Abstract
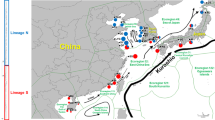
In a previous study on the kuruma shrimp Penaeus japonicus from the South China Sea, we detected high genetic divergence between two morphologically similar varieties (I and II) with distinct color banding patterns on the carapace, indicating the occurrence of cryptic species. In the present study, we clarify the geographical distribution of the two varieties in the western Pacific by investigating the genetic differentiation of the shrimp from ten localities. Two Mediterranean populations are also included for comparison. Based on the mitochondrial DNA sequence data, the shrimps are separated into two distinct clades representing the two varieties. Variety I comprises populations from Japan and China (including Taiwan), while variety II consists of populations from Southeast Asia (Vietnam, Singapore and the Philippines), Australia and the Mediterranean. Population differentiation is evident in variety II, as supported by restriction profiles of two mitochondrial markers and analysis of two microsatellite loci. The Australian population is genetically diverged from the others, whereas the Southeast Asian and Mediterranean populations show a close genetic relationship. Variety I does not occur in these three localities, while a small proportion of variety II is found along the northern coast of the South China Sea and Taiwan, which constitute the sympatric zone of the two varieties. The present study reveals high genetic diversity of P. japonicus. Further studies on the genetic structure of this species complex, particularly the populations in the Indian Ocean and Mediterranean, are needed not only to understand the evolutionary history of the shrimp, but also to improve the knowledge-based fishery management and aquaculture development programs of this important biological resource.






Similar content being viewed by others
References
Baldwin JD, Bass AL, Bowen BW, Clark WH Jr (1998) Molecular phylogeny and biogeography of the marine shrimp Penaeus. Mol Phylogenet Evol 10:399–407
Ball AO, Chapman RW (2003) Population genetic analysis of white shrimp Litopenaeus setiferus, using microsatellite genetic markers. Mol Ecol 12:2319–2330
Ball AO, Leonard S, Chapman RW (1998) Characterization of (GT)n microsatellites from native white shrimp (Penaeus setiferus). Mol Ecol 7:1251–1253
Balss H (1927) Berichte uber die Crustacea Decapoda (Natantia und Anomura). Zoological Results of the Cambridge Expedition to the Suez Canal, 1924, XIV. Trans Zool Soc Lond 22:221–227
Banks MA, Eichert W (2000) WHICHRUN (Version 3.2) a computer program for population assignment of individuals based on multilocus genotype data. J Hered 91:87–89
Barnard KH (1950) Descriptive catalogue of South African decapod Crustacea. Ann South Afr Mus 38:1–837
Barton NH (1989) Founder effect speciation. In: Otte D, Endler JA (eds) Speciation and its consequences. Sinauer, Sunderland, pp 229–256
Bate CS (1888) Report on the Crustacea Macrura collected by H.M.S. “Challenger” during the years 1873–76. Report on the scientific results of the voyage of H.M.S. “Challenger” during the years 1873–76 24:1–942
Benzie JAH (1999) Genetic structure of coral reef organisms—ghost of dispersal past. Am Zool 39:131–145
Benzie JAH, Ballment E, Forbes AT, Demetriades NT, Sugama K, Haryanti, Moria S (2002) Mitochondrial DNA variation in Indo-Pacific populations of the giant tiger prawn, Penaeus monodon. Mol Ecol 11:2553–2569
Bloom AL (1971) Glacial eustatic and isostatic controls of sea level since the last glaciation. In: Turekian KK (ed) The late Cenozoic glacial ages. Yale University Press, New Haven, pp 355–380
Brooker AL, Benzie JAH, Blair D, Versini JJ (2000) Population structure of the giant tiger prawn Penaeus monodon in Australian waters, determined using microsatellite markers. Mar Biol 136:149–157
Camin JH, Sokal RR (1965) A method for deducing branching sequences in phylogeny. Evolution 19:311–326
