Abstract
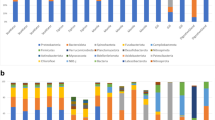
This work analyzes bacterial diversity of sediments transiting through the gut of Holothuria scabra which is an important bioturbator in tropical shallow waters. This edible holothurian species has a social and economic importance for coastal populations in many developing countries. Bacterial biodiversity was analyzed by sequencing the 16S rRNA of bacterial cultures and clones. DAPI and FISH methods were used to determine and compare the number of bacteria found in the various gut compartments. A total of 116 phylotypes belonging to the γ-Proteobacteria (60.5 %), α-Proteobacteria (24.5 %), Bacteroidetes (6 %), Actinobacteria (2.75 %), Fusobacteria (1.75 %), Firmicutes (1.75 %), Cyanobacteria (1.75 %) and δ-Proteobacteria (1 %) were identified. The number of bacteria is significantly greater (1.5×) in the foregut than in the ambient sediments. The number of bacteria significantly decreases in the midgut and remains stable until defecation. Some γ-Proteobacteria, especially Vibrio, are less affected by digestion than other bacterial taxa. The season has an impact on the bacterial diversity found in the sediments transiting through the gut: in the dry season, γ-Proteobacteria are the most abundant taxon, while α-Proteobacteria dominate in the rainy season. Vibrio is the most frequent genus with some well-known opportunistic pathogens like V. harveyi, V. alginolyticus and V. proteolyticus. Findings show that sediment-associated microbial communities are significantly modified by H. scabra during their transit through the gut which supports the view that holothurians play a substantial role in the structuring of bacterial communities at the sediment–seawater interface.




Similar content being viewed by others
References
Acinas SG, Sarma-Rupavtarm R, Klepac-Ceraj V, Polz MF (2005) PCR-induced sequence artifacts and bias: insights from comparison of two16SrRNA clone libraries constructed from the same sample. Appl Environ Microbiol 71:8966–8969
Amann RI, Binder BJ, Olson RJ, Chisholm SW, Devereux R, Stahl DA (1990) Combination of 16S rRNA-targeted oligonucleotide probes with flow cytometry for analyzing mixed microbial populations. Appl Environ Microbiol 56:1919–1925
Amaro T, Luna GM, Danovaro R, Billett DSM, Cunha MR (2012) High prokaryotic biodiversity associated with gut contents of the holothurian Molpadia musculus from the Nazare Canyon (NE Atlantic). Deep Sea Res 63:82–90
Amon RMW, Herndl GJ (1991) Deposit feeding and sediment: I. Interrelationship between Holothuria tubulosa (Holothuroidea, Echinodermata) and the sediment microbial community. Mar Ecol 12:163–174
Austin B (1989) Methods in aquatic bacteriology. Wiley, Chichester
Bakus GJ (1973) The biology and ecology of tropical holothurians. In: Jones OA, Endean R (eds) Biology and geology of coral reefs, vol 2. Academic Press, New York, pp 325–367
Bassler BN, Greenberg EP, Stevens AM (1997) Cross-species induction of luminescence in the quorum-sensing bacterium Vibrio harveyi. J Bacteriol 179:4043–4045
Becker P, Gillan D, Lanterbecq D, Jangoux M, Rasolofonirina R, Rakotovao J, Eeckhaut I (2004) The skin ulceration disease in cultivated juveniles of Holothuria scabra (Holothuroidea, Echinodermata). Aquaculture 242:13–30
Birkeland C (1988) The influence of echinoderms on coral-reef communities. Echinoderm Stud 3:1–79
Buchholz-Cleven BEE, Rattunde B, Straub KL (1997) Screening for genetic diversity of isolates of anaerobic Fe(II)-oxidizing bacteria using DGGE and whole-cell hybridization. Syst Appl Microbiol 20:301–309
Deming JW, Colwell RR (1982) Barophilic bacteria associated with digestive tracts of abyssal holothurians. Appl Environ Microbiol 44:1222–1230
Deming JW, Tabor PS, Colwell RR (1981) Barophilic growth of bacteria from intestinal tracts of deep-sea invertebrates. Microb Ecol 7:85–94
Dilmore LA, Hood MA (1986) Vibrios of some deep-water invertebrates. FEMS Microbiol Lett 35:221–224
