Abstract
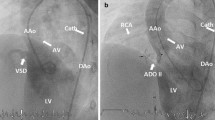
Untreated ventricular septal defect (VSD) is an important cause of congestive heart failure in early infancy. Growth is impaired in this population, and surgical closure is challenging because of congestion in the lungs, making infants prone to respiratory infection, and because of their poor nutritional status. The aim of this study is to share our experience with percutaneous VSD closure in patients under 1 year of age. Patients with hemodynamically significant left-to-right shunt, less than 1 year of age, and with VSD diameter ≤ 6 mm were retrospectively included in the study between December 2014 and January 2017. The median length of follow-up was 8.5 (4–14.2) months. Twelve patients from 2 to 12 months of age, with a median weight of 6.75 (5.4–8) kg, were included. The mean VSD diameter as measured by angiography from the left ventricle side was 4.7 ± 0.25 mm, and from the right ventricle side was 3.4 ± 1.1 mm. All were of a perimembranous type except three, which were muscular. All defects were closed with the Amplatzer Duct Occluder II (ADO II) or the ADO II-additional size. The mean fluoroscopy duration and total radiation dose were 22.6 ± 18.7 min and 1674 ± 851 cGy/min, respectively. No aortic regurgitation associated with device closure was seen in any of the patients. Complete atrioventricular block occurred in one patient 6 months after the procedure, and was treated with a permanent pacemaker. VSD closure is challenging, regardless of whether a surgical or percutaneous procedure is used. The risks are higher for children younger than 1 year with low body weight. Percutaneous closure, which carries similar risks but is less invasive than surgery, may be the preferred alternative in early infancy.

Similar content being viewed by others
References
Schipper M, Slieker MG, Schoof PH et al (2017) Surgical repair of ventricular septal defect; contemporary results and risk factors for a complicated course. Pediatr Cardiol 38:264–270
Anderson B, Stevens K, Nicolson S et al (2013) Contemporary outcomes of surgical ventricular septal defect closure. J Thorac Cardiovasc Surg 145:641–647
Wollenek G, Wyse R, Sullivan I et al (1996) Closure of muscular ventricular septal defects through a left ventriculotomy. Eur J Cardiothorac Surg 10:595–598
Kitagawa T, Durham LA III, Mosca RS et al (1998) Techniques and results in the management of multiple ventricular septal defects. J Thorac Cardiovasc Surg 115:848–856
Waight DJ, Bacha EA, Kahana M et al (2002) Catheter therapy of Swiss cheese ventricular septal defects using the Amplatzer muscular VSD occluder. Catheter Cardiovasc Interv 55:355–361
Hijazi ZM, Hakim F, Al-Fadley F et al (2000) Transcatheter closure of single muscular ventricular septal defects using the amplatzer muscular VSD occluder: initial results and surgical considerations. Catheter Cardiovasc Interv 49:167–172
Diab KA, Cao QL, Mora BN et al (2007) Device closure of muscular ventricular septal defects in infants less than one year of age using the Amplatzer devices: feasibility and outcome. Catheter Cardiovasc Interv 70:90–97
Zartner P, Christians C, Stelter JC et al (2014) Transvascular closure of single and multiple muscular ventricular septal defects in neonates and infants < 20 kg. Catheter Cardiovasc Interv 1:83:564–570
Warnes CA, Williams RG, Bashore TM et al (2008) ACC/AHA 2008 guidelines for the management of adults with congenital heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation 118:714–833
Baumgartner H, Bonhoeffer P, De Groot NM, Task Force on the Management of Grown-up Congenital Heart Disease of the European Society of Cardiology (ESC), Association for European Paediatric Cardiology (AEPC), ESC Committee for Practice Guidelines (CPG) et al (2010) ESC guidelines for the management of grown-up congenital heart disease (new version 2010). Eur Heart J 31:2915–2957
Butera G, Chessa M, Carminati M (2007) Percutaneous closure of ventricular septal defects. Cardiol Young 17:243–253
Pamukcu O, Narin N, Baykan A, Sunkak S, Tasci O, Uzum K (2017) Mid-term results of percutaneous ventricular septal defect closure with Amplatzer Duct Occluder-II in children. Cardiol Young. https://doi.org/10.1017/S104795111700107X
Butera G, Carminati M, Chessa M et al (2007) Transcatheter closure of perimembranous ventricular septal defects: early and long-term results. J Am Coll Cardiol 50:1189–1195
Holzer R, de Giovanni J, Walsh KP et al (2006) Transcatheter closure of perimembranous ventricular septal defects using the Amplatzer membranous VSD occluder: immediate and midterm results of an international registry. Catheter Cardiovasc Interv 68:620–628
Saurav A, Kaushik M, Mahesh Alla V et al (2015) Comparison of percutaneous device closure versus surgical closure of peri-membranous ventricular septal defects: a systematic review and meta-analysis. Catheter Cardiovasc Interv 86:1048–1056
Narin N, Baykan A, Pamukcu O et al (2015) ADO II in percutaneous VSD closure in pediatric patients. J Interv Cardiol 28:479–484
Freedman SB, Haladyn JK, Floh A et al (2007) Pediatric myocarditis: emergency department clinical findings and diagnostic evaluation. Pediatrics 120:1278–1285
Batra AS, Epstein D, Silka MJ (2003) The clinical course of acquired complete heart block in children with acute myocarditis. Pediatr Cardiol 24(5):495–497
Ichikawa R, Sumitomo N, Komori A, Abe Y, Nakamura T, Fukuhara J, Matsumura M, Miyashita M, Kanamaru H, Ayusawa M, Mugishima H (2011) The follow-up evaluation of electrocardiogram and arrhythmias in children with fulminant myocarditis. Circ J 75(4):932–938
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have a conflict of interest.
Rights and permissions
About this article
Cite this article
Narin, N., Pamukcu, O., Tuncay, A. et al. Percutaneous Ventricular Septal Defect Closure in Patients Under 1 Year of Age. Pediatr Cardiol 39, 1009–1015 (2018). https://doi.org/10.1007/s00246-018-1852-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-018-1852-5