Abstract
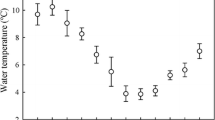
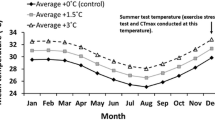
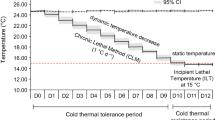
Global climate change is predicted to increase the variability in weather patterns with more extreme weather conditions occurring on a more frequent basis. Little information exists on thermal limits of fishes from highly variable environments. This study evaluated the thermal maximum and minimum of checkered puffers, yellowfin mojarra, schoolmaster snapper, and bonefish across seasons. Thermal scope (i.e., CTmax–CTmin) of nearshore fishes ranged from 24 to 28.6 °C across seasons, with thermal scopes typically being larger in the winter (January 1, 2012–March 22, 2012) than in the summer (June 26, 2012–November 9, 2012). Acclimatization response ratios (AZRR; ΔCTmax ΔT −1 and ΔCTmin ΔT −1) were typically greater than 0.60 for all species, a value greater than most previously reported for fish species from variable thermal environments. Present-day maximum and minimum temperatures in the nearshore environment are approximately equal to or exceed the thermal tolerance limits of the fish in this study, making thermal safety margins (TSM; i.e., the difference between thermal tolerance limit and extreme environmental temperature) very small or negative for nearshore fishes (TSM upper = −4.9 to 0.5; lower = −0.2 to 0.4). The IPCC’s worse-case scenario will push maximum temperatures beyond the TSM of all nearshore fish in this study. Distribution of fishes in the nearshore environment in the future will depend on available thermal refuge, cost of migrating, and food web interactions. Overall, the thermal landscape in the nearshore environment in the future will likely benefit species with positive thermal safety margins that are capable of acclimatizing (e.g., schoolmaster snapper), while relatively intolerant species (e.g., bonefish) may inhabit these systems less frequently or will be absent in the future.

Similar content being viewed by others
References
Barbier EB, Hacker SD, Kennedy C et al (2011) The value of estuarine and coastal ecosystem services. Ecol Monogr 81:169–193. doi:10.1890/10-1510.1
Beitinger TL, Bennett WA (2000) Quantification of the role of acclimation temperature in temperature tolerance of fishes. Environ Biol Fishes 58:277–288. doi:10.1023/A:1007618927527
Beitinger TL, Bennett WA, Mccauley RW (2000) Temperature tolerances of North American freshwater fishes exposed to dynamic changes in temperature. Environ Biol Fishes 58:237–275. doi:10.1023/A:1007676325825
Bennett WA, Beitinger TL (1997) Temperature tolerance of the sheepshead minnow, Cyprinodon variegatus. Copia 1:77–87. doi:10.2307/1447842
Chipps SR, Clapp DF, Wahl DH (2000) Variation in routine metabolism of juvenile muskellunge: evidence for seasonal metabolic compensation in fishes. J Fish Biol 56:311–318. doi:10.1111/j.1095-8649.2000.tb02108.x
Claussen DL (1977) Thermal acclimation in ambystomatid salamanders. Comp Biochem Physiol Part A Mol Integr Physiol 58:333–340. doi:10.1016/0300-9629(77)90150-5
Coumou D, Rahmstorf S (2012) A decade of weather extremes. Nat Clim Chang 2:491–496. doi:10.1038/NCLIMATE1452
Deutsch CA, Tewksbury JJ, Huey RB et al (2008) Impacts of climate warming on terrestrial ectotherms across latitude. Proc Natl Acad Sci 105:6668–6672. doi:10.1073/pnas.0709472105