Castric V, Bernatchez L, Belkhir K, Bonhomme F (2002) Heterozygote deficiencies in small lacustrine populations of brook charr Salvelinus fontinalis Mitchill (Pisces, Salmonidae): a test of alternative hypotheses. Heredity 89:27–35
Chaitiamvong S, Supongpan M (eds) (1992) A guide to penaeoid shrimps found in Thai waters. Australian Institute of Marine Science, Townsville, p 77
Chan TY (1998) Shrimps and prawns. In: Carpenter KE, Niem VH (eds) FAO Species identification guide for fishery purposes. The living marine resources of the western central Pacific vol 2. FAO, Rome, p 917
Chan TY (2004) The “Plesionika rostricrescentis (Bate, 1888)” and “P. lophotes Chace, 1985” species groups of Plesionika Bate, 1888, with descriptions of five new species (Crustacea: Decapoda: Pandalidae). In: Marshall B, Richer de Forges B (eds) Tropical deep-sea benthos vol 23. Mémoires du Muséum national d’Histoire naturelle 191:293–318
Chan TY, Crosnier A (1997) Crustacea Decapoda: Deep sea shrimps of the genus Plesionika Bate, 1888 (Pandalidae) from French Polynesia, with descriptions of five new species. In: Crosnier A (ed), Resultats des Campangnes MUSORSTOM vol 18. Mémoires du Muséum national d’Histoire naturelle 176:187–234
Chappell J, Omura A, Esat T, McCulloch M, Pandolfi J, Ota Y, Pillans B (1996) Reconciliation of late Quaternary sea levels derived from coral terraces at Huon Peninsula with deep sea oxygen isotope records. Earth Planet Sci Lett 141:227–236
Chu TY (1972) A study on the water exchange between Pacific Ocean and the South China Sea. Acta Ocean Taiwanica 2:11–24
Chu KH, Li CP, Tam YK, Lavery S (2003) Application of mitochondrial control region in population genetic studies of the shrimp Penaeus. Mol Ecol Notes 3:120–122
Chu P, Chen Y, Kuninaka A (2005) Seasonal variability of the Yellow Sea/East China Sea surface fluxes and thermohaline structure. Adv Atmo Sci 2:11–24
Clark PF (1990) Asian prawns go wild in the Channel. New Sci 125:30
Crow J, Kimura M (1965) Evolution in sexual and asexual populations. Am Nat 99:439–450
Dall W (1991) Zoogeography of the Penaeidae. Mem Queensland Mus 31:39–50
Dall W, Hill J, Rothlisberg PC, Staples DJ (1990) The biology of Penaeidae. In: Blaxter JHS, Southward AJ (eds) Advances in marine biology vol 27. Academic, New York, USA
Demetropoulos A, Neocleous D (1969) The fishes and crustaceans of Cyprus. Fisheries Bulletin of the Department of Fisheries, Cyprus 1:3–21
Duda TF Jr, Palumbi SR (1999) Population structure of the black tiger prawn, Penaeus monodon, among western Indian Ocean and western Pacific populations. Mar Biol 134:705–710
Farris A, Wimbush M (1996) Wind-induced Kuroshio intrusion into the South China Sea. J Oceanogr 52:771–784
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376
Fleminger A (1986) The Pleistocene equatorial barrier between the Indian and Pacific Oceans and a likely cause for Wallace’s line. UNESCO Tech Pap Mar Sci 49:84–97
Freitas de A, Silva C (1990) Lagostas, Camarões, Caranguejos. In: Fischer W (ed) Fischas FAO de Identificação de Espécies Para Actividades de Pesca. Guía de campo das espécies comerciais marinhas e de águas salobras de Moçambique. FAO, Rome, pp 275–315
Fukami H, Budd AF, Levitan DR, Jara J, Kersanach R, Knowlton N (2004) Geographic differences in species boundaries among members of the Montastraea annularis complex based on molecular and morphological markers. Evolution 58:324–337
Galil B, Zenetos A (2002) A sea change—exotics in the eastern Mediterranean. In: Leppäkoski E, Olenin S, Gollasch S (eds) Invasive aquatic species of Europe: distributions, impacts and management. Kluwer Academic Publishers, Dordrecht, pp 325–336
Galil B, Froglia C, Noël P (2002) CIESM atlas of exotic species in the Mediterranean. In: Briand F (ed) Crustaceans: Decapods and Stomatopods vol 2. CIESM Publishers, Monaco, p 192