Dunbar J, White S, Forney L (1997) Genetic diversity through the looking glass: effect of enrichment bias. Appl Environ Microbiol 63:1326–1331
Eeckhaut I, Lavitra T, Rasolofonirina R, Rabenevanana MW, Gildas P, Jangoux M (2009) Madagascar Holothurie SA: la première entreprise commerciale axée sur l’aquaculture des holothuries à Madagascar. Beche-de-mer Inf Bull 28:22–23
Epstein SS, Rossel J (1995) Enumeration of sandy sediment bacteria: search for optimal protocol. Mar Ecol Prog Ser 117:289–298
Eriksson H, Robinson G, Slater MJ, Troell M (2012) Sea cucumber aquaculture in the Western Indian Ocean: challenges for sustainable livelihood and stock improvement. Ambio 41:109–121
Féral J-P (1980) Cuticule et bactéries associées des épidermes digestif et tégumentaire de Leptosynaopta gallieninei (Herapath) (Holothurioidea: Apoda)-premières données. In: Jangoux M (ed) Echinoderms: present and past. AA Balkema, Rotterdam, pp 285–290
Féral J-P, Massin C (1982) Digestive systems: Holothuroidea, In: Jangoux M, Lawrence JM (eds) Echinoderm Nutrition AA Balkema, Rotterdam, p 191–212
Gillan DC, Pernet P (2007) Adherent bacteria in heavy metal contaminated marine sediments. Biofouling 23:1–13
Gillan DC, Danis B, Pernet P, Joly G, Dubois P (2005) Structure of sediment-associated microbial communities along a heavy-metal contamination gradient in the marine environment. Appl Environ Microbiol 71:679–690
Hamel J-F, Conand C, Pawson DL, Mercier A (2001) The sea cucumber Holothuria scabra (Holothuroidea: Echinodermata): its biology and exploitation as Bêche-de-mer. Adv Mar Biol 41:129–223
Hammond LS (1983) Nutrition of deposit-feeding holothurians and echinoids (Echinodermata) from a shallow reef lagoon, Discovery Bay, Jamaica. Mar Ecol Prog Ser 10:297–305
Harris JM (1993) The presence, nature, and role of gut microflora in aquatic invertebrates: a synthesis. Microb Ecol 25:195–231
Hatmanti A, Purwati P (2011) Bacteria associated holothurians: the key of habitat preference, diet and functions. Jurnal Ilmu dan Teknologi Kelautan Tropis 3:73–81
Huber T, Faulkner G, Hugenholtz P (2004) Bellerophon: a program to detect chimeric sequences in multiple sequence alignments. Bioinformatics 20:2317–2319
Khripounoff A, Sibuet M (1980) La nutrition d’échinodermes abyssaux I. Alimentation des holothuries. Mar Biol 60:17–26
Kirchman D, Sigda J, Kapuscinski R, Mitchell R (1982) Statistical analysis of the direct count method for enumeration bacteria. Appl Environ Microbiol 44:376–382
Lavitra T, Rasolofonirina R, Eeckhaut I (2010) The effect of sediment quality and stocking density on survival and growth of the sea cucumber Holothuria scabra reared in nursery ponds and sea pens. WIOJMS 9(2):153–164
Léonet A, Rasolofonirina R, Wattiez R, Jangoux M, Eeckhaut I (2009) A new method to induce oocyte maturation in holothuroids (Echinodermata). Invertebr Reprod Dev 53:13–21
Liu P-C, Lee K–K, Chen S-N (1996) Pathogenicity of different isolates of Vibrio harveyi in tiger prawn, Penaeus monodon. Lett Appl Microbiol 22:413–416
Mangion P, Taddei D, Frouin P, Conand C (2004) Feeding rate and impact of sediment reworking by two deposit feeders Holothuria leucospilota and Holothuria atra on fringing reef (Reunion Island, Indian Ocean). In: Heinzeller TT, Nebelsick J (eds) Echinoderms: München, Taylor and Francis Group, London, p 311–317
Manz W, Amann R, Ludwig W, Wagner M, Schleifer KH (1992) Phylogenetic oligonucleotide probes for the major subclasses of Proteobacteria: problems and solutions. Syst Appl Microbiol 15:593–600
Manz W, Amann R, Ludwig W, Vancanney M, Schleifer KF (1996) Application of a suite of 16S rRNA-specific oligonucleotide probes designed to investigate bacteria of the phylum cytophaga-flavobacter-bacteriodes in the natural environment. Microbiology 142:1097–1106
Manz W, Eisenbrecher M, Neu TR, Szewzyk U (1998) Abundance and spatial organization of gram negative sulfate-reducing bacteria in activated sludge investigated by in situ probing with specific 16S rRNA targeted oligonucleotides. FEMS Microbiol Ecol 25:43–61
