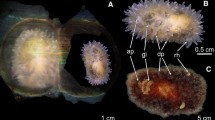
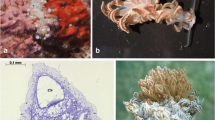
Abstract
The loss of the shell in nudibranch gastropods has been related to the acquisition of chemical defensive strategies during evolution, such as the use of natural products to deter predation. In the present study, we investigated the origin, location, and putative role of granuloside (1), a homosesterterpene lactone, recently isolated from the Antarctic nudibranch Charcotia granulosa Vayssière, 1906. Several adults, egg masses, and its bryozoan prey, Beania erecta Waters, 1904, were chemically analyzed by chromatographic and spectroscopic techniques. Light- and transmission electron microscopy of the mantle revealed complex glandular structures, which might be associated with the storage of defensive compounds in analogy to mantle dermal formations described in other nudibranchs. Although preliminary in situ repellence bioassays with live specimens of the nudibranch showed avoidance against the Antarctic generalist sea star predator Odontaster validus, the specific role of the terpene granuloside requires further investigation. The egg masses do not present granuloside, and the glandular structures are absent in the trochophore larvae. Our results suggest that C. granulosa synthesizes granuloside de novo in early stages of its ontogeny, instead of obtaining it from the prey. Considering the wide geographic area inhabited by this slug, this may be advantageous, because natural products produced by the slug will not be affected by fluctuant food availability. Overall, the Antarctic sea slug C. granulosa seems to possess defensive strategies that are similar, in terms of production and storage, to nudibranchs from other regions of the world. This species is one of the few cladobranchs investigated so far that present de novo biosynthesis of a defensive compound.






Similar content being viewed by others
References
Arnaud PM, Troncoso JS, Ramos A (2001) Species diversity and assemblages of macrobenthic Mollusca from the South Shetland Islands and Bransfield Strait (Antarctica). Polar Biol 24:105–112
Avila C (1995) Natural products of Opisthobranch molluscs: a biological review. Oceanogr Mar Biol 33:487–559
Avila C, Durfort M (1996) Histology of epithelia and mantle glands of selected species of doridacean mollusks with chemical defensive strategies. Veliger 39:148–163
Avila C, Paul V (1997) Chemical ecology of the nudibranch Glossodoris pallida: is the location of diet-derived metabolites important for defense? Mar Ecol Prog Ser 150:171–180. doi:10.3354/meps150171
Avila C, Cimino G, Fontana A et al (1991) Defensive strategy of two Hypselodoris nudibranchs from Italian and Spanish coasts. J Chem Ecol 17:625–636
Avila C, Iken K, Fontana A, Cimino G (2000) Chemical ecology of the Antarctic nudibranch Bathydoris hodgsoni Eliot, 1907: defensive role and origin of its natural products. J Exp Mar Bio Ecol 252:27–44
Avila C, Taboada S, Núñez-Pons L (2008) Antarctic marine chemical ecology: what is next? Mar Ecol 29:1–71. doi:10.1111/j.1439-0485.2007.00215.x
Barnes DKA, Brockington S (2003) Zoobenthic biodiversity, biomass and abundance at Adelaide Island, Antarctica. Mar Ecol Prog Ser 249:145–155
Barnes DKA, Bullough LW (1996) Some observations on the diet and distribution of nudibranchs at Signy Island, Antarctica. J Molluscan Stud 62:281–287
Benkendorff K, Davis AR, Bremner JB (2001) Chemical defense in the egg masses of benthic invertebrates: an assessment of antibacterial activity in 39 mollusks and 4 polychaetes. J Invertebr Pathol 78:109–118. doi:10.1006/jipa.2001.5047
Carbone M, Gavagnin M, Haber M, Guo Y-W, Fontana A, Manzo E, Genta-Jouve G, Tsoukatou M, Rudman WB, Cimino G, Ghiselin MT, Mollo E (2013) Packaging and delivery of chemical weapons: a defensive trojan horse stratagem in chromodorid nudibranchs. PLoS One 8:e62075. doi:10.1371/journal.pone.0062075
