Abstract
For many jellyfish, the magnitude and timing of medusae blooms are recognized to result from the benthic stage dynamics. However, information on the scyphistomae of jellyfish populations in the wild remains scarce. Here, bi-mensual underwater photoquadrat surveys were combined with scyphistomae sampling and observation to describe the annual (February 2017–January 2018) benthic stage dynamics and asexual reproduction strategy of Aurelia coerulea in the Thau lagoon (43°25′31.1″N; 03°42′0.9″E). Our results revealed unexpected seasonal patterns of variation: scyphistoma coverage peaked in the spring (11.6 ± 3.7% on 21st April) and was minimal in the summer and autumn (1.4 ± 1.3% on 10th October). The increase in scyphistoma coverage mainly resulted from an intense production of buds between February and April during the spring rise in water temperature (peak of 12,800 buds m−2 on 21st April), but scyphistoma coverage appeared to be negatively influenced by the interaction of high summer temperatures and salinities. Strobilation was observed from November to April. It peaked on 17th November, with 33.1% of the scyphistomae strobilating and an average production of 19,100 strobila disks m−2. However, the low scyphistoma coverage at this time of the year (< 2%) likely limited the intensity of ephyrae liberation and the subsequent medusae bloom. The final population size of A. coerulea thus results from a complex interaction of abiotic and biotic factors. Our results bring into question how the different populations of Aurelia spp. will respond to the predicted global warming scenarios.

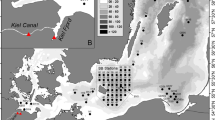
Source: IGN (Institut Geographique National) and IFREMER (Institut Français de Recherche pour l'exploitation de la Mer)






Similar content being viewed by others
References
Amorim K, Mattmüller RM, Algueró-Muñiz M, Meunier CL, Alvarez-Fernandez S, Boersma M, Morais P, Teodósio MA (2018) Winter river discharge may affect summer estuarine jellyfish blooms. Mar Ecol Prog Ser 591:253–265
Arai MN (2009) The potential importance of podocysts to the formation of scyphozoan blooms: a review. Hydrobiologia 616:241–246. https://doi.org/10.1007/s10750-008-9588-5
Boero F (2013) Review of jellyfish blooms in the Mediterranean and Black Sea. Studies and reviews. General fisheries commission for the Mediterranean. No 92. FAO, Rome
Bonnet D, Molinero J-C, Schohn T, Daly-Yahia MN (2012) Seasonal changes in the population dynamics of Aurelia aurita in Thau lagoon. Cah Biol Mar 53:343–347
Brotz L, Cheung WWL, Kleisner K, Pakhomov E, Pauly D (2012) Increasing jellyfish populations: trends in large marine ecosystems. Hydrobiologia 690:3–20. https://doi.org/10.1007/s10750-012-1039-7
Dawson MN, Jacobs DK (2001) Molecular evidence for cryptic species of Aurelia aurita (Cnidaria, Scyphozoa). Biol Bull 200:92–96. https://doi.org/10.3102/00346543067001043
Dawson MN, Martin DL (2001) Geographic variation and ecological adaptation in Aurelia aurita (Scyphozoa, Semaeostomeae): some implications from molecular phylogenetics. Hydrobiologia 451:259–273
Dawson MN, Sen Gupta A, England MH (2005) Coupled biophysical global ocean model and molecular genetic analyses identify multiple introductions of cryptogenic species. PNAS 102:11968–11973. https://doi.org/10.1073/pnas.0503811102
Dong Z, Sun T, Wang L (2018) The biogenic reefs formed by the alien polychaete Hydroides dianthus (Serpulidae, Annelida) favor the polyp stage of Aurelia coerulea (Cnidaria, Scyphozoa) in a coastal artificial lake. Mar Pollut Bull 129:86–91. https://doi.org/10.1016/j.marpolbul.2018.02.016
Dray S, Dufour A (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20. https://doi.org/10.18637/jss.v022.i04
Feng S, Wang SW, Zhang GT, Sun S, Zhang F (2017) Selective suppression of in situ proliferation of scyphozoan polyps by biofouling. Mar Pollut Bull 114:1046–1056. https://doi.org/10.1016/j.marpolbul.2016.10.062
