Abstract
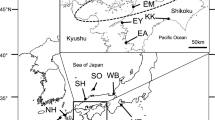
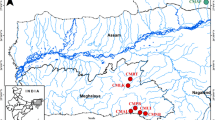
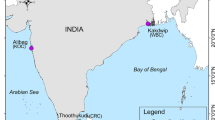
Crassostrea ariakensis is an important aquacultured oyster species in Asia, its native region. During the past decade, consideration was given to introducing C. ariakensis into Chesapeake Bay, in the United States, to help revive the declining native oyster industry and bolster the local ecosystem. Little is known about the ecology and biology of this species in Asia due to confusion with nomenclature and difficulty in accurately identifying the species of wild populations in their natural environment. Even less research has been done on the population genetics of native populations of C. ariakensis in Asia. We examined the magnitude and pattern of genetic differentiation among 10 wild populations of C. ariakensis from its confirmed distribution range using eight polymorphic microsatellite markers. Results showed a small but significant global θST (0.018), indicating genetic heterogeneity among populations. Eight genetically distinct populations were further distinguished based on population pairwise θST comparisons, including one in Japan, four in China, and three populations along the coast of South Korea. A significant positive association was detected between genetic and geographic distances among populations, suggesting a genetic pattern of isolation by distance. This research represents a novel observation on wild genetic population structuring in a coastal bivalve species along the coast of the northwest Pacific.



Similar content being viewed by others
References
Aranishi F (2006) A novel mitochondrial intergenic spacer reflecting population structure of Pacific oyster. J Appl Genet 47:119–123
Arnaud-Haond S, Bonhomme F, Blanc F (2003) Large discrepancies in differentiation of allozymes, nuclear and mitochondrial DNA loci in recently founded Pacific populations of the pearl oyster Pinctada margaritifera. J Evol Biol 16:388–398
Arnaud-Haond S, Vonau V, Rouxel C, Bonhomme F, Prou J, Goyard E, Boudry P (2008) Genetic structure at different spatial scales in the pearl oyster (Pinctada margaritifera cumingii) in French Polynesian lagoons: be aware of sampling strategy and genetic patchiness. Mar Biol 155:147–157
Avise JC (1994) Molecular makers, natural history, and evolution. Chapman and Hall, New York
Barber PH, Palumbi SR, Erdmann MV, Moosa MK (2002) Sharp genetic breaks among populations of Haptosquilla pulchella (Stomatopoda) indicate limits to larval transport: patterns, causes, and consequences. Mol Ecol 11:659–674
Belkhir K, Borsa P, Chikhi L, Raufaste N, Bonhomme F (1996–2004) GENETIX 4.05, logiciel sous Windows TM pour la génétique des populations. Laboratoire Génome, populations, interactions, CNRS UMR 5171, Université de Montpellier II, Montpellier (France)
Boudry P, Heurtebise S, Lapègue S (2003) Mitochondrial and nuclear DNA sequence variation of presumed Crassostrea gigas and Crassostrea angulata specimens: a new oyster species in Hong Kong? Aquaculture 228:15–25
Cavalli-Sforza LL, Edwards AW (1967) Phylogenetic analysis models and estimation procedures. Evolution 21:550–570
Chen C, Beardsley RC, Limeburner R, Kim K (1994) Comparison of winter and summer hydrographic observations in the Yellow and East China Seas and adjacent Kuroshio during 1986. Cont Shelf Res 14:909–929
Chen CA, Ablan MCA, McManus JW, Bell JD, Tuan VS, Cabanban AS, Shao K (2006) Population structure and genetic variability of Six Bar Wrasse (Thallasoma Hardwicki) in northern South China Sea revealed by mitochondrial control region sequences. Mar Biotechnol 6:312–326
Cordes JF, Xiao J, Reece KS (2008) Discrimination of nine Crassostrea oyster species based on restriction fragment-length polymorphism (RFLP) analysis of nuclear and mitochondrial DNA markers. J Shellfish Res 27:1155–1162