Gilberto R, Héctor S (2001) Anthropogenic dispersal of decapod crustaceans in aquatic environments. Interciencia 26:282–288
Goodman SJ (1997) RST CALC: a collection of computer programs for calculating estimates of genetic differentiation from microsatellite data and a determining their significance. Mol Ecol 6:881–885
Gopurenko D, Hughes JM, Keenan CP (1999) Mitochondrial DNA evidence for rapid colonisation of the Indo-West Pacific by the mudcrab Scylla serrata. Mar Biol 134:227–233
Goudet J (1995) FSTAT version 1.2: a computer program to calculate F-statistics. J Hered 86:485–486
Grey DL, Dall W, Baker A (eds) (1983) A guide to the Australian penaeid prawns. Northern Territory Government Printing Office, Darwin, Australia, p 140
Gruvel A (1928) Répartition géographique de quelques crustacés comestibles sur les côtes d’Egypte et de Syrie. Comptes rendus de la société de biogéographie 5:45–46
Guo SW, Thompson EA (1992) Performing the exact test of Hardy-Weinberg proportion for multiple alleles. Biometrics 48:361–372
Gusmão J, Lazoski CVS, Solé-Cava AM (2000) A new species of Penaeus (Crustacea: Penaeidae) revealed by allozyme and cytochrome oxidase I analysis. Mar Biol 137:435–446
Hasegawa M, Kishino H, Yano TA (1985) Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J Mol Evol 22:160–174
Hewitt DR, Duncan PF (2001) Effect of high water temperature on the survival, moulting and food consumption of Penaeus (Marsupenaeus) japonicus (Bate, 1888). Aquacult Res 32:305–313
Holthuis LB (1980) Shrimps and prawns of the world. An annotated catalogue of species of interest to fisheries. FAO Fisheries Synopsis No. 125, 1:1–271
Holthuis LB (1984) Lobsters. In: FAO species identification sheets, western Indian Ocean (Fishery Area 51), FAO, Rome
Holthuis LB, Gottlieb E (1958) An annotated list of the decapod Crustacea of the Mediterranean coast of Israel, with an appendix listing the Decapoda of the eastern Mediterranean. Bull Res Counc Isr 7B:1–126
Hualkasin W, Sirimontaporn P, Chotigeat W, Quercic J, Phongdara A (2003) Molecular phylogenetic analysis of white prawns species and the existence of two clades in Penaeus merguiensis. J Exp Mar Biol Ecol 296:1–11
Hudinaga M (1935) Studies on the development of Penaeus japonicus (Bate). Hayatomo Fishery Institute Report 1
Hudinaga M (1942) Reproduction, development and rearing of Penaeus japonicus Bate. Japan J Zool 10:305–393
Jan S, Wang J, Chern CS, Chao SY (2002) Seasonal variation of the circulation in the Taiwan Strait. J Mar Syst 35:249–268
Jerry DR, Preston NP, Crocos PJ, Keys S, Meadows JRS, Li Y (2004) Parentage determination of kuruma shrimp Penaeus (Marsupenaeus) japonicus using microsatellite markers (Bate). Aquaculture 235:237–247
Kapiris K (1997) Shrimp culture in Hellas (Greece). Research and development in sustainable coastal aquaculture ecosystems, Unit 4. School of Biological Sciences, UC Irvine
Kevrekidis K, Kevrekidis T (1996) The occurrence of Penaeus japonicus in the Aegean Sea. Crustaceana 69:925–929
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Klinbunga S, Penman DJ, McAndrew BJ, Tassanakajon A (1999) Mitochondrial DNA diversity in three populations of the giant tiger shrimp Penaeus monodon. Mar Biotechnol 1:113–121
Klinbunga S, Siludjai D, Wudthijinda W, Tassanakajon A, Jarayabhand P, Menasveta P (2001) Genetic heterogeneity of the giant tiger shrimp (Penaeus monodon) in Thailand revealed by RAPD and mitochondrial DNA RFLP analyses. Mar Biotechnol 3:428–438
Knowlton N (1986) Cryptic and sibling species among the decapod Crustacea. J Crustac Biol 6:356–363
Knowlton N (1993) Sibling species in the sea. Annu Rev Ecol Syst 24:189-216
Knowlton N, Keller BD (1985) Two more sibling species of alpheid shrimps associated with the Caribbean sea anemones Bartholomea annulata and Heteractis lucida. Bull Mar Sci 37:893–904
Knowlton N, Weigt LA (1998) New dates and new rates for divergence across the Isthmus of Panama. Proc R Soc Lond B Biol Sci 265:2257–2263