Feng S, Wang SW, Sun S, Zhang F, Zhang GT, Liu MT, Uye SI (2018) Strobilation of three scyphozoans (Aurelia coelurea, Nemopilema nomurai, and Rhopilema esculentum) in the field at Jiaozhou Bay, China. Mar Ecol Prog Ser 591:141–153. https://doi.org/10.3354/meps12276
Fu Z, Shibata M, Makabe R, Ikeda H, Uye SI (2014) Body size reduction under starvation, and the point of no return, in ephyrae of the moon jellyfish Aurelia aurita. Mar Ecol Prog Ser 510:255–263. https://doi.org/10.3354/meps10799
Graham WM, Gelcich S, Robinson KL, Duarte CM, Brotz L, Purcell JE, Madin LP, Mianzan H, Sutherland KR, Uye SI, Pitt KA, Lucas CH, Bøgeberg M, Brodeur RD, Condon RH (2014) Linking human well-being and jellyfish: ecosystem services, impacts, and societal responses. Front Ecol Environ 12:515–523. https://doi.org/10.1890/130298
Gröndahl F (1988) A comparative ecological study on the scyphozoans Aurelia aurita, Cyanea capillata and C. lamarckii in the Gullmar Fjord, western Sweden, 1982 to 1986. Mar Biol 97:541–550. https://doi.org/10.1007/BF00391050
Han C-H, Uye S-I (2010) Combined effects of food supply and temperature on asexual reproduction and somatic growth of polyps of the common jellyfish Aurelia aurita s.l. Plankton Benthos Res 5:98–105. https://doi.org/10.3800/pbr.5.98
Harrison XA, Donaldson L, Correa-cano ME, Evans J, Fisher DN, Goodwin CED, Robinson BS, Hodgson DJ, Inger R (2018) A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 6(e4794):1–32. https://doi.org/10.7717/peerj.4794
Hocevar S, Malej A, Boldin B, Purcell JE (2018) Seasonal fluctuations in population dynamics of Aurelia aurita polyps in situ with a modelling perspective. Mar Ecol Prog Ser 591:155–166. https://doi.org/10.3354/meps12387
Holst S (2012) Effects of climate warming on strobilation and ephyra production of North Sea scyphozoan jellyfish. Hydrobiologia 690:127–140. https://doi.org/10.1007/s10750-012-1043-y
Holst S, Jarms G (2007) Substrate choice and settlement preferences of planula larvae of five Scyphozoa (Cnidaria) from German Bight, North Sea. Mar Biol 151:863–871. https://doi.org/10.1007/s00227-006-0530-y
Holst S, Jarms G (2010) Effects of low salinity on settlement and strobilation of Scyphozoa (Cnidaria): is the lion’s mane Cyanea capillata (L.) able to reproduce in the brackish Baltic Sea? Hydrobiologia 645:53–68. https://doi.org/10.1007/s10750-010-0214-y
Hoover RA, Purcell JE (2009) Substrate preferences of scyphozoan Aurelia labiata polyps among common dock-building materials. Hydrobiologia 616:259–267. https://doi.org/10.1007/s10750-008-9595-6
Hoover RA, Armour R, Dow I, Purcell JE (2012) Nudibranch predation and dietary preference for the polyps of Aurelia labiata (Cnidaria: Scyphozoa). Hydrobiologia 690:199–213. https://doi.org/10.1007/s10750-012-1044-x
Hubot N, Lucas CH, Piraino S (2017) Environmental control of asexual reproduction and somatic growth of Aurelia spp. (Cnidaria, Scyphozoa) polyps from the Adriatic Sea. PLoS One. https://doi.org/10.1371/journal.pone.0178482
Ikeda H, Mizota C, Uye S (2017) Bioenergetic characterization in Aurelia aurita (Cnidaria: Scyphozoa) polyps and application to natural polyp populations. Mar Ecol Prog Ser 568:87–100
IPCC (2014) Climate change 2014 synthesis report. Contribution of working groups I, II and III to the fifth assessment report of the intergovernmental panel on climate change. IPCC, Geneva
Ishii H, Katsukoshi K (2010) Seasonal and vertical distribution of Aurelia aurita polyps on a pylon in the innermost part of Tokyo Bay. J Oceanogr 66:329–336. https://doi.org/10.1007/s10872-010-0029-5
Kamiyama T (2011) Planktonic ciliates as a food source for the scyphozoan Aurelia aurita (s.l.): feeding activity and assimilation of the polyp stage. J Exp Mar Biol Ecol 407:207–215. https://doi.org/10.1016/j.jembe.2011.06.016
Kamiyama T (2013) Planktonic ciliates as food for the scyphozoan Aurelia aurita (s.l.): effects on asexual reproduction of the polyp stage. J Exp Mar Biol Ecol 445:21–28. https://doi.org/10.1016/j.jembe.2013.03.018
Kawahara M, Ohtsu K, Uye SI (2013) Bloom or non-bloom in the giant jellyfish Nemopilema nomurai (Scyphozoa: Rhizostomeae): roles of dormant podocysts. J Plankton Res 35:213–217. https://doi.org/10.1093/plankt/fbs074