Duarte H, Tejedo M, Katzenberger M et al (2012) Can amphibians take the heat? Vulnerability to climate warming in subtropical and temperate larval amphibian communities. Glob Chang Biol 18:412–421. doi:10.1111/j.1365-2486.2011.02518.x
Eliason EJ, Clark TD, Hague MJ et al (2011) Differences in thermal tolerance among sockeye salmon populations. Science 332:109–112. doi:10.1126/science.1199158
Evans DO (1984) Temperature independence of the annual cycle of standard metabolism in the pumpkinseed. Trans Am Fish Soc 113:494–512. doi:10.1577/1548-8659(1984)113<494:TIOTAC>2.0.CO;2
Fader SC, Yu Z, Spotila JR (1994) Seasonal variation in heat shock proteins (hsp70) in stream fish under natural conditions. J Therm Biol 19:335–341. doi:10.1016/0306-4565(94)90070-1
Fangue N, Bennett W (2003) Thermal tolerance responses of laboratory-acclimated and seasonally acclimatized Atlantic stingray, Dasyatis sabina. Copeia 2:315–325. doi:10.1643/0045-8511(2003)003[0315:TTROLA]2.0.CO;2
Feidantsis K, Antonopoulou E, Lazou A et al (2013) Seasonal variations of cellular stress response of the gilthead sea bream (Sparus aurata). J Comp Physiol B 183:625–639. doi:10.1007/s00360-012-0735-y
Ghalambor CK, Huey RB, Martin PR et al (2006) Are mountain passes higher in the tropics? Janzen’s hypothesis revisited. Integr Comp Biol 46:5–17. doi:10.1093/icb/icj003
Gunderson AR, Stillman JH (2015) Plasticity in thermal tolerance has limited potential to buffer ectotherms from global warming. Proc R Soc B Biol Sci 282:20150401. doi:10.1098/rspb.2015.0401
Hartley HO (1950) The maximum F-ratio as a short-cut test for heterogeneity of variance. Biometrika 37:308–312. doi:10.2307/2332383
Hofmann GE, Todgham AE (2010) Living in the now: physiological mechanisms to tolerate a rapidly changing environment. Annu Rev Physiol 72:127–145. doi:10.1146/annurev-physiol-021909-135900
Hofmann GE, Barry JP, Edmunds PJ et al (2010) The effect of ocean acidification on calcifying organisms in marine ecosystems: an organism-to-ecosystem perspective. Annu Rev Ecol Evol Syst 41:127–147. doi:10.1146/annurev.ecolsys.110308.120227
Hopkin RS, Qari S, Bowler K et al (2006) Seasonal thermal tolerance in marine Crustacea. J Exp Mar Biol Ecol 331:74–81. doi:10.1016/j.jembe.2005.10.007
Huey RB, Tewksbury JJ (2009) Can behavior douse the fire of climate warming? Proc Natl Acad Sci 106:3647–3648. doi:10.1073/pnas.0900934106
IPCC (2013) Climate change 2013: The physical basis. Contribution of working group 1 to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jayasundara N, Somero GN (2013) Physiological plasticity of cardiorespiratory function in a eurythermal marine teleost, the longjaw mudsucker, Gillichthys mirabilis. J Exp Biol 216:2111–2121. doi:10.1242/jeb.083873
Kerr RA (2011) Humans are driving extreme weather; time to prepare. Science 334:1040. doi:10.1126/science.334.6059.1040
Knutson TR, McBride JL, Chan J et al (2010) Tropical cyclones and climate change. Nat Geosci 3:157–163. doi:10.1038/NGEO779
Komoroske LM, Connon RE, Lindberg J et al (2014) Ontogeny influences sensitivity to climate change stressors in an endangered fish. Conserv Physiol. doi:10.1093/conphys/cou008
Lam K, Tsui T, Nakano K, Randall DJ (2006) Physiological adaptations of fishes to tropical intertidal environments. In: Val AL, De Almeida-Val VMF, Randall DJ (eds) Fish physiology: the physiology of tropical fishes, vol 21. Academic Press Inc, London, pp 501–581