Cimino G, Ghiselin MT (2009) Chemical defense and the evolution of opisthobranch gastropods. Proc Calif Acad Sci 60:175–422
Cutignano A, Zhang W, Avila C, Cimino G, Fontana A (2011) Intrapopulation variability in the terpene metabolism of the Antarctic opisthobranch mollusc Austrodoris kerguelenensis. Eur J Org Chem 2011:5383–5389. doi:10.1002/ejoc.201100552
Cutignano A, Moles J, Avila C, Fontana A (2015) Granuloside, a unique linear homosesterterpene from the Antarctic nudibranch Charcotia granulosa. J Nat Prod 78:1761–1764. doi:10.1021/acs.jnatprod.5b00378
Davies-Coleman MT (2006) Secondary metabolites from the marine gastropod molluscs of Antarctica, Southern Africa and South America. In: Cimino G, Gavagnin M (eds) Molluscs: from chemo-ecological study to biotechnological application, vol 43. Series: progress in molecular and subcellular biology. Subseries: marine molecular biotechnology. Springer, Berlin, pp 133–157
Davis J, Fricke WF, Hamann MT, Esquenazi E, Dorrestein PC, Hill RT (2013) Characterization of the bacterial community of the chemically defended Hawaiian sacoglossan Elysia rufescens. Appl Environ Microbiol 79:7073–7081. doi:10.1128/AEM.01568-13
Dayton PK, Robilliard GA, Paine RT, Dayton LB (1974) Biological accommodation in the benthic community at McMurdo Sound, Antarctica. Ecol Monogr 44:105–128
Diyabalanage T, Iken KB, McClintock JB, Amsler CD, Baker BJ (2010) Palmadorins A–C, diterpene glycerides from the Antarctic nudibranch Austrodoris kerguelenensis. J Nat Prod 73:416–421. doi:10.1021/np900617m
Figuerola B, Núñez-Pons L, Moles J, Avila C (2013) Feeding repellence in Antarctic bryozoans. Naturwissenschaften 100:1069–1081. doi:10.1007/s00114-013-1112-8
Fontana A, Giménez F, Marin A, Mollo E, Cimino G (1994) Transfer of secondary metabolites from the sponges Dysidea fragilis and Pleraplysilla spinifera to the mantle dermal formations (MDFs) of the nudibranch Hypserlodoris webbi. Experientia 50:510–516
García-Gómez JC, Cimino G, Medina A (1990) Studies on the defensive behaviour of Hypselodoris species (Gastropoda: Nudibranchia): ultrastructure and chemical analysis of the mantle dermal formations (MDFs). Mar Biol 106:245–250
Gavagnin M, Trivellone E, Castelluccio F, Cimino G, Cattaneo-Vietti R (1995) Glyceryl ester of a new halimane diterpenoic acid from the skin of the Antarctic nudibranch Austrodoris kerguelenensis. Tetrahedron Lett 36:7319–7322
Gavagnin M, Castelluccio F, Cimino G (1999a) Austrodorin-A and -B: first tricyclic diterpenoid 2′-monoglyceryl esters from an Antarctic nudibranch. Tetrahedron Lett 40:8471–8475
Gavagnin M, De Napoli A, Cimino G, Iken K, Avila C, Garcia FJ (1999b) Absolute configuration of diterpenoid diacylglycerols from the Antarctic nudibranch Austrodoris kerguelenensis. Tetrahedron Asymmetry 10:2647–2650. doi:10.1016/S0957-4166(99)00273-6
Gavagnin M, Mollo E, Castelluccio F, Ghiselin MT, Calado G, Cimino G (2001) Can molluscs biosynthesize typical sponge metabolites? The case of the nudibranch Doriopsilla areolata. Tetrahedron 57:8913–8916. doi:10.1016/S0040-4020(01)00876-6
Gavagnin M, Carbone M, Mollo E, Cimino G (2003a) Austrodoral and austrodoric acid: nor-sesquiterpenes with a new carbon skeleton from the Antarctic nudibranch Austrodoris kerguelenensis. Tetrahedron Lett 44:1495–1498
Gavagnin M, Carbone M, Mollo E, Cimino G (2003b) Further chemical studies on the Antarctic nudibranch Austrodoris kerguelenensis: new terpenoid acylglycerols and revision of the previous stereochemistry. Tetrahedron 59:5579–5583. doi:10.1016/S0040-4020(03)00775-0
Hayward P (1995) Antarctic cheilostomatous bryozoa. Oxford University Press, Oxford
Iken K, Avila C, Ciavatta ML, Fontana A, Cimino G (1998) Hodgsonal, a new drimane sesquiterpene from the mantle of the Antarctic nudibranch Bathydoris hodgsoni. Tetrahedron Lett 39:5635–5638. doi:10.1016/S0040-4039(98)01095-8
