Abstract
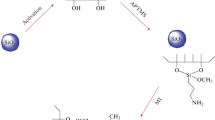
In this study, a ternary mixed-mode silica sorbent (TMSS) with octamethylene, carboxyl, and amino groups was prepared via Cu(I)-catalyzed azide-alkyne cycloaddition (CuAAC) click reaction and a subsequent reduction of azide to primary amine. While used in solid-phase extraction (SPE), the retention behavior of TMSS towards a total of nine kinds of basic, neutral, and acidic drugs was investigated in detail. The results revealed that hydrophobic, ion-exchange interaction, and electrostatic repulsion between TMSS and the analytes were closely related to the retention behavior of TMSS. Besides, the log Kow value of the analyte was also a factor influencing the retention behavior of analytes on TMSS. The nine analytes could be retained by TMSS simultaneously and then, were eluted into two fractions according to the acid-base property of the analytes for further determinations. The acidic and neutral analytes were in one fraction, and the basic ones in the other fraction. When used to treat the human serum spiked with the nine drugs, TMSS offered higher recoveries than BakerBond CBA and comparable recoveries to Oasis WCX. It should be noted TMSS had better purifying capability for human serum than Oasis WCX. Under the optimized SPE conditions, a method of SPE hyphenated to high-performance liquid chromatography-ultraviolet detection (HPLC-UV) for determination of the basic, neutral, and acidic drugs spiked in human serum was established. For the nine drugs, the linear ranges were all between 5.0 and 1000 μg L−1 with correlation coefficients (R2) above 0.9990, and the limits of detection (LODs) were in the range of 0.8–2.3 μg L−1. The intra-day and inter-day relative standard deviations (RSDs) were less than 5.3 and 8.8%, respectively.

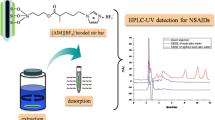
Treating drugs in human serum by SPE with ternary mixed-mode silica sorbent.








Similar content being viewed by others
References
Delamoye M, Duverneuil C, Paraire F, Mazancourt PD, Alvarez J-C. Simultaneous determination of thirteen β-blockers and one metabolite by gradient high-performance liquid chromatography with photodiode-array UV detection. Forensic Sci Int. 2004;141(1):23–31.
Braza AJ, Modamio P, Mariño EL. Two reproducible and sensitive liquid chromatographic methods to quantify atenolol and propranolol in human plasma and determination of their associated analytical error functions. J Chromatogr B. 2000;738(2):225–31.
Li Y, Yang JJ, Huang CN, Wang LX, Wang JC, Chen JP. Dendrimer-functionalized mesoporous silica as a reversed-phase/anion-exchange mixed-mode sorbent for solid phase extraction of acid drugs in human urine. J Chromatogr A. 2015;1392:28–36.
Boonjob W, Sklenarova H, Lara FJ, Garcia-Campana AM, Solich P. Retention and selectivity of basic drugs on solid-phase extraction sorbents: application to direct determination of beta-blockers in urine. Anal Bioanal Chem. 2014;406(17):4207–15.
Laven M, Alsberg T, Yu Y, Adolfsson-Erici M, Sun HW. Serial mixed-mode cation- and anion-exchange solid-phase extraction for separation of basic, neutral and acidic pharmaceuticals in wastewater and analysis by high-performance liquid chromatography-quadrupole time-of-flight mass spectrometry. J Chromatogr A. 2009;1216(1):49–62.
Hu XG, Pan JL, Hu YL, Li GK. Preparation and evaluation of propranolol molecularly imprinted solid-phase microextraction fiber for trace analysis of beta-blockers in urine and plasma samples. J Chromatogr A. 2009;1216(2):190–7.
Mullett WM, Levsen K, Lubda D, Pawliszyn J. Bio-compatible in-tube solid-phase microextraction capillary for the direct extraction and high-performance liquid chromatographic determination of drugs in human serum. J Chromatogr A. 2002;963(1–2):325–34.