Massin C (1982) Effects of feeding on the environment: Holothuroidea. In: Jangoux M, Lawrence JM (eds) Echinoderm nutrition. AA Balkema, Rotterdam, pp 493–497
McLaughlin J, DePaola A, Bopp CA, Martinek KA, Napolilli NP, Allison CG, Murray SL, Thompson EC, Bird MM, Middaugh MD (2005) Outbreak of Vibrio parahaemolyticus gastroenteritis associated with Alaskan oysters. N Eng J Med 353:1463–1470
Mezzali K, Soualili D L (2013) The ability of holothurians to select sediment particles and organic matter. Bêche-de-Mer Inf Bull 33:38–43
Molitoris E, Joseph SW, Krichevsky MI, Sindhuhardja W, Colwell RR (1985) Characterization and distribution of Vibrio alginolyticus and Vibrio parahaemolyticus isolated in Indonesia. Appl Environ Microbiol 50:1388–1394
Montagna PA (1982) Sampling design and enumeration statistics for bacteria extracted from marine sediment. Appl Environ Microbiol 43:1366–1372
Moriarty DJW (1982) Feeding of Holothuria atra and Stichopus chloronotus on bacteria, organic carbon and organic nitrogen in sediments of the Great Barrier Reef. Aust J Mar Freshw Res 33:255–263
Moriarty DJW, Pollard PC, Hunt WG, Moriarty CM, Wassenberg TJ (1985) Productivity of bacteria and microalgae and the effect of grazing by holothurians in sediments on a coral reef flat. Mar Biol 85:293–300
Odintsov VS (1981) Nitrogen fixation (acetylene reduction) in the digestive tract of some echinoderms from Vostok Bay in the Sea of Japan. Mar Biol Lett 2:259–263
Plotieau (2012) Analyses de certains éléments nutritionels essentiels à Holothuria scabra (Echinodermata, Holothuroidea): influence de la qualité du sédiment sur le développement des holothuries en aquaculture et importance des bactéries. Phd Thesis. University of Mons, Mons, Belgium
Purcell SW (2004) Rapid growth and bioturbation activity of the sea cucumber Holothuria scabra in earthen ponds. In: Proceedings of Australasian Aquaculture 2004, Sydney, Australia, p 244
Purcell SW, Mercier A, Conand C, Hamel J-F, Toral-Granda MV, Lovatelli A, Uthicke S (2013) Sea cucumber fisheries: global analysis of stocks, management measures and drivers of overfishing. Fish Fish 14(1):34–59
Ravenschlag K, Sahm K, Amann R (2001) Quantitative molecular analysis of the microbial community in marine Arctic sediments (Svalbard). Appl Environ Microbiol 67:387–395
Robinson G, Pascal B (2009) De l’écloserie au village—Premier programme communautaire d’holothuriculture à Madagascar. Bêche-de-mer Inf Bull 29:37–41
Robinson G, Pascal B (2012) Sea cucumber farming experiences in south-western Madagascar. In: Hair CA, Pickering TD, Mills DJ (eds) Proceedings of an international symposium. Noumea, New Caledonia, pp 40–49
Schneider K, Silverman J, Woolsey E, Eriksson H, Byrne M, Caldeira K (2011) Potential influence of sea cucumbers on coral reef CaCO3 budget: a case study at One Tree Reef. J Geophys Res. doi:10.1029/2011JG001755
So JJ, Hamel J-F, Mercier A (2010) Habitat utilisation, growth and predation of Cucumaria frondosa: implications for an emerging sea cucumber fishery. Fish Manag Ecol 17:473–484
Su YC, Liu C (2007) Vibrio parahaemolyticus: a concern of seafood safety. Food Microbiol 24:549–558
Swofford D (1998) PAUP*. Phylogenetics analysis using parsimony (*and others methods). Version 4.0b10. Sinauer Associates, Sunderland, Massachussetts
Taddéi D (2006) Transfert de matière et d’énergie dans les sédiments d’un complexe récifal anthropisé (Ile de la réunion, Océan Indien). PhD thesis, Université de la Réunion
Teske A, Wawer C, Muyzer G, Ramsing NB (1996) Distribution of sulfate-reducing bacteria in a stratified fjord (Mariager Fjord, Denmark) as evaluated by most-probable-number counts and denaturing gradient gel electrophoresis of PCR-amplified ribosomal DNA fragments. Appl Environ Microbiol 62:1405–1415