Lucas CH, Graham WM, Widmer C (2012) Jellyfish life histories: role of polyps in forming and maintaining scyphomedusa populations, 1st edn. Elsevier Ltd, Amsterdam
Makabe R, Furukawa R, Takao M, Uye SI (2014) Marine artificial structures as amplifiers of Aurelia aurita s.l. blooms: a case study of a newly installed floating pier. J Oceanogr 70:447–455. https://doi.org/10.1007/s10872-014-0249-1
Malej A, Kogovšek T, Ramšak A, Catenacci L (2012) Blooms and population dynamics of moon jellyfish in the northern Adriatic. Cah Biol Mar 53:337–342
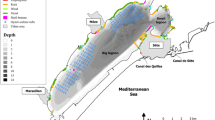
Marques R, Cantou M, Soriano S, Molinero JC, Bonnet D (2015a) Mapping distribution and habitats of Aurelia sp. Polyps in Thau lagoon, north-western Mediterranean sea (France). Mar Biol 162:1441–1449. https://doi.org/10.1007/s00227-015-2680-2
Marques R, Albouy-Boyer S, Delpy F, Carré C, Le Floc’H É, Roques C, Molinero JC, Bonnet D (2015b) Pelagic population dynamics of Aurelia sp. in French Mediterranean lagoons. J Plankton Res 37:1019–1035. https://doi.org/10.1093/plankt/fbv059
Marques R, Bouvier C, Darnaude AM, Molinero J-C, Przybyla C, Soriano S, Tomasini J-A, Bonnet D (2016) Jellyfish as an alternative source of food for opportunistic fishes. J Exp Mar Biol Ecol. https://doi.org/10.1016/j.jembe.2016.08.008
Melica V, Invernizzi S, Caristi G (2014) Logistic density-dependent growth of an Aurelia aurita polyps population. Ecol Model 291:1–5. https://doi.org/10.1016/j.ecolmodel.2014.07.009
Mills C (2001) Are population increasing globally in response to changing ocean conditions? Hydrobiologia 451:55–68
Miyake H, Terazaki M, Kakinuma Y (2002) On the polyps of the common jellyfish Aurelia aurita in Kagoshima Bay. J Oceanogr 58:451–459. https://doi.org/10.1023/A:1021628314041
Östman C (1997) Abundance, feeding behaviour and nematocysts of scyphopolyps (Cnidaria) and nematocysts in their predator, the nudibranch Coryphella verrucosa (Mollusca). In: Naumov AD, Hummel H, Sukhotin AA, Ryland JS (eds) Interactions and adaptation strategies of marine organisms. Developments in hydrobiology, vol 121. Springer, Dordrecht
Purcell JE (2005) Climate effects on formation of jellyfish and ctenophore blooms: a review. J Mar Biol Assoc UK 85:461–476. https://doi.org/10.1017/S0025315405011409
Purcell JE (2007) Environmental effects on asexual reproduction rates of the scyphozoan Aurelia labiata. Mar Ecol Prog Ser 348:183–196. https://doi.org/10.3354/meps07056
Purcell JE (2012) Jellyfish and ctenophore blooms coincide with human proliferations and environmental perturbations. Ann Rev Mar Sci 4:209–235. https://doi.org/10.1146/annurev-marine-120709-142751
Purcell JE, Uye SI, Lo WT (2007) Anthropogenic causes of jellyfish blooms and their direct consequences for humans: a review. Mar Ecol Prog Ser 350:153–174. https://doi.org/10.3354/meps07093
Purcell JE, Hoover RA, Schwarck NT (2009) Interannual variation of strobilation by the scyphozoan Aurelia labiata in relation to polyp density, temperature, salinity, and light conditions in situ. Mar Ecol Prog Ser 375:139–149. https://doi.org/10.3354/meps07785
Purcell JE, Atienza D, Fuentes V, Olariaga A, Tilves U, Colahan C, Gili JM (2012) Temperature effects on asexual reproduction rates of scyphozoan species from the northwest Mediterranean Sea. Hydrobiologia 690:169–180. https://doi.org/10.1007/s10750-012-1047-7
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Richardson AJ, Bakun A, Hays GC, Gibbons MJ (2009) The jellyfish joyride: causes, consequences and management responses to a more gelatinous future. Trends Ecol Evol 24:312–322. https://doi.org/10.1016/j.tree.2009.01.010
Schiariti A, Morandini AC, Jarms G, Von Glehn Paes R, Franke S, Mianzan H (2014) Asexual reproduction strategies and blooming potential in Scyphozoa. Mar Ecol Prog Ser 510:241–253. https://doi.org/10.3354/meps10798
