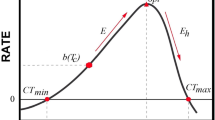
Abstract
Temperature is one of the more important factors affecting metabolism and fitness of aquatic ectothermic animals. The present study aimed, therefore, at quantifying and comparing the oxygen consumption rates of several subtropical macrobenthos organisms living in two South African estuarine systems. Oxygen fluxes were measured in the laboratory at an individual level for temperatures ranging between 16 and 30 °C. The metabolic response differed significantly among taxa, developmental stage, and microhabitat, with higher activation energy for the animals from the permanently open estuary compared to the one from the temporarily open/closed system. The invasive snails showed an exceptional higher metabolic performance at higher temperatures compared to the other organisms collected within the temporarily open/closed system. The results suggest that estuaries experiencing prolonged closure with stable environmental conditions and lower biodiversity host the least performing native species. Setting this pattern in a future global change scenario, this study highlights that longer closures of the estuarine mouth caused by changing rainfall regimes and increased freshwater abstractions from catchments are likely to create the ideal conditions for the proliferation of more adaptable invasive species in temporally open/closed subtropical estuaries.






Similar content being viewed by others
References
Allanson B, Baird D (2008) Estuaries of South Africa. Cambridge University Press, New York
Angilletta MJ, Niewiarowski PH, Navas CA (2002) The evolution of thermal physiology in ectotherms. J Therm Biol 27:249–268. https://doi.org/10.1016/S0306-4565(01)00094-8
Angilletta MJ, Huey RB, Frazier MR (2010) Thermodynamic effects on organismal performance: is hotter better? Physiol Biochem Zool Ecol Evol Approaches 83:197–206
Appleton CC, Forbes AT, Demetriades NT (2009) The occurrence, bionomics and potential impacts of the invasive freshwater snail Tarebia granifera (Lamarck, 1822)(Gastropoda: Thiaridae) in South Africa. Zool Med Leiden 83:525–536
Arrhenius S (1889) Über die Dissociationswärme und den Einfluss der Temperatur auf den Dissociationsgrad der Elektrolyte. Z Für Phys Chem 4:226–248. https://doi.org/10.1515/zpch-1889-0108
Baird D, Milne H (1981) Energy flow in the Ythan estuary, Aberdeenshire, Scotland. Estuar Coast Shelf Sci 13:455–472. https://doi.org/10.1016/S0302-3524(81)80041-2
Baldanzi S, Weidberg NF, Fusi M, Cannicci S, McQuaid CD, Porri F (2015) Contrasting environments shape thermal physiology across the spatial range of the sandhopper Talorchestia capensis. Oecologia 179:1067–1078. https://doi.org/10.1007/s00442-015-3404-5
Brey T (2010) An empirical model for estimating aquatic invertebrate respiration. Methods Ecol Evol 1:92–101. https://doi.org/10.1111/j.2041-210X.2009.00008.x
Brodersen KP, Pedersen O, Walker IR, Jensen MT (2008) Respiration of midges (Diptera; Chironomidae) in British Columbian lakes: oxy-regulation, temperature and their role as palaeo-indicators. Freshw Biol 53:593–602. https://doi.org/10.1111/j.1365-2427.2007.01922.x
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789. https://doi.org/10.1890/03-9000
Bucher D, Saenger P (1994) A classification of tropical and subtropical Australian estuaries. Aquat Conserv Mar Freshw Ecosyst 4:1–19. https://doi.org/10.1002/aqc.3270040102
Chown SL, Haupt TM, Sinclair BJ (2016) Similar metabolic rate-temperature relationships after acclimation at constant and fluctuating temperatures in caterpillars of a sub-Antarctic moth. J Insect Physiol 85:10–16. https://doi.org/10.1016/j.jinsphys.2015.11.010
Clarke A (2004) Is there a universal temperature dependence of metabolism? Funct Ecol 18:252–256. https://doi.org/10.1111/j.0269-8463.2004.00842.x
Clavier J, Castets MD, Bastian T, Hily C, Boucher G, Chauvaud L (2009) An amphibious mode of life in the intertidal zone: aerial and underwater contribution of Chthamalus montagui to CO2 fluxes. Mar Ecol Prog Ser 375:185–194
Cloern JE, Foster SQ, Kleckner AE (2014) Phytoplankton primary production in the world’s estuarine-coastal ecosystems. Biogeosciences 11:2477–2501
De Kock KN, Wolmarans CT (2007) Distribution and habitats of Corbicula fluminalis africana (Mollusca: Bivalvia) in South Africa. Water Sa 33:709–715
