Abstract
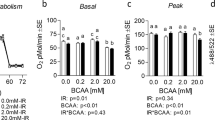
Uncarboxylated osteocalcin (ucOC) stimulates muscle glucose uptake in mice EDL and soleus muscles. However, whether ucOC also exerts a similar effect in insulin-stimulated muscles in a muscle type-specific manner is currently unclear. We aimed to test the hypothesis that, with insulin stimulation, ucOC per se has a greater effect on oxidative muscle compared with glycolytic muscle, and to explore the underlying mechanisms. Mouse (C57BL6, male 9–12 weeks) extensor digitorum longus (EDL) and soleus muscles were isolated and longitudinally split into halves. Muscle samples were treated with varying doses of recombinant ucOC (0, 0.3, 1, 3, 30 ng/ml), followed by insulin addition. Muscle glucose uptake, protein phosphorylation and total expression of protein kinase B (Akt), Akt substrate of 160 kDa (AS160), extracellular signal-regulated kinase isoform 2 (ERK2), and adenosine monophosphate-activated protein kinase subunit α (AMPKα) were assessed. ucOC treatment at 30 ng/ml enhanced muscle glucose uptake in insulin-stimulated soleus, a mainly oxidative muscle (17.5%, p < 0.05), but not in EDL—a mostly glycolytic muscle. In insulin-stimulated soleus only, ucOC treatment (3 and 30 ng/ml) increased phosphorylation of AS160 and ERK2, but not Akt or AMPKα. The ucOC-induced increase in ERK2 phosphorylation in soleus was not associated with the increase in glucose uptake or AS160 phosphorylation. ucOC enhances glucose uptake and AS160 phosphorylation in insulin-stimulated oxidative but not glycolytic muscle, via upstream mechanisms which appear to be independent of ERK or AMPK.




Similar content being viewed by others
References
Brennan-Speranza TC, Henneicke H, Gasparini SJ, Blankenstein KI, Heinevetter U, Cogger VC, Svistounov D, Zhang Y, Cooney GJ, Buttgereit F (2012) Osteoblasts mediate the adverse effects of glucocorticoids on fuel metabolism. J Clin Invest 122:4172
Ferron M, Hinoi E, Karsenty G, Ducy P (2008) Osteocalcin differentially regulates β cell and adipocyte gene expression and affects the development of metabolic diseases in wild-type mice. Proc Natl Acad Sci USA 105:5266–5270
Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, Confavreux C, Dacquin R, Mee PJ, McKee MD, Jung DY (2007) Endocrine regulation of energy metabolism by the skeleton. Cell 130:456–469
Brennan-Speranza TC, Conigrave AD (2015) Osteocalcin: an osteoblast-derived polypeptide hormone that modulates whole body energy metabolism. Calcif Tissue Int 96:1–10
Kanazawa I (2015) Osteocalcin as a hormone regulating glucose metabolism. World J Diabetes 6:1345
Ferron M, McKee MD, Levine RL, Ducy P, Karsenty G (2012) Intermittent injections of osteocalcin improve glucose metabolism and prevent type 2 diabetes in mice. Bone 50:568–575
Zhou B, Li H, Xu L, Zang W, Wu S, Sun H (2013) Osteocalcin reverses endoplasmic reticulum stress and improves impaired insulin sensitivity secondary to diet-induced obesity through nuclear factor-κB signaling pathway. Endocrinol 154:1055–1068
Levinger I, Lin X, Zhang X, Brennan-Speranza TC, Volpato B, Hayes A, Jerums G, Seeman E, McConell G (2016) The effects of muscle contraction and recombinant osteocalcin on insulin sensitivity ex vivo. Osteoporos Int 27:653–663
Tsuka S, Aonuma F, Higashi S, Ohsumi T, Nagano K, Mizokami A, Kawakubo-Yasukochi T, Masaki C, Hosokawa R, Hirata M (2015) Promotion of insulin-induced glucose uptake in C2C12 myotubes by osteocalcin. Biochem Biophys Res Commun 459:437–442
Lin X, Parker L, Mclennan E, Zhang X, Hayes A, McConell GK, Brennan-Speranza TC, Levinger I (2017) Recombinant uncarboxylated osteocalcin per se enhances mouse skeletal muscle Glucose uptake in both extensor digitorum longus and soleus muscles. Front Endocrinol 8:1–11
Zierath JR, Hawley JA (2004) Skeletal muscle fiber type: influence on contractile and metabolic properties. PLoS Biol 2:e348
Westerblad H, Bruton JD, Katz A (2010) Skeletal muscle: energy metabolism, fiber types, fatigue and adaptability. Exp Cell Res 316:3093–3099
Wang Y-X, Zhang C-L, Ruth TY, Cho HK, Nelson MC, Bayuga-Ocampo CR, Ham J, Kang H, Evans RM (2004) Regulation of muscle fiber type and running endurance by PPARδ. PLoS Biol 2:e294