Schiariti A, Melica V, Kogovšek T, Malej A (2015) Density-dependent effects control the reproductive strategy and population growth of Aurelia aurita s.l. scyphistomae. Mar Biol 162:1665–1672. https://doi.org/10.1007/s00227-015-2704-y
Scorrano S, Aglieri G, Boero F, Dawson MN, Piraino S (2016) Unmasking Aurelia species in the Mediterranean Sea: an integrative morphometric and molecular approach. Zool J Linn Soc. https://doi.org/10.1111/zoj.12494
Sokołowski A, Brulińska D, Olenycz M, Wołowicz M (2016) Does temperature and salinity limit asexual reproduction of Aurelia aurita polyps (Cnidaria: Scyphozoa) in the Gulf of Gdańsk (southern Baltic Sea)? An experimental study. Hydrobiologia 773:49–62. https://doi.org/10.1007/s10750-016-2678-x
Takao M, Okawachi H, Uye S (2014) Natural predators of polyps of Aurelia aurita s.l. (Cnidaria: Scyphozoa: Semaeostomeae) and their predation rates. Plankton Benthos Res 9:105–113. https://doi.org/10.3800/pbr.9.105
Thein H, Ikeda H, Uye SI (2012) The potential role of podocysts in perpetuation of the common jellyfish Aurelia aurita s.l. (Cnidaria: Scyphozoa) in anthropogenically perturbed coastal waters. Hydrobiologia 690:157–167. https://doi.org/10.1007/s10750-012-1045-9
Toyokawa M, Furota T, Terazaki M (2000) Life history and seasonal abundance of Aurelia aurita medusae in Tokyo Bay, Japan. Plankton Biol Ecol 47:48–58
Toyokawa M, Aoki K, Yamada S, Yasuda A, Murata Y, Kikuchi T (2011) Distribution of ephyrae and polyps of jellyfish Aurelia aurita (Linnaeus 1758) sensu lato in Mikawa Bay, Japan. J Oceanogr 67:209–218. https://doi.org/10.1007/s10872-011-0021-8
Uye S, Shimauchi H (2005) Population biomass, feeding, respiration and growth rates, and carbon budget of the scyphomedusa Aurelia aurita in the Inland Sea of Japan. J Plankton Res 27:237–248. https://doi.org/10.1093/plankt/fbh172
Watanabe T, Ishii H (2001) In situ estimation of ephyrae liberated from polyps of Aurelia aurita using settling plates in Tokyo Bay, Japan. Hydrobiologia 451:247–258. https://doi.org/10.1023/A:1011856929443
Widmer CL, Fox CJ, Brierley AS (2016) Effects of temperature and salinity on four species of northeastern Atlantic scyphistomae (Cnidaria: Scyphozoa). Mar Ecol Prog Ser 559:73–88. https://doi.org/10.3354/meps11879
Willcox S, Moltschaniwskyj NA, Crawford C (2007) Asexual reproduction in scyphistomae of Aurelia sp.: effects of temperature and salinity in an experimental study. J Exp Mar Biol Ecol 353:107–114. https://doi.org/10.1016/j.jembe.2007.09.006
Willcox S, Moltschaniwskyj NA, Crawford CM (2008) Population dynamics of natural colonies of Aurelia sp. scyphistomae in Tasmania, Australia. Mar Biol 154:661–670. https://doi.org/10.1007/s00227-008-0959-2
Zeileis A, Kleiber C, Jackman S (2008) Regression models for count data in R. J Stat Softw 27:1–25. https://doi.org/10.18637/jss.v027.i08
Acknowledgements
We thank Nicholas Nouguier and Remy Valdes for their technical support and expertise during SCUBA dives and field work. Chlorophyll a and mesozooplankton data are part of a long-term monitoring programme on microbial communities in Thau lagoon funded by Observatoire des Sciences de l’Univers OREME (OSU-OREME). Data are available on (http://www.oreme.univ-montp2.fr/). We also thank the reviewers and the handling editor for valuable comments on this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The jellyfish Aurelia coerulea is an invertebrate which is not endangered or protected species and therefore, no permits were needed for sampling their scyphistomae. All bivalves with attached scyphistomae were released alive after laboratory observation.
Additional information
Responsible Editor: J. Purcell.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by S. Hocevar Ch. Widmer and an undisclosed expert.
Rights and permissions
About this article
Cite this article
Marques, R., Darnaude, A.M., Schiariti, A. et al. Dynamics and asexual reproduction of the jellyfish Aurelia coerulea benthic life stage in the Thau lagoon (northwestern Mediterranean). Mar Biol 166, 74 (2019). https://doi.org/10.1007/s00227-019-3522-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-019-3522-4