del Giorgio PA, le Williams PJB (2005) Respiration in aquatic ecosystems. Oxford University Press, Oxford
Dell AI, Pawar S, Savage VM (2011) Systematic variation in the temperature dependence of physiological and ecological traits. Proc Natl Acad Sci 108:10591–10596. https://doi.org/10.1073/pnas.1015178108
Deutsch CA, Tewksbury JJ, Huey RB, Sheldon KS, Ghalambor CK, Haak DC, Martin PR (2008) Impacts of climate warming on terrestrial ectotherms across latitude. Proc Natl Acad Sci 105:6668–6672. https://doi.org/10.1073/pnas.0709472105
Dore MHI (2005) Climate change and changes in global precipitation patterns: what do we know? Environ Int 31:1167–1181. https://doi.org/10.1016/j.envint.2005.03.004
DWS D of W&S (2015) Resource directed measures: reserve determination study of selected surface water and roundwater resources in the Usutu/Mhlathuze Water Management Area. Mlalazi Estuary Rapid Environmental Water Requirements Determination. CRUZ Environmental for Tlou Consulting (Pty) Ltd
Elliott M, Quintino V (2007) The estuarine quality paradox, environmental homeostasis and the difficulty of detecting anthropogenic stress in naturally stressed areas. Mar Pollut Bull 54:640–645. https://doi.org/10.1016/j.marpolbul.2007.02.003
Ferreira JG, Wolff WJ, Simas TC, Bricker SB (2005) Does biodiversity of estuarine phytoplankton depend on hydrology? Ecol Model 187:513–523. https://doi.org/10.1016/j.ecolmodel.2005.03.013
Folguera G, Bastías DA, Caers J, Rojas JM, Piulachs M-D, Bellés X, Bozinovic F (2011) An experimental test of the role of environmental temperature variability on ectotherm molecular, physiological and life-history traits: implications for global warming. Comp Biochem Physiol A Mol Integr Physiol 159:242–246. https://doi.org/10.1016/j.cbpa.2011.03.002
Gherardi F, Russo S, Lazzara L (2002) The function of wandering in the sand-bubbler crab, Dotilla fenestrata. J Crustac Biol 22:521–531. https://doi.org/10.1651/0278-0372(2002)022[0521:TFOWIT]2.0.CO;2
Gillooly JF, Brown JH, West GB, Savage VM, Charnov EL (2001) Effects of size and temperature on metabolic rate. Science 293:2248–2251. https://doi.org/10.1126/science.1061967
Gillooly JF, Londoño GA, Allen AP (2008) Energetic constraints on an early developmental stage: a comparative view. Biol Lett 4:123–126. https://doi.org/10.1098/rsbl.2007.0460
Glazier DS (2005) Beyond the ‘3/4-power law’: variation in the intra- and interspecific scaling of metabolic rate in animals. Biol Rev 80:611–662
Glazier DS (2006) The 3/4-power law is not universal: evolution of isometric, ontogenetic metabolic scaling in pelagic animals. Bioscience 56:325–332
Harrison TD (2004) Physico-chemical characteristics of South African estuaries in relation to the zoogeography of the region. Estuar Coast Shelf Sci 61:73–87. https://doi.org/10.1016/j.ecss.2004.04.005
Hill BJ (1966) A contribution to the ecology of the Umlalazi Estuary. Zool Afr 2:1–24
Hillebrand H, Dürselen C-D, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424
Huey RB, Kearney MR, Krockenberger A, Holtum JAM, Jess M, Williams SE (2012) Predicting organismal vulnerability to climate warming: roles of behaviour, physiology and adaptation. Philos Trans R Soc B Biol Sci 367:1665–1679. https://doi.org/10.1098/rstb.2012.0005
Ivleva IV (1980) The dependence of crustacean respiration rate on body mass and habitat temperature. Int Rev Gesamten Hydrobiol Hydrogr 65:1–47. https://doi.org/10.1002/iroh.19800650102
Litulo C, Mahanjane Y, Mantelatto FLM (2005) Population biology and breeding period of the sand-bubbler crab Dotilla fenestrata (Brachyura: Ocypodidae) from southern Mozambique. Aquat Ecol 39:305–313. https://doi.org/10.1007/s10452-005-3443-9
Madeira D, Narciso L, Cabral HN, Vinagre C (2012) Thermal tolerance and potential impacts of climate change on coastal and estuarine organisms. J Sea Res 70:32–41. https://doi.org/10.1016/j.seares.2012.03.002
Mclachlan A (1974) Notes on the biology of some estuarine bivalves. Zool Afr 9:15–34. https://doi.org/10.1080/00445096.1974.11448518
Mclachlan A, Erasmus T (1974) Temperature tolerances and osmoregulation in some estuarine bivalves. Zool Afr 9:1–13. https://doi.org/10.1080/00445096.1974.11448517