Uthicke S (1999) Sediment bioturbation and impact of feeding activity of Holothuria (Halodeima) atra and Stichopus chloronotus, two sediment feeding holothurians, at Lizard island, Great Barrier Reef. Bull Mar Sci 64:129–141
Uthicke S (2001) Interactions between sediment-feeders and microalgae on coral reefs: grazing losses versus production enhancement. Mar Ecol Prog Ser 210:125–138
Uthicke S, Klumpp DW (1998) Microphytobenthos community production at a near-shore coral reef: seasonal variation and response to ammonium recycled by holothurian. Mar Ecol Prog Ser 169:1–11
Wallner G, Amann R, Beisker W (1993) Optimizing fluorescent in situ-hybridization with rRNA-targeted oligonucleotide probes for flow cytometric identification of microorganisms. Cytometry 14:136–143
Ward-Rainey N, Rainey FA, Stackebrandt E (1996) A study of the bacterial flora associated with Holothuria atra. J Exp Mar Bio Ecol 203:11–26
Wolkenhauer SM, Uthicke S, Burridge C, Skewes T, Pitcher R (2010) The ecological role of Holothuria scabra (Echinodermata: Holothuroidea) within subtropical seagrass beds. J Mar Biol Assoc UK 90:215–223
Wright ES, Yilmaz LS, Noguera DR (2012) Methods: DECIPHER, a search-based approach to chimera identification for 16s rRNA sequences. Appl Environ Microbiol 78:717–725
Yingst J (1976) The utilization of organic matter in shallow marine sediments by an epibenthic deposit-feeding holothurian. J Exp Mar Bio Ecol 23:55–69
Acknowledgments
We wish to thank the FNRS (Fonds National pour la Recherche Scientifique), CUD (Commission Universitaire pour le Développement) and AMPA (Agence Malgache de la Pêche et de l’Aquaculture) for financing assignments in Madagascar. T.P. benefited from a doctoral grant of the FRIA (Fonds pour la formation à la Recherche dans l’Industrie et l’Agriculture). This work is a contribution of the CIBIM (Centre Interuniversitaire de Biologie Marine) and of the PRU (Polyaquaculture Research Unit). We thank very much Jean-François Hamel and Annie Mercier for checking the English and two anonymous reviewers for their useful comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. kuhl.
Electronic supplementary material
Below is the link to the electronic supplementary material.
227_2013_2297_MOESM1_ESM.eps
Fig. 1S. Distance tree showing the relationships among the 16S rRNA clones and isolates of the γ-Proteobacteria obtained in this study. The bacteria indicated by names in the tree are those whose similarity is higher than 99 % during a blast search. Green nonsulfur bacterium served as the outgroup. A matrix of 121 OTUs was used. GenBank accession numbers are listed for the close relatives of the clones and isolates. Bootstrap values are shown above branches (1000 replicates). The origin of bacteria are indicated in brackets: (SW) = seawater, (S) = substrate, (F) = foregut, (M) = midgut, (H) = hindgut and (Fa) = faeces. (EPS 36204 kb)
227_2013_2297_MOESM2_ESM.eps
Fig. 2S. Distance tree showing the relationships among the 16S rRNA clones and isolates of Cyanobacteria, Actinobacteria, Firmicutes, α-Proteobacteria, δ-Proteobacteria, Fusobacteria and Bacteroidetes obtained in this study. The bacteria indicated by names in the tree are those whose similarity is higher than 99 % during a blast search. Green nonsulfur bacterium served as the outgroup. A matrix of 76 OTUs was used. GenBank accession numbers are listed for the close relatives of the clones and isolates. Bootstrap values are shown above branches (1000 replicates). The origin of bacteria are indicated in brackets: (SW) = seawater, (S) = substrate, (F) = foregut, (M) = midgut, (H) = hindgut and (Fa) = faeces. (EPS 9263 kb)
Rights and permissions
About this article
Cite this article
Plotieau, T., Lavitra, T., Gillan, D.C. et al. Bacterial diversity of the sediments transiting through the gut of Holothuria scabra (Holothuroidea; Echinodermata). Mar Biol 160, 3087–3101 (2013). https://doi.org/10.1007/s00227-013-2297-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-013-2297-2