Kumar S, Tamura K, Nei M (2004) MEGA3: Integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
Lavery S, Chan TY, Tam YK, Chu KH (2004) Phylogenetic relationships and evolutionary history of the shrimp genus Penaeus s. l. derived from mitochondrial DNA. Mol Phylogenet Evol 31:39–49
Lefébure T, Douady CJ, Gouy M, Gibert J (2006) Relationship between morphological taxonomy and molecular divergence within Crustacea: proposal of a molecular threshold to help species delimitation. Mol Phylogenet Evol (in press)
Liao IC, Chien YH (1994) Culture of kuruma prawn (Penaeus japonicus) in Asia. World Aquacult 25(1):18–33
Lumare F, Palmegiano GB (1980) Acclimatazione di Penaeus japonicus Bate nella Laguna di Lesina (Italia sud orientale). Rivista Italiana di Piscicoltura e Ittiopatologia 15:53–58
Lumare F, Scordella G, Pastore M, Prato E, Zanella L, Tessarin C, Sanna A (2000) Pond management and environmental dynamics in semiextensive culture of Penaeus japonicus (Decapoda, Penaeidae) on the northern Adriatic coast of Italy. Rivista Italiana di Acquacoltura 35:15–43
Maggioni R, Rogers AD, Maclean N, D’Incao F (2001) Molecular phylogeny of western Atlantic Farfantepenaeus and Litopenaeus shrimp based on mitochondrial 16S partial sequences. Mol Phylogenet Evol 18:66–73
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Mayr E (1942) Systematics and the origin of species. Columbia University Press, New York
McElroy D, Moran P, Bermingham E, Kornfield I (1991) REAP: The restriction enzyme analysis package, ver 4.0. Department of Zoology, University of Maine, Orono
McLaughlin PA (ed) (1980) Comparative morphology of recent Crustacea. WH Freeman, San Francisco
McMillen-Jackson AL, Bert TM (2003) Disparate patterns of population genetic structure and population history in two sympatric penaeid shrimp species (Farfantepenaeus aztecus and Litopenaeus setiferus) in the eastern United States. Mol Ecol 12:2895–2905
McMillen-Jackson AL, Bert TM (2004) Genetic diversity in the mtDNA control region and population structure in the pink shrimp Farfantepenaeus duorarum. J Crustac Biol 24:101–109
Merril CR, Switzer RC, Van Keuren ML (1979) Trace polypeptides in cellular extracts and human body fluid detected by two-dimensional electrophoresis and a highly sensitive silver stain. Proc Natl Acad Sci USA 76:4335–4339
Michalakis Y, Excoffier L (1996) A generic estimation of population subdivision using distances between alleles with special reference for microsatellite loci. Genetics 142:1061–1064
Miyake S (1998) Japanese crustacean decapods and stomatopods in color. 1. Macrura, Anomura and Stomatopoda. Hoiksuha, Osaka, p 261
Monod T (1930) Über einige indo-pazifischer Decapoden der Meeresfauna Syriens. Zool Anz 92:135–141
Moore SS, Whan V, Davis GP, Byrne K, Hetzel DJS, Preston N (1999) The development and application of genetic markers for the kuruma prawn Penaeus japonicus. Aquaculture 173:19–32
Morton B, Blackmore G (2001) South China Sea. Mar Pollut Bull 42:1236–1263
Motoh H (ed) (1980) Field guide for the edible Crustacea of the Philippines. SEAFDEC Aquaculture Department, Iloilo, p 96
Nei M (1977) F-statistics and analysis of gene diversity in subdivided populations. Ann Hum Genet 41:225-233
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269-5273
Nei M, Tajima F (1981) DNA polymorphism detectable by restriction endonucleases. Genetics 97:145-163
Niino H, Emery KO (1961) Sediments of shallow portions of East China Sea and South China Sea. Geol Soc Am Bull 72:731–762
Palumbi SR, Benzie J (1991) Large mitochondrial DNA differences between morphologically similar penaeid shrimp. Mol Mar Biol Biotechnol 1:27–34
Pérez Farfante I (1976) A redescription of Penaeus (Melicertus) canaliculatus (Olivier, 1811), a wide-ranging Indo-West Pacific shrimp (Crustacea, Decapoda, Penaeidae). Zoologische Mededelingen 50:23–37
