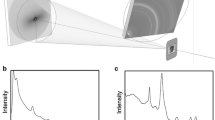
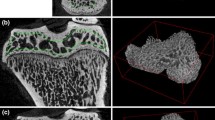
Abstract
The effect of SrCl2 treatment on bone nanostructure in a rat ovariectomy model was studied using scanning small-angle X-ray scattering (sSAXS). Twelve 6-month-old female Wistar rats were used. Six animals were ovariectomized (+ovx) and six were left intact after sham surgery (−ovx). Six animals, three +ovx and three −ovx, were treated with 4 mmol SrCl2 (aq)/kg/day (+Sr), whereas the remaining six received placebo (−Sr) for 140 days. Rats were labeled with flourochromes at days 7, 126, and 136. Femoral cross sections were studied using fluorescence microscopy, scanning electron microscopy including energy-dispersive X-ray analysis, and sSAXS. The SAXS data comprised about 5,500 measurements and provided information about mineral crystal thickness and orientation in new and old bone. The newly formed bone contained higher levels of Sr2+ in +Sr than in −Sr animals, indicating that the Sr2+ was incorporated into the new bone. Mineral plates were significantly thicker in old bone, 2.62 nm (95% CI 2.58–2.66), than in new bone, 2.41 nm (95% CI 2.36–2.46). Surprisingly, mineral plates in new bone were significantly thicker (2.52 [95% CI 2.47–2.57] nm vs. 2.41 [95% CI 2.36–2.46] nm, P = 0.017) in +ovx rats than in −ovx rats. However, no significant effect of SrCl2 on mineral plate thicknesses in new bone was observed. The statistical model yielded estimates of the difference in bone mineral plate thickness induced by Sr. The estimated effect of Sr was −0.09 (95% CI −0.21 to 0.03) and 0.02 (95% CI −0.10 to 0.14) nm for new bone in −ovx and +ovx rats, respectively.





Similar content being viewed by others
References
Marie PJ (2005) Strontium as therapy for osteoporosis. Curr Opin Pharmacol 5:633–636
Delannoy P, Bazot D, Marie PJ (2002) Long-term treatment with strontium ranelate increases vertebral bone mass without deleterious effect in mice. Metabolism 51:906–911
Marie PJ, Hott M, Modrowski D, De Pollak C, Guillemain J, Deloffre P, Tsouderos Y (1993) An uncoupling agent containing strontium prevents bone loss by depressing bone resorption and maintaining bone formation in estrogen-deficient rats. J Bone Miner Res 8:607–615
Marie PJ, Garba MT, Hott M, Miravet L (1985) Effect of low doses of stable strontium on bone metabolism in rats. Miner Electrolyte Metab 11:5–13
Buehler J, Chappuis P, Saffar JL, Tsouderos Y, Vignery A (2001) Strontium ranelate inhibits bone resorption while maintaining bone formation in alveolar bone in monkeys (Macaca fascicularis). Bone 29:176–179
Grynpas MD, Marie PJ (1990) Effects of low doses of strontium on bone quality and quantity in rats. Bone 11:313–319
Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel EM, Pors-Nielsen S, Rizzoli R, Genant HK, Reginster JY (2004) The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 350:459–468
Reginster JY, Deroisy R, Dougados M, Jupsin I, Colette J, Roux C (2002) Prevention of early postmenopausal bone loss by strontium ranelate: the randomized, two-year, double-masked, dose-ranging, placebo-controlled PREVOS trial. Osteoporos Int 13:925–931
Reginster JY, Seeman E, De Vernejoul MC, Adami S, Compston J, Phenekos C, Devogelaer JP, Curiel MD, Sawicki A, Goemaere S, Sorensen OH, Felsenberg D, Meunier PJ (2005) Strontium ranelate reduces the risk of nonvertebral fractures in postmenopausal women with osteoporosis: treatment of Peripheral Osteoporosis (TROPOS) study. J Clin Endocrinol Metab 90:2816–2822
Marie PJ, Ammann P, Boivin G, Rey C (2001) Mechanisms of action and therapeutic potential of strontium in bone. Calcif Tissue Int 69:121–129
Omdahl JL, DeLuca HF (1971) Strontium induced rickets: metabolic basis. Science 174:949–951
