Abstract
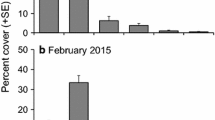
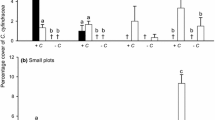
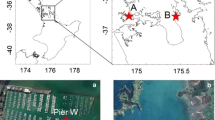
Disturbance can play an important role in accelerating the spread of invasive species in coastal habitats by increasing the availability of resources. Thus, reductions in native canopy cover often facilitate the establishment of invasive macroalgae. In this study, the invasion process of the kelp Undaria pinnatifida in disturbed patches on a rocky low-intertidal shore in central New Zealand was investigated. The study site was initially uninvaded, but adjacent to (~150 m) intertidal areas with established U. pinnatifida populations. Experimental plots (0.25 m2) were established that were either partially (50%) or totally (100%) cleared of native algal cover at two different times of year (summer: December 2008 and winter: June 2009). U. pinnatifida recruitment was monitored for 3, 6, 9 and 12 months for the summer run, and for 3 and 6 months for the winter run. Although the timing of first appearance varied across the two runs, we were unable to detect an effect of clearing native algae on U. pinnatifida recruitment. By the end of the experiment, U. pinnatifida had recruited into all plot treatment types at low densities (on average 1.4 plants 0.25 m−2). The fact that U. pinnatifida recruited into the uncleared control plots indicated that invasion of this kelp into new areas does not require disturbance of the native algal community. These results suggest that if light and space are not limiting resources, as is likely the case in this low-intertidal habitat, then disturbance may not facilitate U. pinnatifida recruitment.


Similar content being viewed by others
References
Airoldi L (1998) Roles of disturbance, sediment stress, and substratum retention on spatial dominance in algal turf. Ecology 79:2759–2770
Alpert P, Bone E, Holzapfel C (2000) Invasiveness, invasibility and the role of environmental stress in the spread of non-native plants. Perspect Plant Ecol 3:52–66
Andrew NL, Viejo RM (1998) Ecological limits to the invasion of Sargassum muticum in northern Spain. Aquat Bot 60:251–263
Araújo R, Vaselli S, Almeida M, Serrão E, Sousa-Pinto I (2009) Effects of disturbance on marginal populations: human trampling on Ascophyllum nodosum assemblages at its southern distribution limit. Mar Ecol Prog Ser 378:81–92
Arenas F, Sánchez I, Hawkins SJ, Jenkins SR (2006) The invasibility of marine algal assemblages: role of functional diversity and identity. Ecology 87:2851–2861
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models Using lme4. J Stat Softw 67:1–48
Benedetti-Cecchi L, Cinelli F (1994) Recovery of patches in an assemblage of geniculate coralline algae: variability at different successional stages. Mar Ecol Prog Ser 110:9–18
Bertness MD, Leonard GH, Levine JM, Schmidt PR, Ingraham AO (1999) Testing the relative contribution of positive and negative interactions in rocky intertidal communities. Ecology 80:2711–2726
Bolton JJ, Levitt GJ (1985) Light and temperature requirements for growth and reproduction in gametophytes of Ecklonia maxima (Alariaceae: laminariales). Mar Biol 87:131–135
Britton-Simmons KH (2006) Functional group diversity, resource pre-emption and the genesis of invasion resistance in a community of marine algae. Oikos 113:395–401
Carnell PE, Keough MJ (2014) Spatially variable synergistic effects of disturbance and additional nutrients on kelp recruitment and recovery. Oecologia 175:409–416
Choi HC, Kim YS, Lee SJ, Park EJ, Nam KW (2005) Effects of daylength, irradiance and settlement density on the growth and reproduction of Undaria pinnatifida gametophytes. J Appl Phycol 17:423–430
Dayton PK (1971) Competition, disturbance, and community organization: the provision and subsequent utilization of space in a rocky intertidal community. Ecol Monogr 41:351–389
Dayton PK (1975) Experimental evaluation of ecological dominance in a rocky intertidal algal community. Ecol Monogr 45:137–159
Dellatore FG, Amoroso R, Saravia J, Orensanz JM (2014) Rapid expansion and potential range of the invasive kelp Undaria pinnatifida in the Southwest Atlantic. Aquat Invasions 9:467–478
Demello R, Phillips NE (2011) Variation in mussel and barnacle recruitment parallels a shift in intertidal community structure in the Cook Strait region of New Zealand. Mar Freshw Res 62:1221–1229
Deysher LE, Dean TA (1986) In situ recruitment of sporophytes of the giant kelp, Macrocystis pyrifera (L.) C.A. Agardh: effects of physical factors. J Exp Mar Biol Ecol 103:41–63
