Abstract
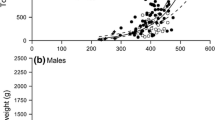
The gonad index, GI, is widely used as a measure of changes in reproductive state. There are, however, problems with its use because it is based on the implicit assumption of an isometric relationship between gonad size and some measure of total size. If, for example, gonad weight and total weight are used, the exponent for an allometric relationship usually is ignored and hence assumed to be 1.0. It is further assumed that this exponent is fixed for all states of the reproductive cycle and that gonads begin to develop at size = 0. Data for the purple sea urchin Strongylocentrotus purpuratus at Gregory Point, Oregon, USA, gathered over a period of 31 months showed that these assumptions cannot be supported. The relationship is better modeled with a function that (1) takes into account size of initial gonad production and (2) allows allometric exponents that vary with site or season. Thus, a better approach is to use a wide range of sizes to estimate size when gonads begin to develop and then, with this correction, ANCOVA to test for differences of gonad size among samples. Gonad changes at Gregory Point were estimated using fixed sizes of 5 cm diameter and 60 g total weight. Publishing means for X and Y, the standard error of the estimate, R 2, and slope for each regression are shown to be sufficient to compare our results with results across studies.






Similar content being viewed by others
References
Atchley WR, Gaskins CT, Anderson D (1976) Statistical properties of ratios. I. Empirical results. Syst Zool 25:137–148. doi:10.2307/2412740
Barker MF, Xu RA (1991) Population differences in gonad and pyloric caeca cycles of the New Zealand seastar Sclerasterias mollis (Echinodermata: Asteroidea). Mar Biol 108:97–103. doi:10.1007/BF01320228
Beaupre SJ, Dunham AE (1995) A comparison of ratio-based and covariance analyses of a nutritional data set. Funct Ecol 9:876–880
Bennett J, Giese AC (1955) The annual reproductive and nutritional cycles in two western sea urchins. Biol Bull 109:226–237
Bernard FR (1977) Fishery and reproductive cycle of the Red Sea urchin, Strongylocentrotus franciscanus, in British Columbia. J Fish Res Bd Can 34:604–610
Bigatti, G, Marzinelli EM, Cledón M, Penchaszadeh PE (2004) Gonadal cycle of Pseudechinus magellanicus (Philippi, 1857) (Echinoidea: Temnopleuridae) from Patagonia, Argentina. In: Heinzeller T, Nebelsick JH (eds) Echinoderms: München. Proceedings, 11th international echinoderm conference, A. A. Balkema, Leiden, The Netherlands, pp 11–14
Boivin Y, Larrivée D, Himmelman JH (1986) Reproductive cycle of the subarctic brooding asteroid Leptasterias polaris. Mar Biol 92:329–337. doi:10.1007/BF00392673
Bourgoin A, Guillou M (1990) Variations in the reproductive cycle of Acrocinida brachiat a (Echinodermata: Ophiuroidea) according to environment in the Bay of Douarnenez (Brittany). J Mar Biol Ass UK 70:57–66. doi:10.1017/S0025315400034196
Byrne M (1992) Reproduction of sympatric populations of Patiriella gunnii, P. calcar and P. exigua in New South Wales, asterinid sea stars with direct development. Mar Biol 114:297–316. doi:10.1007/BF00349533
Byrne M, Morrice MG, Wolf B (1997) Introduction of the northern Pacific asteroid Asterias amurensis to Tasmania: reproduction and current distribution. Mar Biol 127:673–685. doi:10.1007/s002270050058
Byrne M, Andrew NL, Worthington DG, Brett PA (1998) Reproduction in the diadematoid sea urchin Centrostephanus rodgersii in contrasting habitats along the coast of New South Wales, Australia. Mar Biol 132:305–318. doi:10.1007/s002270050396