Cabrera WE, Schrooten I, De Broe ME, D’Haese PC (1999) Strontium and bone. J Bone Miner Res 14:661–668
Neufeld EB, Boskey AL (1994) Strontium alters the complexed acidic phospholipid content of mineralizing tissues. Bone 15:425–430
Jethi RK, Wadkins CL, Mackey MG, Meredith PD (1972) Studies of mechanism of biological calcification. 3. Interaction of strontium with a calcifiable matrix from beef tendon. Calcif Tissue Res 9:310–324
Boivin G, Meunier PJ (2003) The mineralization of bone tissue: a forgotten dimension in osteoporosis research. Osteoporos Int 14(Suppl 3):S19–S24
Mann S (2002) Biomineralization, Principles and concepts in bioinorganic materials chemistry. Oxford University Press, Oxford
Weiner S, Traub W (1992) Bone structure: from angstroms to microns. FASEB J 6:879–885
Landis WJ (1999) An overview of vertebrate mineralization with emphasis on collagen-mineral interaction. Gravit Space Biol Bull 12:15–26
Ammann P, Shen V, Robin B, Mauras Y, Bonjour JP, Rizzoli R (2004) Strontium ranelate improves bone resistance by increasing bone mass and improving architecture in intact female rats. J Bone Miner Res 19:2012–2020
Christoffersen J, Christoffersen MR, Kolthoff N, Barenholdt O (1997) Effects of strontium ions on growth and dissolution of hydroxyapatite and on bone mineral detection. Bone 20:47–54
Farlay D, Boivin G, Panczer G, Lalande A, Meunier PJ (2005) Long-term strontium ranelate administration in monkeys preserves characteristics of bone mineral crystals and degree of mineralization of bone. J Bone Miner Res 20:1569–1578
Skoryna SC (1981) Effects of oral supplementation with stable strontium. Can Med Assoc J 125:703–712
Dahl SG, Allain P, Marie PJ, Mauras Y, Boivin G, Ammann P, Tsouderos Y, Delmas PD, Christiansen C (2001) Incorporation and distribution of strontium in bone. Bone 28:446–453
Grynpas MD, Hamilton E, Cheung R, Tsouderos Y, Deloffre P, Hott M, Marie PJ (1996) Strontium increases vertebral bone volume in rats at a low dose that does not induce detectable mineralization defect. Bone 18:253–259
Fratzl P, Groschner M, Vogl G, Plenk H Jr, Eschberger J, Fratzl-Zelman N, Koller K, Klaushofer K (1992) Mineral crystals in calcified tissues: a comparative study by SAXS. J Bone Miner Res 7:329–334
Fratzl P, Schreiber S, Klaushofer K (1996) Bone mineralization as studied by small-angle X-ray scattering. Connect Tissue Res 34:247–254
Hauge Bunger M, Foss M, Erlacher K, Bruun Hovgaard M, Chevallier J, Langdahl B, Bunger C, Birkedal H, Besenbacher F, Skov Pedersen J (2006) Nanostructure of the neurocentral growth plate: insight from scanning small angle X-ray scattering, atomic force microscopy and scanning electron microscopy. Bone 39:530–541
Rinnerthaler S, Roschger P, Jakob HF, Nader A, Klaushofer K, Fratzl P (1999) Scanning small angle X-ray scattering analysis of human bone sections. Calcif Tissue Int 64:422–429
Oxlund HTJ, Andreassen TT (2005) Low-dose strontium increased the formation of new lamellar bone at the periosteal surface, with normal mechanical competence. J Bone Miner Res 20(Suppl 1):S414
Pedersen JS (2004) A flux- and background-optimized version of the NanoSTAR small-angle X-ray scattering camera for solution scattering. J Appl Crystallogr 37:369–380
Fratzl P (1994) Statistical model of the habit and arrangement of mineral crystals in the collagen of bone. J Stat Phys 77:125–143
Fratzl P, Fratzl-Zelman N, Klaushofer K, Vogl G, Koller K (1991) Nucleation and growth of mineral crystals in bone studied by small-angle X-ray scattering. Calcif Tissue Int 48:407–413
Fratzl P, Roschger P, Eschberger J, Abendroth B, Klaushofer K (1994) Abnormal bone mineralization after fluoride treatment in osteoporosis: a small-angle X-ray-scattering study. J Bone Miner Res 9:1541–1549