Edgar GJ, Barrett NS, Morton AJ, Samson CR (2004) Effects of algal canopy clearance on plant, fish and macroinvertebrate communities on eastern Tasmanian reefs. J Exp Mar Biol Ecol 312:67–87
Farrell TM (1989) Succession in a rocky intertidal community: the importance of disturbance size and position within a disturbed patch. J Exp Mar Biol Ecol 128:57–73
Farrell P, Fletcher RL (2006) An investigation of dispersal of the introduced brown alga Undaria pinnatifida (Harvey) Suringar and its competition with some species on the man-made structures of Torquay Marina (Devon, UK). J Exp Mar Biol Ecol 334:236–243
Floc’h JY, Pajot R, Wallentinus I (1991) The Japanese brown alga Undaria pinnatifida on the coast of France and its possible establishment in European waters. J Cons 47:379–390
Forrest BM, Brown SN, Taylor MD, Hurd CL, Hay CH (2000) The role of natural dispersal mechanisms in the spread of Undaria pinnatifida (Laminariales, Phaeophyceae). Phycologia 39:547–553
Gagnon K, McKindsey CW, Johnson LE (2015) Roles of dispersal mode, recipient environment and disturbance in the secondary spread of the invasive seaweed Codium fragile. Biol Invasions 17:1123–1136
Harrison XA (2014) Using observation-level random effects to model overdispersion in count data in ecology and evolution. Peer J. doi:10.7717/peerj.616
Hauck WW Jr, Donner A (1977) Wald’s test as applied to hypotheses in logit analysis. J Am Stat Assoc 72:851–853
Hay CH, Luckens PA (1987) The Asian kelp Undaria-Pinnatifida (Phaeophyta, Laminariales) found in a New-Zealand harbor. N Z J Bot 25:329–332
Hay CH, Villouta E (1993) Seasonality of the adventive Asian kelp Undaria pinnatifida in New-Zealand. Bot Mar 36:461–476
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6:324–337
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Hsiao SI, Dreuhl LD (1973) Environmental control of gametogenesis in Laminaria saccharina. IV. In situ development of gametophytes and young sporophytes. J Phycol 9:160–164
Jenkins SR, Norton TA, Hawkins SJ (2004) Long term effects of Ascophyllum nodosum canopy removal on mid shore community structure. J Mar Biol Assoc UK 84:327–329
Kennelly SJ (1987) Physical disturbances in an Australian kelp community. 1. Temporal effects. Mar Ecol Prog Ser 40:145–153
Keough MJ (1984) Effects of patch size on the abundance of sessile marine invertebrates. Ecology 65:423–437
Keough MJ, Quinn GP (1998) Effects of periodic disturbance from trampling on rocky intertidal algal beds. Ecol Appl 8:141–161
Kim JH, DeWreede RE (1996) Effect of size and season of disturbance on algal patch recovery in a rocky intertidal community. Mar Ecol Prog Ser 133:217–228
Kuznetsova A, Brockhoff PB, Christensen RHB (2013) lmerTest: tests for random and fixed effects for linear mixed effect models (lmer objects of lme4 package)R-Version 1.1-0 http://cran.rproject.org/web/packages/lmerTest/index.html
Levin PS, Coyer JA, Petrik R, Good TP (2002) Community-wide effects of nonindigenous species in temperate rocky reefs. Ecology 83:3182–3193
Lilley SA, Schiel DR (2006) Community effects following the deletion of a habitat-forming alga from rocky marine shores. Oecologia 148:672–681
Luning K, Neushul M (1978) Light and temperature demands for growth and reproduction of laminarian gametophytes in southern and central California. Mar Biol 45:297–309
Middelboe AL, Binzer T (2004) Importance of canopy structure on photosynthesis in single-and multi-species assemblages of marine macroalgae. Oikos 170:422–432
Milazzo M, Chemello R, Badalamenti F, Riggio S (2002) Short-term effect of human trampling on the upper infralittoral macroalgae of Ustica Island MPA (western Mediterranean, Italy). J Mar Biol Assoc UK 82:745–748
Morelissen B (2012) Ecological effects of Undaria pinnatifida (Harvey) Suringar and nutrient-enrichment on intertidal assemblages in the Wellington region of New Zealand. Victoria University of Wellington, Wellington
Morelissen B, Dudley BD, Geange SW, Phillips NE (2013) Gametophyte reproduction and development of Undaria pinnatifida under varied nutrient and irradiance conditions. J Exp Mar Biol Ecol 448:197–206
Morita T, Kurashima A, Maegawa M (2003) Temperature requirements for the growth and maturation of the gametophytes of Undaria pinnatifida and U. undarioides (Laminariales, Phaeophyceae). Phycol Res 51:154–160
Paine RT (1979) Disaster, catastrophe, and local persistence of the sea palm Postelsia palmaeformis. Science 205:685–687
Paine RT, Levin SA (1981) Intertidal landscapes: disturbance and the dynamics of pattern. Ecol Monogr 51:145–178
Raffo MP, Eyras MC, Iribarne OO (2009) The invasion of Undaria pinnatifida to a Macrocystis pyrifera kelp in Patagonia (Argentina, south-west Atlantic). J Mar Biol Assoc UK 89:1571–1580