Pérez Farfante I, Kensley B (1997) Penaeoid and sergestoid shrimps and prawns of the world. Keys and diagnoses for the families and genera. Mém Mus Natl Hist Nat 175:1–233
Posada D, Crandall KA (1998) MODELTEST: testing the model of DNA substitution. Bioinformatics 14:817-818
Raymond M, Rousset F (1995a) An exact test for population differentiation. Evolution 49:1280–1283
Raymond M, Rousset F (1995b) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249
Reynolds J, Weir BS, Cockerham CC (1983) Estimation of the coancestry coefficient: basis for a short-term genetic distance. Genetics 105:767–779
Roff DA, Bentzen P (1989) The statistical analysis of mitochondrial DNA polymorphisms: χ2 and the problem of small samples. Mol Biol Evol 6:539–545
Rosenberry B (2001) World shrimp farming 2001. Shrimp News International, San Diego
Rousset F (1996) Equilibrium values of measure of population subdivision for stepwise mutation processes. Genetics 142:1357–1362
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sbordoni V, DeMatthaeis E, Cobolli Sbordoni M, La Rosa G, Mattoccia M (1986) Bottleneck effects and the depression of genetic variability in hatchery stocks of Penaeus japonicus (Crustacea, Decapoda). Aquaculture 57:239–251
Schneider S, Roessli D, Excoffier L (2000) Arlequin Ver 2.000: A software for population genetics data analysis. Genetics and Biometry Laboratory, University of Geneva, Switzerland
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701
Slatkin M (1977) Gene flow and genetic drift in a species subject to frequent local extinctions. Theor Popul Biol 12:253–262
Slatkin M (1985) Gene flow in natural populations. Ann Rev Ecol Syst 16:393–430
Sokal RR, Rohlf FJ (eds) (1995) Biometry: The principles and practice of statistics in biological research. WH Freeman and Company, New York
Stebbing TRR (1914) South African Crustacea. (Part VII of S. A. Crustacea, for the Marine Investigations in South Africa). Ann South Afr Mus 15:1–55
Sturmbauer C, Levinton JS, Christy J (1996) Molecular phylogeny analysis of fiddler crabs: test of the hypothesis of increasing behavioral complexity in evolution. Proc Natl Acad Sci USA 93:10855–10857
Sugaya T, Ikeda M, Taniguchi N (2002a) Relatedness structure estimated by microsatellites DNA and mitochondrial DNA polymerase chain reaction-restriction fragment length polymorphisms analyses in the wild population of kuruma prawn Penaeus japonicus. Fish Sci 68:793–802
Sugaya T, Ikeda M, Mori H, Taniguchi N (2002b) Inheritance mode of microsatellite DNA markers and their use for kinship estimation in kuruma prawn Penaeus japonicus. Fish Sci 68:299–305
Supungul P, Sootanan P, Klinbunga S, Kamonrat W, Jarayabhand P, Tassanakajon A (2000) Microsatellite polymorphism and the population structure of the black tiger shrimp (Penaeus monodon) in Thailand. Mar Biotechnol 2:339–347
Swofford DL (2000) PAUP*: Phylogenetic analysis using parsimony (*and other methods), ver. 4. Sinauer, Sunderland, MA
Tautz D (1989) Hypervariability of simple sequences as a general source for polymorphic DNA markers. Nucleic Acids Res 17:6463–6471
Thompson JD, Higgins DG, Gibson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tirmizi NW (1971) Marsupenaeus, a new subgenus of Penaeus Fabricius, 1798 (Decapoda, Natantia). Pakistan J Zool 3:193–194
Tong JG, Chan TY, Chu KH (2000) A preliminary phylogenetic analysis of Metapenaeopsis (Decapoda: Penaeidae) based on mitochondrial DNA sequences of selected species from the Indo-West Pacific. J Crustac Biol 20:541–549
Tsoi KH, Wang ZY, Chu KH (2005) Genetic divergence between two morphologically similar varieties of the kuruma shrimp Penaeus japonicus. Mar Biol 147:367–379
Tzeng TD, Yeh SY (1999) Analysis of the morphometric characters of the kuruma shrimp (Penaeus japonicus) in the East China Sea and the Taiwan Strait. J Fish Soc Taiwan 26:203–212
Tzeng TD, Yeh SY (2002) Multivariate allometric comparisons for kuruma shrimp (Penaeus japonicus) off Taiwan. Fish Res 59:279–288