Crandall ED, Frey MA, Grosberg RK, Barber PH (2008) Contrasting demographic history and phylogeographical patterns in two Indo-Pacific gastropods. Mol Ecol 17:611–626
DeWoody JA, Avise JC (2000) Microsatellite variation in marine, freshwater and anadromous fishes compared with other animals. J Fish Biol 56:461–473
Excoffier L, Laval G, Schneider S (2005) Arlequin ver 3.0: an integrated software package for population genetics data analysis. Evol Bioinform. Online 1:47–50
Felsenstein J (1989) PHYLIP–phylogeny inference package (Version 3.2). Cladistics 5:164–166
Goudet J (2001) FSTAT, a program to estimate and test gene diversities and fixation indices (version 2.9.3). Available from http://www.unil.ch/izea/softwares/fstat.html. Updated from Goudet (1995)
Guo X, Ford SE, Zhang F (1999) Molluscan aquaculture in China. J Shellfish Res 18:19–31
Guo X, Zhang G, Qian L, Wang H, Liu X, Wang A (2006) Oysters and oyster farming in China: a review. J Shellfish Res 25:734 (Abstract)
Hare MP, Avise JC (1998) Population structure in the American oyster as inferred by nuclear gene genealogies. Mol Biol Evol 15:119–128
Hedgecock D (1994) Does variance in reproductive success limit effective population sizes of marine organisms? In: Beaumont AR (ed) Genetics and evolution of aquatic organisms. Chapman & Hall, London, pp 122–134
Hedgecock D, Li G, Banks MA, Kain Z (1999) Occurrence of the Kumamoto oyster Crassostrea sikamea in the Ariake Sea, Japan. Mar Biol 133:65–68
Huvet A, Lapègue S, Magoulas A, Boudry P (2000) Mitochondrial and nuclear DNA phylogeography of Crassostrea angulata, the Portuguese oyster endangered in Europe. Conserv Gen 1:251–262
ICES: International Council for the Exploration of the Sea (2005) Code of practice on the introductions and transfers of marine organisms 2005, 30 pp
Karl SA, Avise JC (1992) Balancing selection at allozyme loci in oysters: implications from nuclear RFLPs. Science 256:100–102
Kelly DW, MacIsaac HJ, Heath DD (2006) Vicariance and dispersal effects on phylogeographic structure and speciation in a wild spread estuarine invertebrate. Evolution 60:257–267
Kim S, Lee S, Park D, An H (2000) Phylogenetic relationship among four species of Korean oysters based on mitochondrial 16S rDNA and COI gene. Korean J Biol Sci 16:203–211
Kinlan BP, Gaines SD (2003) Propagule dispersal in marine and terrestrial environments: a community perspective. Ecology 84:2007–2020
Kong L, Li Q, Qiu Z (2007) Genetic and morphological differentiation in the clam Coelomactra antiquata (Bivalvia: Veneroida) along the coast of China. J Exp Mar Biol Ecol 343:110–117
Lam K, Morton B (2003) Mitochondrial DNA and morphological identification of a new species of Crassostrea (Bivalvia: Ostreidae) cultured for centuries in the Pearl River Delta, Hong Kong, China. Aquaculture 228:1–13
Langdon CJ, Robinson AM (1996) Aquaculture potential of the Suminoe oyster (Crassostrea ariakensis Fugita 1913). Aquaculture 144:321–338
Launey S, Ledu C, Boudry P, Bonhomme F, Naciri-Graven Y (2002) Geographic structure in the European flat oyster (Ostrea edulis L.) as revealed by microsatellite polymorphism. J Hered 93:331–338
Li G, Hubert S, Bucklin K, Ribes V, Hedgecock D (2003) Characterization of 79 microsatellite DNA markers in the Pacific oyster Crassostrea gigas. Mol Ecol Notes 3:228–232
Li Q, Yu H, Yu R (2006) Genetic variability assessed by microsatellite in cultured populations of the Pacific oyster (Crassostrea gigas) in China. Aquaculture 259:95–102
Lind CE, Evans BS, Taylor JJU, Jerry DR (2007) Population genetics of a marine bivalve, Pinctada maxima, throughout the Indo-Australian archipelago shows differentiation and decreased diversity at range limits. Mol Ecol 16:5193–5203
Mantel N (1967) Detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Mariani S, Ketmaier V, de Matthaeis E (2002) Genetic structuring and gene flow in Cerastoderma glaucum (Bivalvia: Cardiidae): evidence from allozyme variation at different geographic scales. Mar Biol 140:687–697
