis in childhood: radiographic manifestations. Radiology 182:87–91
Marais BJ, Gie RP, Schaaf HS et al (2004) A proposed radiological classification of childhood intra-thoracic tuberculosis. Pediatr Radiol 34:886–894
Cremin BJ, Jamieson DH (1995) Childhood tuberculosis: modern imaging and clinical concepts. Springer, Berlin, pp 11, 20, 26, 33, 37, 46, 109
Shewchuk JR, Reed MH (2002) Paediatric postprimary pulmonary tuberculosis. Pediatr Radiol 32:648–665
McAdams HP, Erasmus J, Winter JA (1995) Radiological manifestations of pulmonary tuberculosis. Radiol Clin North Am 33:655–678
South African Department of Health (2000) The S.A. tuberculosis control programme-practical guidelines. Pretoria, South Africa, pp 32–37 http://www.capegateway.gov.za/text/2003/tb_guidelines2000.pdf Cited 16/01/2007
Marais BJ, Hesseling AC, Gie RP et al (2006) The burden of childhood tuberculosis and the accuracy of community-based surveillance data. Int J Tuberc Lung Dis 10:259–263
Kisembo HN, Kawooya MG, Zirembu G et al (2001) Serial chest radiographs in the management of children with a clinical suspicion of pulmonary tuberculosis. J Trop Paediatr 47:276–283
Koh DM, Bell JR, Burkill GJ et al (2001) Mycobacterial infections: still a millennium bug – the imaging features of mycobacterial infections. Clin Radiol 56:535–544
Weber HC, Beyers N, Gie RP et al (2000) The clinical and radiological features of tuberculosis in adolescents. Ann Trop Paediatr 20:5–10
Agrons GA, Markowitz RI, Kramer SS (1993) Pulmonary tuberculosis in children. Semin Roentgenol 28:158–172
Goussard P, Gie RP, Kling S et al (2004) Expansile pneumonia in children caused by Mycobacterium tuberculosis: clinical, radiological and bronchoscopic appearances. Pediatr Pulmonol 38:451–455
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Griffith-Richards, S.B., Goussard, P., Andronikou, S. et al. Cavitating pulmonary tuberculosis in children: correlating radiology with pathogenesis. Pediatr Radiol 37, 798–804 (2007). https://doi.org/10.1007/s00247-007-0496-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-007-0496-z