Madeira D, Narciso L, Cabral HN, Vinagre C (2012) Thermal tolerance and potential impacts of climate change on coastal and estuarine organisms. J Sea Res 70:32–41. doi:10.1016/j.seares.2012.03.002
Magozzi S, Calosi P (2014) Integrating metabolic performance, thermal tolerance, and plasticity enables for more accurate predictions on species vulnerability to acute and chronic effects of global warming. Glob Chang Biol 21:181–194. doi:10.1111/gcb.12695
Mora C, Ospina AF (2001) Tolerance to high temperatures and potential impact of sea warming on reef fishes of Gorgona Island (tropical eastern Pacific). Mar Biol 139:765–769. doi:10.1007/s002270100626
Murchie KJ, Danylchuk SE, Pullen CE et al (2009) Strategies for the capture and transport of bonefish, Albula vulpes, from tidal creeks to a marine research laboratory for long-term holding. Aquac Res 40:1538–1550. doi:10.1111/j.1365-2109.2009.02255.x
Murchie KJ, Cooke SJ, Danylchuk AJ et al (2011) Thermal biology of bonefish (Albula vulpes) in Bahamian coastal waters and tidal creeks: an integrated laboratory and field study. J Therm Biol 36:38–48. doi:10.1016/j.jtherbio.2010.10.005
Murchie KJ, Cooke SJ, Danylchuk AJ et al (2013) Movement patterns of bonefish (Albula vulpes) in tidal creeks and coastal waters of Eleuthera, The Bahamas. Fish Res 147:404–412. doi:10.1016/j.fishres.2013.03.019
Nilsson GE, Crawley N, Lunde IG, Munday PL (2009) Elevated temperature reduces the respiratory scope of coral reef fishes. Glob Chang Biol 15:1405–1412. doi:10.1111/j.1365-2486.2008.01767.x
NOAA (2010) National Oceanic and Atmospheric Administration’s National Weather Source. National Data Buoy Center. http://www.ndbc.noaa.gov/N
Norin T, Malte H, Clark TD (2014) Aerobic scope does not predict the performance of a tropical eurythermal fish at elevated temperatures. J Exp Biol 217:244–251. doi:10.1242/jeb.089755
Parmesan C, Root TL, Willig MR (2000) Impacts of extreme weather and climate on terrestrial biota. Bull Am Meteorol Soc 81:443–450. doi:10.1175/1520-0477(2000)081<0443:IOEWAC>2.3.CO;2
Perry AL, Low PJ, Ellis JR, Reynolds JD (2005) Climate change and distribution shifts in marine fishes. Science 308:1912–1915. doi:10.1126/science.1111322
Poloczanska ES, Brown CJ, Sydeman WJ et al (2013) Global imprint of climate change on marine life. Nat Clim Chang 3:919–925. doi:10.1038/nclimate1958
Pörtner HO (2002) Climate variations and the physiological basis of temperature dependent biogeography: systemic to molecular hierarchy of thermal tolerance in animals. Comp Biochem Physiol A: Mol Integr Physiol 132:739–761. doi:10.1016/S1095-6433(02)00045-4
Pörtner HO, Farrell AP (2008) Physiology and climate change. Science 322:690–692. doi:10.1126/science.1163156
Pörtner HO, Peck MA (2010) Climate change effects on fishes and fisheries: towards a cause-and-effect understanding. J Fish Biol 77:1745–1779. doi:10.1111/j.1095-8649.2010.02783.x
Reyes I, Díaz F, Re AD, Pérez J (2011) Behavioral thermoregulation, temperature tolerance and oxygen consumption in the Mexican bullseye puffer fish, Sphoeroides annulatus Jenyns (1842), acclimated to different temperatures. J Therm Biol 36:200–205. doi:10.1016/j.jtherbio.2011.03.003
Roessig JM, Woodley CM, Cech JJ, Hansen LJ (2005) Effects of global climate change on marine and estuarine fishes and fisheries. Rev Fish Biol Fish 14:251–275. doi:10.1007/s11160-004-6749-0
Rummer JL, Couturier CS, Stecyk JAW et al (2013) Life on the edge: thermal optima for aerobic scope of equatorial reef fishes are close to current day temperatures. Glob Chang Biol 20:1055–1066. doi:10.1111/gcb.12455