Cameron JL, Fankboner PV (1986) Reproductive biology of the commercial sea cucumber Parastichopus californicus (Stimpson) (Echinodermata: Holothuroidea). I. Reproductive periodicity and spawning behavior. Can J Zool 64:168–175. doi:10.1139/z86-027
Chadwick-Furman NE, Spiegel M, Nir I (2000) Sexual reproduction in the tropical corallimorpharian Rhodactis rhodostoma. Invert Biol 119:361–369
Chao S-M, Chen C-P, Alexander PS (1995) Reproductive cycles of tropical sea cucumbers (Echinodermata: Holothuroidea) in southern Taiwan. Mar Biol 122:289–295. doi:10.1007/BF00348942
Chen B-Y, Chen C-P (1992) Reproductive cycle, larval development, juvenile growth and population dynamics of Patiriella psudoexigua (Echinodermata: Asteroidea) in Taiwan. Mar Biol 113:271–280. doi:10.1007/BF00347281
Christians JK (1999) Controlling for body mass effects: is part-whole correlation important? Physiol Biochem Zool 72:250–253. doi:10.1086/316661
Costa PAS, Fernandes FC (1993) Reproductive cycle of Loligo sanpaulensis (Cephalopoda: Loliginidae) in the Cabo Frio region, Brazil. Mar Ecol Prog Ser 101:91–97
Damián GG, Zaixso HE, Tolosano JA (2009) Brooding of the sub-Antarctic heart urchin, Abatus cavernosus (Spatangoida: Schizasteridae), in southern Patagonia. Mar Biol 156:1647–1657. doi:10.1007/s00227-009-1200-7
de Vlaming V, Grossman G, Chapman F (1982) On the use of the gonosomatic index. Comp Biochem Physiol A 73:31–39. doi:10.1016/0300-9629(82)90088-3
Diez MJ, Lovrich GA (2010) Reproductive biology of the crab Halicarcinus planatus (Brachyura, Hymenosomatidae) in sub-Antarctic waters. Polar Biol doi:10.1007/s00300-009-0716-0
Ebert TA (1968) Growth rates of the sea urchin Strongylocentrotus purpuratus related to food availability and spine abrasion. Ecology 49:1075–1091. doi:10.2307/1934491
Ebert TA (1975) Growth and mortality of post-larval echinoids. Am Zool 15:755–775
Ebert TA (1998) An analysis of the importance of Allee effects in management of the red sea urchin Strongylocentrotus franciscanus. In: Mooi R, Telford M (eds) Echinoderms: San Francisco. Proceedings, 9th international echinoderm conference, A. A. Balkema, Brookfield, Vermont, pp 619–627
Ebert TA (2007) Growth and survival of postsettlement sea urchins. In: Lawrence JM (ed) Edible sea urchins: biology and ecology. Developments in aquaculture and fisheries science, vol 37. Elsevier, Amsterdam, pp 95–134
Ebert TA (2008) Longevity and lack of senescence in the red sea urchin Strongylocentrotus franciscanus. Exp Gerontol 43:734–738. doi:10.1016/j.exger.2008.04.015
Ebert TA (2010a) Demographic patterns of the purple sea urchin Strongylocentrotus purpuratus along a latitudinal gradient, 1985–1987. Mar Ecol Prog Ser 406:105–120. doi:10.3354/meps08547
Ebert TA (2010b) Potential use of production and biomass for life-history comparisons of sea urchins. Proceedings 13th international echinoderm conference, Hobart, Tasmania (in press)
Ebert TA, Russell MP (1994) Allometry and model II nonlinear regression. J Theor Biol 168:367–372
Ford CE (1964) Reproduction in the aggregating sea anemone Anthopleura elegantissima. Pac Sci 18:138–145
Giese AC (1966) On the biochemical constitution of some echinoderms. In: Boolootian RA (ed) Physiology of Echinodermata. Interscience Publishers (Wiley), New York, pp 757–796
Giese AC (1967) Changes in body-component indexes and respiration with size in the purple sea urchin Strongylocentrotus purpuratus. Physiol Zool 40:194–200
Giese AC, Greenfield L, Huang H, Farmanfarmaian A, Boolootian R, Lasker R (1959) Organic productivity in the reproductive cycle of the purple sea urchin. Biol Bull 116:49–58