Iken K, Avila C, Fontana A, Gavagnin M (2002) Chemical ecology and origin of defensive compounds in the Antarctic nudibranch Austrodoris kerguelenensis (Opisthobranchia: Gastropoda). Mar Biol 141:101–109. doi:10.1007/s00227-002-0816-7
Lin Z, Torres JP, Ammon MA, Marett L, Teichert RW, Reilly CA, Kwan JC, Hughen RW, Flores M, Tianero MD, Peraud O, Cox JE, Light AR, Villaraza AJL, Haygood MG, Concepcion GP, Olivera BM, Schmidt EW (2013) A bacterial source for mollusk pyrone polyketides. Chem Biol 20:73–81. doi:10.1016/j.chembiol.2012.10.019.A
Martin R, Hild S, Walther P, Ploss K, Boland W, Tomaschko K-H (2007a) Granular chitin in the epidermis of nudibranch molluscs. Biol Bull 213:307–315
Martin R, Tomaschko K-H, Walther P (2007b) Protective skin structures in shell-less marine gastropods. Mar Biol 150:807–817. doi:10.1007/s00227-006-0402-5
Martin R, Heß M, Schrödl M, Tomaschko KH (2009) Cnidosac morphology in dendronotacean and aeolidacean nudibranch molluscs: from expulsion of nematocysts to use in defense? Mar Biol 156:261–268
McClintock JB, Baker BJ (1997a) A review of the chemical ecology of Antarctic marine invertebrates. Integr Comp Biol 37:329–342. doi:10.1093/icb/37.4.329
McClintock JB, Baker BJ (1997b) Palatability and chemical defense of eggs, embryos and larvae of shallow-water antarctic marine invertebrates. Mar Ecol 154:121–131
McClintock JB, Janssen J (1990) Pteropod abduction as a chemical defence in a pelagic antarctic amphipod. Nature 346:462–464. doi:10.1038/346462a0
McClintock JB, Baker BJ, Slattery M, Heine JN, Bryan PJ, Yoshida W, Davies-Coleman MT, Faulkner DJ (1994) Chemical defense of common Antarctic shallow-water nudibranch Tritoniella belli Eliot (Mollusca:Tritonidae) and its prey, Clavularia frankliniana Rouel (Cnidaria: Octocorallia). J Chem Ecol 20:3361–3372
McPhail KL, Davies-Coleman MT, Starmer J (2001) Sequestered chemistry of the Arminacean nudibranch Leminda millecra in Algoa Bay, South Africa. J Nat Prod 64:1183–1190
Moles J, Avila C, Kim I-H (2015a) Anthessius antarcticus n. sp. (Copepoda: Poecilostomatoida: Anthessidae) from Antarctic waters living in association with Charcotia granulosa (Mollusca: Nudibranchia: Charcotiidae). J Crustac Biol 35:97–104. doi:10.1163/1937240X-00002290
Moles J, Figuerola B, Campanyà N, Monleón-Getino T, Taboada S, Avila C (2015b) Distribution patterns in Antarctic and Subantarctic echinoderms. Polar Biol 38:799–813. doi:10.1007/s00300-014-1640-5
Morrill JB (1964) Protein content and dipeptidase activity of normal and cobalt-treated embryos of Limnaea palustris. Acta Embryol Morphol Exp 7:131–142
OBIS (2014) Global biodiversity indices from the ocean biogeographic information system. Intergovernmental Oceanographic Commission of UNESCO. http://www.iobis.org
Pika J, Faulkner DJ (1994) Four sesquiterpenes from the South African nudibranch Leminda millecra. Tetrahedron 50:3065–3070. doi:10.1016/S0040-4020(01)81106-6
Pola M, Gosliner TM (2010) The first molecular phylogeny of cladobranchian opisthobranchs (Mollusca, Gastropoda, Nudibranchia). Mol Phylogenet Evol 56:931–941. doi:10.1016/j.ympev.2010.05.003
Putz A, König GM, Wägele H (2010) Defensive strategies of Cladobranchia (Gastropoda, Opisthobranchia). Nat Prod Rep 27:1386–1402. doi:10.1039/b923849m
Putz A, Kehraus S, Díaz-Agras G, Wägele H, König GM (2011) Dotofide, a guanidine-interrupted terpenoid from the marine slug Doto pinnatifida (Gastropoda, Nudibranchia). Eur J Org Chem 20–21:3733–3737
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Rhoades DF, Gates RG (1976) Toward a general theory of plant antiherbivore chemistry. Recent Adv Phytochem 10:168–213
Schmekel L (1982) Vorkommen und Feinstruktur der Vakuolenepidermis von Nudibranchiern (Gastropoda Opisthobranchia). Malacologia 22:631–635