Miranda NAF, Perissinotto R (2014) Benthic assemblages of wetlands invaded by Tarebia granifera (Lamarck, 1822) (Caenogastropoda: Thiaridae) in the iSimangaliso Wetland Park, South Africa. Molluscan Res 34:40–48. https://doi.org/10.1080/13235818.2013.866177
Miranda NA, Perissinotto R, Appleton CC (2010) Salinity and temperature tolerance of the invasive freshwater gastropod Tarebia granifera. S Afr J Sci 106:01–07
Miranda NAF, Perissinotto R, Appleton CC (2011) Population structure of an invasive parthenogenetic gastropod in coastal lakes and estuaries of Northern KwaZulu-Natal, South Africa. PLOS One 6:e24337. https://doi.org/10.1371/journal.pone.0024337
Naidu S, Hounsome R, Iyer K (2006) Climatic future for Durban, prepared by CSIR Environmentek. In: Roberts D, Mather A, Maganlal M (eds) EThekwini Municipality
Ortega-Cisneros K, Scharler UM (2014) Variability and temporal stability of communities in estuaries (Mlalazi and Mpenjati, South Africa). Mar Ecol Prog Ser 500:11–24. https://doi.org/10.3354/meps10658
Ortega-Cisneros K, Scharler UM (2015) Nutrient dynamics of estuarine invertebrates are shaped by feeding guild rather than seasonal river flow. PLoS One 10:e0137417. https://doi.org/10.1371/journal.pone.0137417
Ortega-Cisneros K, Scharler UM, Whitfield AK (2016) Carbon and nitrogen system dynamics in three small South African estuaries, with particular emphasis on the influence of seasons, river flow and mouth state. Mar Ecol Prog Ser 557:17–30
Pawar S, Dell AI, Savage VM, Knies JL (2016) Real versus artificial variation in the thermal sensitivity of biological traits. Am Nat 187:E41–E52. https://doi.org/10.1086/684590
Peck LS, Conway LZ (2000) The myth of metabolic cold adaptation: oxygen consumption in stenothermal Antarctic bivalves. Geol Soc Lond Spec Publ 177:441–450. https://doi.org/10.1144/GSL.SP.2000.177.01.29
Pillay D, Branch GM, Forbes AT (2007) The influence of bioturbation by the sandprawn Callianassa kraussi on feeding and survival of the bivalve Eumarcia paupercula and the gastropod Nassarius kraussianus. J Exp Mar Biol Ecol 344:1–9. https://doi.org/10.1016/j.jembe.2006.10.044
Pörtner H-O (2002) Climate variations and the physiological basis of temperature dependent biogeography: systemic to molecular hierarchy of thermal tolerance in animals. Comp Biochem Physiol A Mol Integr Physiol 132:739–761
Pörtner H-O (2008) Contribution to the theme section ‘Effects of ocean acidification on marine ecosystems’ Ecosystem effects of ocean acidification in times of ocean warming: a physiologist’s view. Mar Ecol Prog Ser 373:203–217. https://doi.org/10.3354/meps07768
Rosati I, Barbone E, Basset A (2012) Length–mass relationships for transitional water benthic macroinvertebrates in Mediterranean and Black Sea ecosystems. Estuar Coast Shelf Sci 113:231–239. https://doi.org/10.1016/j.ecss.2012.08.008
Roy PS, Williams RJ, Jones AR, Yassini I, Gibbs PJ, Coates B, West RJ, Scanes PR, Hudson JP, Nichol S (2001) Structure and function of south-east Australian estuaries. Estuar Coast Shelf Sci 53:351–384. https://doi.org/10.1006/ecss.2001.0796
Scharler UM, Baird D (2005) A comparison of selected ecosystem attributes of three South African estuaries with different freshwater inflow regimes, using network analysis. J Mar Syst 56:283–308. https://doi.org/10.1016/j.jmarsys.2004.12.003
Schulte PM, Healy TM, Fangue NA (2011) Thermal Performance curves, phenotypic plasticity, and the time scales of temperature exposure. Integr Comp Biol 51:691–702. https://doi.org/10.1093/icb/icr097
Seebacher F, White CR, Franklin CE (2015) Physiological plasticity increases resilience of ectothermic animals to climate change. Nat Clim Change 5:61–66. https://doi.org/10.1038/nclimate2457
Seibel BA, Drazen JC (2007) The rate of metabolism in marine animals: environmental constraints, ecological demands and energetic opportunities. Philos Trans R Soc Lond B Biol Sci 362:2061–2078
Smit H, Van Heel ED, Wiersma S (1993) Biovolume as a tool in biomass determination of Oligochaeta and Chironomidae. Freshw Biol 29:37–46. https://doi.org/10.1111/j.1365-2427.1993.tb00742.x
Sokolova IM, Pörtner HO (2003) Metabolic plasticity and critical temperatures for aerobic scope in a eurythermal marine invertebrate (Littorina saxatilis, Gastropoda: Littorinidae) from different latitudes. J Exp Biol 206:195–207. https://doi.org/10.1242/jeb.00054