Gonor JJ (1972) Gonad growth in the sea urchin, Strongylocentrotus purpuratus (Stimpson) (Echinodermata: Echinoidea) and the assumptions of gonad index methods. J Exp Mar Biol Ecol 10:89–103. doi:10.1016/0022-0981(72)90095-0
Guettaf M, San Martin GA, Francour P (2000) Interpopulation variability of the reproductive cycle of Paracentrotus lividus (Echinodermata: Echinoidea) in the south-western Mediterranean. J Mar Biol Ass UK 80:899–907. doi:10.1017/S0025315400002885
Guillou M, Lumingas LJL (1998) The reproductive cycle of the ‘blunt’ sea urchin. Aquacul Internat 6:147–160. doi:10.1023/A:1009290307840
Guillou M, Lumingas LJL (1999) Variation in the reproductive strategy of the sea urchin Sphaerechinus granularis (Echinodermata: Echinoidea) related to food availability. J Mar Biol Ass UK 79:131–136. doi:10.1017/S0025315498000149
Gutt J, Gerdes D, Klages M (1992) Seasonality and spatial variability in the reproduction of two Antarctic holothurians (Echinodermata). Polar Biol 11:533–544. doi:10.1007/BF00237946
Hatfield EMC, Murray AWA (1999) Objective assessment of maturity in the Patagonian squid Loligo gahi (Cephalopoda: Loliginidae) from Falkland Islands waters. ICES J Mar Sci 56:746–756. doi:10.1006/jmsc.1999.0514
Hines AH (1979) Effects of thermal discharge on reproductive cycles in Mytilus edulis and Mytilus californianus (Mollusca, Bivalvia). Fish Bull 77:498–503
Hughes AD, Kelly MS, Barnes DKA, Catarino AI, Black KD (2006) The dual functions of sea urchin gonads are reflected in the temporal variations of their biochemistry. Mar Biol 148:789–798. doi:10.1007/s00227-005-0124-0
Kato S, Schroeter SC (1985) Biology of the red sea urchin, Strongylocentrotus franciscanus, and its fishery in California. Mar Fish Rev 47:1–20
Kelly MS, Owen PV, Pantazis P (2001) The commercial potential of the common sea urchin Echinus esculentus from the west coast of Scotland. Hydrobiol 465:85–94. doi:10.1023/A:1014553010711
Kirby S, Lamare MD, Barker MF (2006) Growth and morphometrics in the New Zealand sea urchin Pseudechinus huttoni (Echinoidea: Temnopleuridae). NZ J Mar Freshwater Res 40:413–428. doi:0028-8330/06/4003-0413
Kramer DE, Nordin DMA (1975) Physical data from a study of size, weight and gonad quality for the red sea urchin [Strongylocentrotus franciscanus (Agassiz)] over a one-year period. Fish Res Bd Can Manuscript Rpt Ser No 1372, 91 pp
Laegdsgaard P, Byrne M, Anderson DT (1991) Reproduction of sympatric populations of Heliocidaris erythrogramma and H. tuberculata (Echinoidea) in New South Wales. Mar Biol 110:359–374. doi:10.1007/BF01344355
Lau DCC, Lau SCK, Qian P-Y, Qiu J-W (2009) Morphological plasticity and resource allocation in response to food limitation and hyposalinity in a sea urchin. J Shellfish Res 28:383–388
Lawrence JM (1973) Level, content, and caloric equivalents of the lipid, carbohydrate, and protein in the body components of Luidia clathrata (Echinodermata: Asteroidea: Platyasterida) in Tampa Bay. J Exp Mar Biol Ecol 11:263–274. doi:10.1016/0022-0981(73)90026-9
Lawrence JM, Byrne M (1994) Allocation of resources to body components in Heliocidaris erythrogramma and Heliocidaris tuberculata (Echinodermata: Echinoidea). Zool Sci 11:133–137
Lawrence JM, Lawrence AL, Holland ND (1965) Annual cycle in the size of the gut of the purple sea urchin, Strongylocentrotus purpuratus (Stimpson). Nature 205:1238–1239. doi:10.1038/2051238a0