McMillan WO, Palumbi SR (1995) Concordant evolutionary patterns among Indo-west Pacific butterflyfishes. P Roy Soc Lond B Bio 260:229–236
Moss JA, Burreson EM, Cordes JF, Dungan CF, Brown GD, Wang A, Wu X, Reece KS (2007) Pathogens in Crassostrea ariakensis and other Asian oyster species: implications for non-native oyster introduction to Chesapeake Bay. Dis Aquat Org 77:207–223
Moss JA, Xiao J, Dungan CF, Reece KS (2008) Description of Perkinsus beihaiensis n. sp., a new Perkinsus sp. parasite in oysters of southern China. J Eukaryot Microbiol 55:117–130
Murray-Jones SE, Ayre DJ (1997) High levels of gene flow in the surf bivalve Donax deltoids (Bivalvia: Donacidae) on the east coast of Australia. Mar Biol 128:83–89
Nei M (1978) Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89:583–590
Nikula R, Väinölä R (2003) Phylogeography of Cerastoderma glaucum (Bivalvia: Cardiidae) across Europe: a major break in the Eastern Mediterranean. Mar Biol 143:339–350
ÓFoighil D, Gaffney PM, Hibish TJ (1995) Differences in mitochondrial 16S ribosomal gene sequences allow discrimination among American [Crassostrea virginica (Gmelin)] and Asian [C. gigas (Thunberg) C. ariakensis Wakiya] oyster species. J Exp Mar Biol Ecol 192:211–220
Page RDM (1996) TREEVIEW: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Palumbi SR (2003) Population genetics, demographic connectivity, and the design of marine reserves. Ecol Appl 13:S146–S158
Palumbi SR, Grabowsky G, Duda T, Geyer L, Tachino N (1997) Speciation and population genetic structure in tropical Pacific sea urchins. Evolution 51:1506–1517
Petit RJ, Moousadik AE, Pons O (1998) Identifying populations for conservation on the basis of genetic markers. Conserv Biol 12:844–855
Planes S, Fauvelot C (2002) Isolation by distance and vicariance drive genetic structure of coral reef fish in the Pacific Ocean. Evolution 56:378–399
Raymond M, Rousset F (1995) GENEPOP (version 1.2)–population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Reeb CA, Avise JC (1990) A genetic discontinuity in a continuously distributed species: mitochondrial DNA in the American oyster, Crasosstrea virginica. Genetics 124:397–406
Reece KS, Cordes JF, Stubbs JB, Hudson KL, Francis EA (2008) Molecular phylogenies help resolve taxonomic confusion with Asian Crassostrea oyster species. Mar Biol 153:709–721
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Ríos C, Sanz S, Saavedra C, Peña JB (2002) Allozyme variation in populations of scallops. Pecten jacobaeus (L.) and P. maximus (L.) (Bivalvia: Pectinidae), across the Almeria-Oran front. J Exp Mar Biol Ecol 267:223–244
Rose CG, Paynter T, Hare MP (2006) Isolation by distance in the eastern oyster, Crassostrea virginica, in Chesapeake Bay. J Hered 97:158–170
Ruesink JL, Lenihan HS, Trimble AC, Heiman KW, Micheli F, Byers JE, Kay MC (2005) Introduction of non-native oysters: ecosystem effects and restoration implications. Annu Rev Ecol Evol Syst 36:643–689
Sellos D, Moal J, Degremont L, Huvet A, Daniel JY, Nicoulaud S, Boudry P, Samain JF, van Wormhoudt A (2003) Structure of amylase genes in populations of the Pacific cupped oyster (Crassostrea gigas): tissue expression and allelic polymorphism. Mar Biotechnol 5:360–372
Shen H, Zhu J, Ding Y, Chen G (2003) RAPD analysis of three wild stocks of Meretrix meretrix in China’s coastal seas. Acta Oceanologica Sinica 25:97–102 (in Chinese with English abstract)
STAC (2004) Identifying and prioritizing research required to evaluate ecological risks and benefits of introducing diploid Crassostrea ariakensis to restore oysters to Chesapeake Bay. Report of the Scientific and Technical Advisory Committee Workshop, 2–3 Dec 2003 in Annapolis, Maryland, 39 pp
Sündermann J, Feng S (2004) Analysis and modeling of the Bohai sea ecosystem–a joint German–Chinese study. J Mar Syst 44:127–140