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org/
Reed DC, Foster MS (1984) The effects of canopy shading on algal recruitment and growth in a giant kelp forest. Ecology 65:937–948
Reed DC, Raimondi PT, Carr MH, Goldwasser L (2000) The role of dispersal and disturbance in determining spatial heterogeneity in sedentary organisms. Ecology 81:2011–2026
Russell LK, Hepburn CD, Hurd CL, Stuart MD (2008) The expanding range of Undaria pinnatifida in southern New Zealand: distribution, dispersal mechanisms and the invasion of wave exposed environments. Biol Invasions 10:103–115
Saito Y (1975) Undaria. In: Tokida J, Hirose H (eds) Advance in phycology in Japan. Dr. W. Junk, The Hague
Scheibling RE, Gagnon P (2006) Competitive interactions between the invasive green alga Codium fragile ssp. tomentosoides and native canopy-forming seaweeds in Nova Scotia (Canada). Mar Ecol Prog Ser 325:1–14
Schiel DR (1988) Algal interactions on shallow subtidal reefs in northern New Zealand: a review. N Z J Mar Fresh 22:481–489
Schiel DR, Lilley SA (2007) Gradients of disturbance to an algal canopy and the modification of an intertidal community. Mar Ecol Prog Ser 339:1–11
Schiel DR, Taylor DI (1999) Effects of trampling on a rocky intertidal algal assemblage in southern New Zealand. J Exp Mar Biol Ecol 235:213–235
Schiel DR, Thompson GA (2012) Demography and population biology of the invasive kelp Undaria pinnatifida on shallow reefs in southern New Zealand. J Exp Mar Biol Ecol 434–435:25–33
Silva PC, Woodfield RA, Cohen AN, Harris LH, Goddard JHR (2002) First report of the Asian kelp Undaria pinnatifida in the northeastern Pacific Ocean. Biol Invasions 4:333–338
Sliwa C, Johnson CR, Hewitt CL (2006) Mesoscale dispersal of the introduced kelp Undaria pinnatifida attached to unstable substrata. Bot Mar 49:396–405
Sousa WP (1979) Experimental investigations of disturbance and ecological succession in a rocky intertidal algal community. Ecol Monogr 49:228–254
Sousa WP (1984a) The role of disturbance in natural communities. Annu Rev Ecol Syst 15:353–391
Sousa WP (1984b) Intertidal mosaics: patch size, propagule availability, and spatially variable patterns of succession. Ecology 65:1918–1935
Thompson GA, Schiel DR (2012) Resistance and facilitation by native algal communities in the invasion success of Undaria pinnatifida. Mar Ecol Prog Ser 468:95–105
Thornber CS, Kinlan BP, Graham MH, Stachowicz JJ (2004) Population ecology of the invasive kelp Undaria pinnatifida in California: environmental and biological controls on demography. Mar Ecol Prog Ser 268:69–80
Underwood AJ (1998) Grazing and disturbance: an experimental analysis of patchiness in recovery from a severe storm by the intertidal alga Hormosira banksii on rocky shores in New South Wales. J Exp Mar Biol Ecol 231:291–306
Underwood AJ (1999) Physical disturbances and their direct effect on an indirect effect: responses of an intertidal assemblage to a severe storm. J Exp Mar Biol Ecol 232:125–140
Valentine JP, Johnson CR (2003) Establishment of the introduced kelp Undaria pinnatifida in Tasmania depends on disturbance to native algal assemblages. J Exp Mar Biol Ecol 295:63–90
Valentine JP, Johnson CR (2004) Establishment of the introduced kelp Undaria pinnatifida following dieback of the native macroalga Phyllospora comosa in Tasmania, Australia. Mar Freshw Res 55:223–230
Valentine JP, Magierowski RH, Johnson CR (2007) Mechanisms of invasion: establishment, spread and persistence of introduced seaweed populations. Bot Mar 50:351–360
Vaz-Pinto F, Olabarria C, Arenas F (2012) Propagule pressure and functional diversity: interactive effects on a macroalgal invasion process. Mar Ecol Prog Ser 471:51–60
Veiga P, Torres AC, Rubal M, Troncoso J, Sousa-Pinto I (2014) The invasive kelp Undaria pinnatifida (Laminariales, Ochrophyta) along the north coast of Portugal: distribution model versus field observations. Mar Pollut Bull 84:363–365
Acknowledgements
This research was funded by Victoria University of Wellington and the New Zealand Biosecurity Institute (awarded to B. M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Additional information
Responsible Editor: K. Bischof.
Reviewed by Undisclosed experts.
This article is part of the Topical Collection on Invasive Species.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Morelissen, B., Dudley, B.D. & Phillips, N.E. Recruitment of the invasive kelp Undaria pinnatifida does not always benefit from disturbance to native algal communities in low-intertidal habitats. Mar Biol 163, 241 (2016). https://doi.org/10.1007/s00227-016-3014-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-3014-8