Lefebvre A, Davoult D, Gentil F, Janquin MA (1999) Spatio-temporal variability in the gonad growth of Ophiothrix fragilis (Echinodermata: Ophiuroidea) in the English Channel and estimation of carbon and nitrogen outputs towards the pelagic system. Hydrobiologia 414:25–34. doi:10.1023/A:1003827532385
Lessios HA (1981) Reproductive periodicity of the echinoids Diadema and Echinometra on the two coasts of Panama. J Exp Mar Biol Ecol 50:47–61. doi:10.1016/0022-0981(81)90062-9
Lester SE, Gaines SD, Kinlan BP (2007) Reproduction on the edge: large-scale patterns of individual performance in a marine invertebrate. Ecology 88:2229–2239. doi:10.1890/06-1784.1
Lewis JB (1958) The biology of the tropical sea urchin Tripneustes esculentus Leske in Barbados, British West Indies. Can J Zool 36:607–621
Lewis JB (1966) Growth and breeding in the tropical echinoid Diadema antillarum Phillipi. Bull Mar Sci 16:151–158
Lewis JB, Storey GS (1984) Differences in morphology and life history traits of the echinoid Echinometra lucunter from different habitats. Mar Ecol Prog Ser 15:207–211
Liermann M, Steel A, Rosingard M, Guttorp P (2004) Random denominators and the analysis of ratio data. Envir Ecol Stat 11:55–71. doi:10.1023/B:EEST.0000011364.71236.f8
Liu JH (1994) The ecology of the Hong Kong limpets Cellana grata (Gould 1859) and Patelloida pygmaea (Dunker 1860): reproductive biology. J Moll Stud 60:97–111
Lozano J, Galera J, López S, Turon X, Palacin C, Morera G (1995) Biological cycles and recruitment of Paracentrotus lividus (Echinodermata: Echinoidea) in two contrasting habitats. Mar Ecol Prog Ser 122:179–191. doi:10.3354/meps122179
MacCord FS, Ventura CRR (2004) Reproductive cycle of the endemic cassiduloid Cassidulus mitis (Echinoidea: Cassiduloida) on the Brazilian coast. Mar Biol 145:603–612. doi:10.1007/s00227-004-1344-4
Marsh AG, Watts SA (2007) Biochemical and energy requirements of gonad development. In: Lawrence JM (ed) Edible sea urchins: biology and ecology. Developments in aquaculture and fisheries science, vol 37. Elsevier, Amsterdam, pp 35–53
McCarthy DA, Young CM (2002) Gametogenesis and reproductive behavior in the echinoid Lytechinus variegatus. Mar Ecol Prog Ser 233:157–168. doi:10.3354/meps233157
McPherson BF (1965) Contributions to the biology of the sea urchin Tripneustes ventricosus. Bull Mar Sci 15:228–244
Moore HB, Jutare T, Jones JA, McPherson BF, Roper CFE (1963) A contribution to the biology of Tripneustes esculentus. Bull Mar Sci Gulf Carib 13:267–281
Nicastro KR, Zardi GI, McQuaid CD (2010) Differential reproductive investment, attachment strength and mortality of invasive and indigenous mussels across heterogeneous environments. Biol Invasions doi:10.1007/s10530-009-9619-9
Nichols D, Barker MF (1984) Reproductive and nutritional periodicities in the starfish, Marthasterias glacialis, from Plymouth Sound. J Mar Biol Ass UK 64:461–470. doi:10.1017/S0025315400030137
Nojima S (1979) Ecological studies of a sea star, Astropecten latespinosus Meissner. I. Survivorship curve and life history. Publ Amakusa Mar Biol Lab 5:45–65
Olive PJW, Porter JS, Sandeman NJ, Wright NH, Bentley MG (1997) Variable spawning success of Nepthys hombergi (Annelida: Polychaeta) in response to environmental variation a life history homeostasis? J Exp Mar Biol Ecol 215:247–268. doi:10.1016/S0022-0981(97)00047-6
Ong Che RG, Gomez ED (1985) Reproductive periodicity of Holothuria scabra jaeger at Calatagan, Batangas, Philippines. Asian Mar Biol 2:21–30
Packard GC, Boardman TJ (1987) The misuse of ratios to scale physiological data that vary allometrically with body size. In: Feder ME, Bennett AF, Burggren WW, Huey RB (eds) New directions in ecological physiology. Cambridge University Press, Cambridge, pp 216–239