Tzeng TD, Yeh SY, Hui CF (2004) Population genetic structure of the kuruma prawn (Penaeus japonicus) in East Asia inferred from mitochondrial DNA sequences. J Mar Sci 61:913–920
Valles-Jimenez R, Cruz P, Perez-Enriquez R (2005) Population genetic structure of Pacific white shrimp (Litopenaeus vannamei) from Mexico to Panama: microsatellite DNA variation. Mar Biotechnol 6:475–484
Valles-Jimenez R, Gaffney PM, Perez-Enriquez R (2006) RFLP analysis of the mtDNA control region in white shrimp (Litopenaeus vannamei) populations from the eastern Pacific. Mar Biol 148:867–873
Vo ST (1998) The hermatypic Scleractinia of South Vietnam. In: Morton B (ed) The marine biology of the South China Sea III. Hong Kong University Press, Hong Kong, pp 11–21
Wahlund S (1928) Zusammensetzung von Populationen und Korrelationsers-chinungen von Standpunkt der Vererbungslehre aus betrachtet. Hereditas 11:65–106
Wang L, Sarnthein M, Erlenkeuser H, Grimalt J, Grootes P, Heilig S, Ivanova E, Kienast M, Pelejero C, Pflaumann U (1999) East Asian monsoon climate during the late Pleistocene: high-resolution sediment records from the South China Sea. Mar Geol 156:245–284
Wanna W, Rolland JL, Bonhomme F, Phongdara A (2004) Population genetic structure of Penaeus merguiensis in Thailand based on nuclear DNA variation. J Exp Mar Biol Ecol 311:63–78
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Williams ST, Knowlton N, Weigt LA, Jara JA (2001) Evidence for three major clades within the snapping shrimp genus Alpheus inferred from nuclear and mitochondrial gene sequence data. Mol Phylogenet Evol 20:375–389
Xu Z, Primavera JH, de la Pena LD, Pettit P, Belak J, Alcivar-Warren A (2001) Genetic diversity of wild and cultured black tiger shrimp (Penaeus monodon) in the Philippines using microsatellites. Aquaculture 199:13–40
Yu HP, Chan TY (eds) (1986) The illustrated penaeoid prawns of Taiwan. Southern Materials Center, Taiwan, pp 88–90
Acknowledgments
Sincere thanks are given to P.O. Ang Jr., C.C. Cheang (The Chinese University of Hong Kong), S.A. Lehnert (CSIRO Division of Livestock Industries, Australia), P.K.L. Ng (National University of Singapore), J. Primavera (SEAFDEC Aquaculture Department, the Philippines), K. Tanaka (Kyorin University, Japan), K.T. Tai (University of Fishery, Vietnam), Y.H. Yung (The Chinese University of Hong Kong), Z.Y. Wang (Jimei University, China), J. Wong and P.H. Wong (Princess Margaret Hospital, Hong Kong) for collecting specimens. We are indebted to Z.Y. Wang (Jimei University, China), J. Tong (Institute of Hydrobiology, Chinese Academy of Sciences), Y.K. Tam, C.P. Li and W. Lau (The Chinese University of Hong Kong) for technical advice, and to Z.Y. Wang, B.S. Galil (National Institute of Oceanography, Israel), A. Crosnier (Museum National d’Histoire Naturelle, Paris), P. Clark (Museum of Natural History, London), F. Coman and N. Preston (CSIRO Marine and Atmospheric Research, Australia) for personal communications on P. japonicus. We also thank T.D. Tzeng (She Te University, Taiwan) for providing us the CR sequences from his study for comparison, and B.K.K. Chan (Academia Sinica, Taiwan), C.K. Wong, J.W. Xu (The Chinese University of Hong Kong) and the anonymous reviewers for constructive comments on the manuscript. The work described in this paper was fully supported by a grant from the Research Grants Council, Hong Kong Special Administrative Region (HKSAR), China (Project No. CUHK4157/01M).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M.S. Johnson, Crawley.
Rights and permissions
About this article
Cite this article
Tsoi, K.H., Chan, T.Y. & Chu, K.H. Molecular population structure of the kuruma shrimp Penaeus japonicus species complex in western Pacific. Mar Biol 150, 1345–1364 (2007). https://doi.org/10.1007/s00227-006-0426-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-006-0426-x