Shields CC, Marko PB, Woods HA, Moran AL (2009) Nudibranchs in the Ross Sea, Antarctica: lineage diversity and divergence estimated using methods of molecular phylogenetics and sequence divergence. Masters thesis, Clemson University, Clemson, South Carolina, 82 pp
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research. Freeman WH, New York
Vayssière A (1906) Mollusques nudibranches et Marséniadés. Expédition Antart. Française commendée par le Dr. Jean Charcot, pp 1–51
Wägele H (1989) On the morphology and ultrastucture of some egg-clutches of Antarctic nudibranchs (Gastropoda). Zool Anz 222:225–243
Wägele H (1991) The distribution of some endemic Antarctic Nudibranchia. Malacol Soc Lond 57:337–345
Wägele H (1996) On egg clutches of some Antarctic Opisthobranchia. Molluscan Reprod Malacol Rev Suppl 6:21–30
Wägele H (1997) Histological investigation of some organs and specialized cellular structures in Opisthobranchia (Gastropoda) with the potential to yield phylogenetically significant characters. Zool Anz 236:119–131
Wägele H (1998) Histological investigation of some organs and specialised cellular structures in Opisthobranchia (Gastropoda) with the potential to yield phylogenetically significant characters. Zool Anz 236:119–131
Wägele H, Klussmann-Kolb A (2005) Opisthobranchia (Mollusca, Gastropoda)—more than just slimy slugs. Shell reduction and its implications on defence and foraging. Front Zool 2:1–18. doi:10.1186/1742-9994-2-3
Wägele H, Willan R (2000) Phylogeny of the Nudibranchia. Zool J Linn Soc 130:83–181. doi:10.1006/zjls
Wägele H, Barnes DKA, Bullough LW (1995) Redescription of Charcotia granulosa Vayssière, 1906 (Nudibranchia: Arminoidea: Charcotiidae) from Signy Island, Antarctica. J Molluscan Stud 61:197–207
Wägele H, Ballesteros M, Avila C (2006) Defensive glandular structures in opisthobranch molluscs—from histology to ecology. Oceanogr Mar Biol 44:197–276
Wägele H, Klussmann-Kolb A, Verbeek E, Schrödl M (2014) Flashback and foreshadowing—a review of the taxon Opisthobranchia. Org Div Evol 14:133–149. doi:10.1007/s13127-013-0151-5
Wilson NG, Maschek JA, Baker BJ (2013) A species flock driven by predation? Secondary metabolites support diversification of slugs in Antarctica. PLoS One 8:e80277. doi:10.1371/journal.pone.0080277
Yoshida WY, Bryan PJ, Baker BJ, Mcclintock JB (1995) Pteroenone: a defensive metabolite of the abducted Antarctic pteropod Clione antarctica. J Org Chem 60:780–782
Zapata F, Wilson NG, Howison M, Andrade SCS, Jörger KM, Schrödl M, Goetz FE, Giribet G, Dunn CW (2014) Phylogenomic analyses of deep gastropod relationships reject Orthogastropoda. Proc R Soc B. doi:10.1098/rspb.2014.1739
Acknowledgments
We wish to thank J. Cristobo, C. Angulo-Preckler, M. Bas, B. Figuerola, L. Núñez-Pons, A. Riesgo, and S. Taboada for their help in the field work. We are indebted to the crews of the BIO-Las Palmas, the BIO-Hespérides, and the “Gabriel de Castilla” Spanish Antarctic Base, which provided logistic support during the ACTIQUIM-3 and 4 cruises. Special thanks are given to the ICB-NMR Service. A. García, R. Rivera, and A. Riesgo provided support in transmission electron microscopy. Funding was provided by the Spanish Government through the ACTIQUIM-II (CTM2010-17415/ANT) and DISTANTCOM (CTM2013-42667/ANT) projects, and by the Italian PNRA through the Research Project 2009/A1.06. J. M. was supported by a FPI of the Spanish Government (BES-2011-045325).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: F. Weinberger.
Reviewed by undisclosed experts.
Rights and permissions
About this article
Cite this article
Moles, J., Wägele, H., Cutignano, A. et al. Distribution of granuloside in the Antarctic nudibranch Charcotia granulosa (Gastropoda: Heterobranchia: Charcotiidae). Mar Biol 163, 54 (2016). https://doi.org/10.1007/s00227-016-2831-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-016-2831-0