James DE, Jenkins AB, Kraegen EW (1985) Heterogeneity of insulin action in individual muscles in vivo: euglycemic clamp studies in rats. Am J Physiol Endocrinol Metab 248:E567–E574
Marette A, Richardson JM, Ramlal T, Balon TW, Vranic M, Pessin JE, Klip A (1992) Abundance, localization, and insulin-induced translocation of glucose transporters in red and white muscle. Am J Physiol Cell Physiol 263:C443–C452
Kjøbsted R, Munk-Hansen N, Birk JB, Foretz M, Viollet B, Björnholm M, Zierath JR, Treebak JT, Wojtaszewski JF (2017) Enhanced muscle insulin sensitivity after contraction/exercise is mediated by AMPK. Diabetes 66:598–612
Lee WJ, Song K-H, Koh EH, Won JC, Kim HS, Park H-S, Kim M-S, Kim S-W, Lee K-U, Park J-Y (2005) α-Lipoic acid increases insulin sensitivity by activating AMPK in skeletal muscle. Biochem Biophys Res Commun 332:885–891
Mera P, Laue K, Ferron M, Confavreux C, Wei J, Galán-Díez M, Lacampagne A, Mitchell SJ, Mattison JA, Chen Y (2016) Osteocalcin signaling in myofibers is necessary and sufficient for optimum adaptation to exercise. Cell Metab 23:1078–1092
Lin X, Hanson E, Betik AC, Brennan-Speranza TC, Hayes A, Levinger I (2016) Hindlimb immobilization, but not castration, induces reduction of undercarboxylated osteocalcin associated with muscle atrophy in rats. J Bone Miner Res 31:1967–1978
Zhou B, Li H, Liu J, Xu L, Guo Q, Zang W, Sun H, Wu S (2016) Autophagic dysfunction is improved by intermittent administration of osteocalcin in obese mice. Int J Obes 40:833–843
Jensen J, O’Rahilly S (2017) AMPK is required for exercise to enhance insulin sensitivity in skeletal muscles. Mol Metab 6:315
Sriwijitkamol A, Coletta DK, Wajcberg E, Balbontin GB, Reyna SM, Barrientes J, Eagan PA, Jenkinson CP, Cersosimo E, DeFronzo RA (2007) Effect of acute exercise on AMPK signaling in skeletal muscle of subjects with type 2 diabetes. Diabetes 56:836–848
Ishikura S, Bilan PJ, Klip A (2007) Rabs 8A and 14 are targets of the insulin-regulated Rab-GAP AS160 regulating GLUT4 traffic in muscle cells. Biochem Biophys Res Commun 353:1074–1079
Geraghty KM, Chen S, Harthill JE, Ibrahim AF, Toth R, Morrice NA, Vandermoere F, Moorhead GB, Hardie DG, MacKintosh C (2007) Regulation of multisite phosphorylation and 14-3-3 binding of AS160 in response to IGF-1, EGF, PMA and AICAR. Biochem J 407:231–241
Liu S, Gao F, Wen L, Ouyang M, Wang Y, Wang Q, Luo L, Jian Z (2017) Osteocalcin induces proliferation via positive activation of the PI3K/Akt, P38 MAPK pathways and promotes differentiation through activation of the GPRC6A-ERK1/2 pathway in C2C12 myoblast cells. Cell Physiol Biochem 43:1100–1112
Ren J-M, Semenkovich CF, Gulve EA, Gao J, Holloszy JO (1994) Exercise induces rapid increases in GLUT4 expression, glucose transport capacity, and insulin-stimulated glycogen storage in muscle. J Biol Chem 269:14396–14401
Beg M, Abdullah N, Thowfeik FS, Altorki NK, McGraw TE (2017) Distinct Akt phosphorylation states are required for insulin regulated Glut4 and Glut1-mediated glucose uptake. eLife 6:1–22
McMillin SL, Schmidt DL, Kahn BB, Witczak CA (2017) Glucose transporter 4 (GLUT4) is not necessary for overload-induced glucose uptake or hypertrophic growth in mouse skeletal muscle. Diabetes 66:1491–1500
Acknowledgements
Associate Professor Itamar Levinger is a Future Leader Fellow supported by Australian Heart Foundation (ID: 100040).
Author information
Authors and Affiliations
Contributions
XL, XZ, TS, and IL designed the study; XL, LP, and EM performed experiments; XL and LP analyzed the data; XL, LP, AH, GM, TS, and IL interpreted the results; XL wrote the first draft of the paper; XL, LP, XZ, AH, GM, TS, and IL edited and revised the manuscript; All authors approved final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Xuzhu Lin, Lewan Parker, Emma Mclennan, Xinmei Zhang, Alan Hayes, Glenn McConell, Tara C. Brennan-Speranza, and Itamar Levinger declare no conflict of interest.
Human and Animal Rights and Informed Consent
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Lin, X., Parker, L., Mclennan, E. et al. Uncarboxylated Osteocalcin Enhances Glucose Uptake Ex Vivo in Insulin-Stimulated Mouse Oxidative But Not Glycolytic Muscle. Calcif Tissue Int 103, 198–205 (2018). https://doi.org/10.1007/s00223-018-0400-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-018-0400-x