Somero GN (2002) Thermal physiology and vertical zonation of intertidal animals: optima, limits, and costs of living. Integr Comp Biol 42:780–789
Somero GN (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers’. J Exp Biol 213:912–920
Stretch DD, Zietsman I (2004) The hydrodynamics of Mhlanga and Mdloti estuaries: flows, residence times, water levels and mouth dynamics, Water Research Commission Project K5/1247-Final Report (Hydrodynamics)
Tagliarolo M, McQuaid C (2015) Sub-lethal and sub-specific temperature effects are better predictors of mussel distribution than thermal tolerance. Mar Ecol Prog Ser 535:145–159. https://doi.org/10.3354/meps11434
Tagliarolo M, Clavier J, Chauvaud L, Koken M, Grall J (2012) Metabolism in blue mussel: intertidal and subtidal beds compared. Aquat Biol 17:167–180. https://doi.org/10.3354/ab00464
Tattersall GJ, Sinclair BJ, Withers PC, Fields PA, Seebacher F, Cooper CE, Maloney SK (2012) Coping with thermal challenges: physiological adaptations to environmental temperatures. Compr Physiol 2:2151–2202
Teske PR, Wooldridge TH (2003) What limits the distribution of subtidal macrobenthos in permanently open and temporarily open/closed South African estuaries? Salinity vs. sediment particle size. Estuar Coast Shelf Sci 57:225–238. https://doi.org/10.1016/S0272-7714(02)00347-5
Tomanek L (2010) Variation in the heat shock response and its implication for predicting the effect of global climate change on species’ biogeographical distribution ranges and metabolic costs. J Exp Biol 213:971–979
Tronstad LM, Tronstad BP, Benke AC (2010) Growth rates of chironomids collected from an ephemeral floodplain wetland. Wetlands 30:827–831. https://doi.org/10.1007/s13157-010-0074-2
Watson S-A, Morley SA, Bates AE, Clark MS, Day RW, Lamare M, Martin SM, Southgate PC, Tan KS, Tyler PA, Peck LS (2014) Low global sensitivity of metabolic rate to temperature in calcified marine invertebrates. Oecologia 174:45–54. https://doi.org/10.1007/s00442-013-2767-8
Weber RE (1980) Functions of invertebrate hemoglobins with special reference to adaptations to environmental hypoxia. Am Zool 20:79–101
Whitfield A, Bate G, Adams J, Cowley P, Froneman P, Gama P, Strydom N, Taljaard S, Theron A, Turpie J, van Niekerk L, Wooldridge T (2012) A review of the ecology and management of temporarily open/closed estuaries in South Africa, with particular emphasis on river flow and mouth state as primary drivers of these systems. Afr J Mar Sci 34:163–180. https://doi.org/10.2989/1814232X.2012.675041
Acknowledgements
We would like to thank Ezemvelo KZN Wildlife for their support during fieldwork and Fiona Mackay (Oceanographic Research Institute, Durban) for the help with identification. Many thanks also to Kajal Lechman, Dane Garvie, Thembeka Radebe, Christopher Waspe, Fifi Dikarabo Rafedile, and Gavin Tweddle for the assistance in the field.
Author information
Authors and Affiliations
Contributions
MT conceived and designed the experiments, performed the experiment, analysed the data, and wrote the manuscript. FP wrote the paper and facilitated the laboratory experiments. UMS wrote the paper and facilitated the field work.
Corresponding author
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Collection permit from the department of environmental affairs of the Republic of South Africa: RES2016-85.
Ethical approval
Animal Ethics protocol reference number: AREC/083/015PD from the University of KwaZulu-Natal.
Funding
This work was founded by the UKZN post-doctoral fellowship (granted to M. Tagliarolo) and the National Research Foundation (NRF) Grant (UID number: 102688) (granted to U. Scharler).
Conflict of interest
No conflict interests exist: M. Tagliarolo declare that she has no conflict of interest. F. Porri declares that she has no conflict of interest. U. M. Scharler declares that she has no conflict of interest.
Additional information
Responsible Editor: H.-O. Pörtner.
Reviewed by Undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tagliarolo, M., Porri, F. & Scharler, U.M. Temperature-induced variability in metabolic activity of ecologically important estuarine macrobenthos. Mar Biol 165, 23 (2018). https://doi.org/10.1007/s00227-017-3276-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-017-3276-9