Seebacher F, White CR, Franklin CE (2014) Physiological plasticity increases resilience of ectothermic animals to climate change. Nat Clim Chang 5:61–66. doi:10.1038/nclimate2457
Shultz AD, Zuckerman ZC, Stewart HA, Suski CD (2014) Seasonal blood chemistry response of sub-tropical nearshore fishes to climate change. Conserv Physiol 2:1–12. doi:10.1093/conphys/cou028.Introduction
Smale DA, Wernberg T (2013) Extreme climatic event drives range contraction of a habitat-forming species. Proc R Soc B Biol Sci 280:20122829. doi:10.1098/rspb.2012.2829
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. W.H Freeman, New York
Somero GN (2010) The physiology of climate change: How potentials for acclimatization and genetic adaptation will determine “winners” and “losers”. J Exp Biol 213:912–920. doi:10.1242/jeb.037473
Somero GN (2012) The physiology of global change: linking patterns to mechanisms. Annu Rev Mar Sci 4:39–61. doi:10.1146/annurev-marine-120710-100935
Stillman JH (2003) Acclimation capacity underlies susceptibility to climate change. Science 301:65. doi:10.1126/science.1083073
Sunday JM, Bates AE, Dulvy NK (2011) Global analysis of thermal tolerance and latitude in ectotherms. Proc R Soc B Biol Sci 278:1823–1830. doi:10.1098/rspb.2010.1295
Sunday JM, Bates AE, Kearney MR et al (2014) Thermal-safety margins and the necessity of thermoregulatory behavior across latitude and elevation. Proc Natl Acad Sci USA 111:5610–5615. doi:10.1073/pnas.1316145111
Szekeres P, Brownscombe JW, Cull F et al (2014) Physiological and behavioural consequences of cold shock on bonefish (Albula vulpes) in The Bahamas. J Exp Mar Biol Ecol 459:1–7. doi:10.1016/j.jembe.2014.05.003
Trenberth KE, Fasullo JT (2012) Climate extremes and climate change: the Russian heat wave and other climate extremes of 2010. J Geophys Res Atmos. doi:10.1029/2012JD018020
Valiela I, Bowen JL, York JK (2001) Mangrove forests: one of the world’s threatened major tropical environments. Bioscience 51:807–815. doi:10.1641/0006-3568(2001)051[0807:MFOOTW]2.0.CO;2
Vanlandeghem MM, Wahl DH, Suski CD (2010) Physiological responses of largemouth bass to acute temperature and oxygen stressors. Fish Manag Ecol 17:414–425. doi:10.1111/j.1365-2400.2010.00740.x
Wernberg T, Smale DA, Tuya F et al (2012) An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat Clim Chang 3:78–82. doi:10.1038/nclimate1627
Acknowledgments
We would like to acknowledge the hard-working interns and staff at the Island School and Cape Eleuthera Institute, in particular Kit Hayward, for his efforts in capturing and maintaining fish in the wetlab. Dr. Jocelyn Curtis-Quick gave constructive criticism on early drafts of this paper. The Environmental Change Institute at the University of Illinois and the Cape Eleuthera Island School provided financial support for this work. This work was conducted under permits issued by the Department of Marine Resources, The Bahamas.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: H.-O. Pörtner.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Shultz, A.D., Zuckerman, Z.C. & Suski, C.D. Thermal tolerance of nearshore fishes across seasons: implications for coastal fish communities in a changing climate. Mar Biol 163, 83 (2016). https://doi.org/10.1007/s00227-016-2858-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-2858-2