Packard GC, Boardman TJ (1999) The use of percentages and size-specific indices to normalize physiological data for variation in body size: wasted time, wasted effort? Comp Biochem Physiol A 122:37–44
Pearse JS (1968) Patterns of reproductive periodicities in four species of Indo-Pacific echinoderms. Proc Indian Acad Sci B 68:247–279
Pérez-González R, Puga-López D, Castro-Longoria R (2009) Ovarian development and size at sexual maturity of the Mexican spiny lobster Panulirus inflatus. NZ J Mar Freshwater Res 43:163–172. doi:10.1080/00288330909509990
Quackenbush LS, Herrnkind WF (1981) Regulation of molt and gonadal development in the spiny lobster, Panulirus argus (Crustacea: Palinuridae): effect of eyestalk ablation. Comp Biochem Physiol A 69:523–627. doi:10.1016/0300-9629(81)93014-0
Raubenheimer D, Simpson SJ (1992) Analysis of covariance: an alternative to nutritional indices. Entomol Exp Appl 62:221–231
Raymond BG, Scheibling RE (1987) Recruitment and growth of the sea urchin Strongylocentrotus droebachiensis (Muller) following mass mortalities off Nova Scotia, Canada. J Exp Mar Biol Ecol 108:31–54. doi:10.1016/0022-0981(87)90129-8
Robb RC (1929) On the nature of hereditary size-limitation. II. The growth of parts in relation to the whole. Brit J Exp Bio 6:311–324
Russell MP (1998) Resource allocation plasticity in sea urchins: rapid, diet induced, phenotypic changes in the green sea urchin, Strongylocentrotus droebachiensis (Müller). J Exp Mar Biol Ecol 220:1–14. doi:10.1016/S0022-0981(97)00079-8
Selden R, Johnson AS, Ellers O (2009) Waterborne cues from crabs induce thicker skeletons, smaller gonads and size-specific changes in growth rate in sea urchins. Mar Biol 156:1057–1071. doi:10.1007/s00227-009-1150-0
SYSTAT (2004) SYSTAT 11. SYSTAT Software, Inc, Richmond, CA
Town JC (1980) Movement, morphology, reproductive periodicity, and some factors affecting gonad production in the seastar, Asterstole scabra (Hutton). J Exp Mar Biol Ecol 44:111–132. doi:10.1016/0022-0981(80)90149-5
Vadas RL Sr, Beal B, Dowlin T, Fegley JC (2000) Experimental field tests of natural algal diets on gonad index and quality in the green sea urchin, Strongylocentrotus droebachiensis: a case for rapid summer production in post-spawned animals. Aquaculture 182:115–135. doi:10.1016/S0044-8486(99)00254-9
Vernon JD, McClintock JB, Hopkins TS, Watts SA, Marion KR (1993) Reproduction of Clypeaster ravenelii (Echinodermata: Echinoidea) in the northern Gulf of Mexico. Invert Reprod Dev 24:71–78
Zar JH (1974) Biostatistical analysis. Prentice-Hall, Inc., Englewood Cliffs, NJ
Acknowledgments
Dissections of sea urchins from Gregory Point were done by B. Miller. Those from Arena Cove, California, were done by M. Morris and N. Sandoval. Assistance with dissections from Yaquina Head, Oregon, was by C. Ebert. The manuscript benefited from comments made by 3 anonymous reviewers. Funding for this work was from the Ocean Sciences Division Biological Oceanography of the US National Science Foundation (grants OCE 84-01415 and OCE-0623934). We are grateful for all of this support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Byrne.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ebert, T.A., Hernandez, J.C. & Russell, M.P. Problems of the gonad index and what can be done: analysis of the purple sea urchin Strongylocentrotus purpuratus . Mar Biol 158, 47–58 (2011). https://doi.org/10.1007/s00227-010-1541-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-010-1541-2