Fratzl P, Schreiber S, Boyde A (1996) Characterization of bone mineral crystals in horse radius by small-angle X-ray scattering. Calcif Tissue Int 58:341–346
Fratzl P, Schreiber S, Roschger P, Lafage MH, Rodan G, Klaushofer K (1996) Effects of sodium fluoride and alendronate on the bone mineral in minipigs: a small-angle X-ray scattering and backscattered electron imaging study. J Bone Miner Res 11:248–253
Bunger MH, Foss M, Erlacher K, Li H, Zou X, Langdahl BL, Bunger C, Birkedal H, Besenbacher F, Pedersen JS (2006) Bone nanostructure near titanium and porous tantalum implants studied by scanning small angle X-ray scattering. Eur Cell Mater 12:81–91
Lindner P, Zemb T (2002) Neutrons, X-rays and light: scattering methods applied to soft condensed matter. Elsevier Science, Amsterdam
Shimada T, Doi M, Okano K (1988) Concentration fluctuation in stiff polymers. I. Static structure factor. J Chem Phys 88:2815–2821
Siperko LM, Landis WJ (2001) Aspects of mineral structure in normally calcifying avian tendon. J Struct Biol 135:313–320
Weiner S, Traub W, Wagner HD (1999) Lamellar bone: structure–function relations. J Struct Biol 126:241–255
Camacho NP, Rinnerthaler S, Paschalis EP, Mendelsohn R, Boskey AL, Fratzl P (1999) Complementary information on bone ultrastructure from scanning small angle X-ray scattering and Fourier-transform infrared microspectroscopy. Bone 25:287–293
Roschger P, Grabner BM, Rinnerthaler S, Tesch W, Kneissel M, Berzlanovich A, Klaushofer K, Fratzl P (2001) Structural development of the mineralized tissue in the human L4 vertebral body. J Struct Biol 136:126–136
Danielsen CC, Mosekilde L, Svenstrup B (1993) Cortical bone mass, composition, and mechanical properties in female rats in relation to age, long-term ovariectomy, and estrogen substitution. Calcif Tissue Int 52:26–33
Kim BT, Mosekilde L, Duan Y, Zhang XZ, Tornvig L, Thomsen JS, Seeman E (2003) The structural and hormonal basis of sex differences in peak appendicular bone strength in rats. J Bone Miner Res 18:150–155
Thompson DD, Simmons HA, Pirie CM, Ke HZ (1995) FDA guidelines and animal models for osteoporosis. Bone 17(Suppl 4):125S–133S
Turner RT, Vandersteenhoven JJ, Bell NH (1987) The effects of ovariectomy and 17 beta-estradiol on cortical bone histomorphometry in growing rats. J Bone Miner Res 2:115–122
Ammann P (2005) Strontium ranelate: a novel mode of action leading to renewed bone. Osteoporos Int 16(Suppl 1):S11–S15
Ammann P (2006) Strontium ranelate: a physiological approach for an improved bone quality. Bone 38(Suppl 1):15–18
Ammann P, Badoud I, Barraud S, Dayer R, Rizzoli R (2007) Strontium ranelate treatment improves trabecular and cortical intrinsic bone tissue quality, a determinant of bone strength. J Bone Miner Res 22:1419–1425
Fantner GE, Hassenkam T, Kindt JH, Weaver JC, Birkedal H, Pechenik L, Cutroni JA, Cidade GA, Stucky GD, Morse DE, Hansma PK (2005) Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat Mater 4:612–616
Acknowledgements
We thank Jacques Chevallier at the Institute of Physics and Astronomy, University of Aarhus, for his kind technical assistance. H. B. thanks the Danish Natural Sciences Research Council for funding through a Skou research associate professor fellowship. Hanne Broch at the Institute of Anatomy is gratefully acknowledged for handling the animals.
Author information
Authors and Affiliations
Corresponding author
Additional information
Rights and permissions
About this article
Cite this article
Bünger, M.H., Oxlund, H., Hansen, T.K. et al. Strontium and Bone Nanostructure in Normal and Ovariectomized Rats Investigated by Scanning Small-Angle X-Ray Scattering. Calcif Tissue Int 86, 294–306 (2010). https://doi.org/10.1007/s00223-010-9341-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-010-9341-8