Taylor JS, Sanny JSP, Breden F (1999) Microsatellite allele size homoplasy in the Guppy (Poecilia reticulata). J Mol Evol 48:245–247
Thorson G (1950) Reproductive and larval ecology of marine bottom invertebrates. Biol Rev 25:1–45
van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO_CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538
van Oppen MJH, Rico C, Turner GF, Hewitt GM (2000) Extensive homoplasy, nonstepwise mutations, and shared ancestral polymorphism at a complex microsatellite locus in Lake Malawi Cichlids. Mol Biol Evol 17:489–498
Wang H, Guo X (2006) Identification of Crassostrea species from China using SNP-based markers. J Shellfish Res 25:789 (Abstract)
Wang H, Guo X (2008) Identification of Crassostrea ariakensis and related oysters by multiplex species-specific PCR. J Shellfish Res 27:481–487
Wang H, Guo X, Zhang G, Zhang F (2004) Classification of jinjiang oysters Crassostrea rivularis (Gould, 1861) from China, based on morphology and phylogenetic analysis. Aquaculture 242:137–155
Wang H, Qian L, Zhang G, Liu X, Wang A, Shi Y, Jiao N, Guo X (2006) Distribution of Crassostrea ariakensis in China. J Shellfish Res 25:789 (Abstract)
Wang H, Zhang G, Liu X, Guo X (2008) Classification of common oysters from North China. J. Shellfish Res 27:495–504
Waples RS (1998) Separating the wheat from the chaff: patterns of genetic differentiation in high gene flow species. J Hered 89:438–450
Ward RD, Woodwark M, Skibinski DOF (1994) A comparison of genetic diversity levels in marine, freshwater, and anadromous fishes. J Fish Biol 44:213–232
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Xiao J, Cordes JF, Jones KC, Reece KS (2008) Eleven novel microsatellite markers for the Asian oyster Crassostrea ariakensis. Mol Ecol Notes 8:843–845
Xu F (1997) Bivalves from China seas. Science Press, Beijing. (In Chinese)
Yoon HS, Jung HT, Choi SD (2008) Suminoe oyster (Crassostrea ariakensis) culture in Korea. J Shellfish Res 27:505–508
Yu DH, Chu KH (2006a) Low genetic differentiation among widely separated populations of the pearl oyster Pinctada fucata as revealed by AFLP. J Exp Mar Biol Ecol 333:140–146
Yu DH, Chu KH (2006b) Genetic variation in wild and cultured populations of the pearl oyster Pinctada fucata from southern China. Aquaculture 258:220–227
Yu H, Li Q, Yu R (2008) Genetic differentiation between Zhe oyster Crassostrea plicatula and Pacific oyster Crassostrea gigas populations in China assessed by microsatellite analysis. Fish Sci 74:88–97
Zhang X, Lou ZK (1956) Studies on oysters in China. Acta Zool Sin 8:65–94 (in Chinese)
Zhang Q, Allen SK, Reece KS (2005) Genetic variation in wild and hatchery stocks of Suminoe oyster (Crassostrea ariakensis) assessed by PCR-RFLP and microsatellite markers. Mar Biotechnol 7:588–599
Zhou M, Allen SK (2003) A review of published work on Crassostrea ariakensis. J Shellfish Res 22:1–20
Acknowledgments
We thank Dr. Hyesuk An for providing valuable samples from South Korea, and Drs. Guofan Zhang, Lumin Qian, Fei Xu, Xiao Liu, Huayong Que and Aiming Wang for assisting with sampling trips in China. We are also grateful to Kelly Johnson and Drs. Ryan Carnegie, Caiwen Li and Jens Carlsson for assistance on sample collection from China in 2005 and 2006. Dr. Xiaoxue Wang kindly sent us additional samples and information that we needed. Dr. Jessica Moss assisted with DNA extractions. This research was funded by NOAA (Award Number: NA04NMF4570432). VIMS contribution number 3082.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Uthicke.
Rights and permissions
About this article
Cite this article
Xiao, J., Cordes, J.F., Wang, H. et al. Population genetics of Crassostrea ariakensis in Asia inferred from microsatellite markers. Mar Biol 157, 1767–1781 (2010). https://doi.org/10.1007/s00227-010-1449-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-010-1449-x