Zhu YL, Yang SW, Chen G, Xing J. Single “click” synthesis of a mixed-mode silica sorbent and application in matrix solid-phase dispersion extraction of β-agonists from porcine liver. J Chromatogr A. 2014;1354:101–8.
Meissner G, Hartonen K, Riekkola ML. Supercritical fluid extraction combined with solid phase extraction as sample preparation technique for the analysis of β-blockers in serum and urine. Fresenius J Anal Chem. 1998;360(5):618–21.
Zhang P, Bui A, Rose G, Allinson G. Mixed-mode solid-phase extraction coupled with liquid chromatography tandem mass spectrometry to determine phenoxy acid, sulfonylurea, triazine and other selected herbicides at nanogram per litre levels in environmental waters. J Chromatogr A. 2014;1325:56–64.
Tölgyesi Á, Sharma VK, Fekete J. Confirmatory analysis of stanozolol metabolites in bovine, pig and sheep urines using an optimized clean-up and liquid chromatography-tandem mass spectrometry. J Pharm Biomed Anal. 2014;88:45–52.
Montes R, García-López M, Rodríguez I, Cela R. Mixed-mode solid-phase extraction followed by acetylation and gas chromatography mass spectrometry for the reliable determination of trans-resveratrol in wine samples. Anal Chim Acta. 2010;673(1):47–53.
Tölgyesi Á, Tölgyesi L, Sharma VK, Sohn M, Fekete J. Quantitative determination of corticosteroids in bovine milk using mixed-mode polymeric strong cation exchange solid-phase extraction and liquid chromatography-tandem mass spectrometry. J Pharm Biomed Anal. 2010;53(4):919–28.
Augusto F, Hantao LW, Mogollón NGS, Braga SCGN. New materials and trends in sorbents for solid-phase extraction. Trends Analyt Chem. 2013;43:14–23.
Fontanals N, Marcé RM, Borrull F, Cormack PAG. Mixed-mode ion-exchange polymeric sorbents: dual-phase materials that improve selectivity and capacity. Trends Analyt Chem. 2010;29(7):765–79.
Mayani VJ, Abdi SHR, Kureshy RI, Khan NH, Agrawal S, Jasra RV. Synthesis and characterization of mesoporous silica modified with chiral auxiliaries for their potential application as chiral stationary phase. J Chromatogr A. 2008;1191(1–2):223–30.
Cho YJ, Choi HJ, Hyun MH. Preparation of two new liquid chromatographic chiral stationary phases based on diastereomeric chiral crown ethers incorporating two different chiral units and their applications. J Chromatogr A. 2008;1191(1–2):193–8.
Vidal L, Parshintsev J, Hartonen K, Canals A, Riekkola ML. Ionic liquid-functionalized silica for selective solid-phase extraction of organic acids, amines and aldehydes. J Chromatogr A. 2012;1226:2–10.
Kolb HC, Finn MG, Sharpless KB. Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed. 2001;40(11):2004–21.
El-Sagheer AH, Brown T. Click nucleic acid ligation: applications in biology and nanotechnology. Acc Chem Res. 2012;45(8):1258–67.
Rana S, Cho JW. Core-shell morphology and characterization of carbon nanotube nanowires click coupled with polypyrrole. Nanotechnology. 2011;22(27):275609.
Punna S, Kaltgrad E, Finn MG. Clickable agarose for affinity chromatography. Bioconjug Chem. 2005;16(6):1536–41.
Cavalieri F, Postma A, Lee L, Caruso F. Assembly and functionalization of DNA-polymer microcapsules. ACS Nano. 2009;3(1):234–40.
Gierlich J, Burley GA, Gramlich PME, Hammond DM, Carell T. Click chemistry as a reliable method for the high-density postsynthetic functionalization of alkyne-modified DNA. Org Lett. 2006;8(17):3639–42.
Slater M, Snauko M, Svec F, Fréchet JMJ. “Click chemistry” in the preparation of porous polymer-based particulate stationary phases for μ-HPLC separation of peptides and proteins. Anal Chem. 2006;78(14):4969–75.
Yang F, Mao J, He XW, Chen LX, Zhang YK. Preparation of a boronate-functionalized affinity hybrid monolith for specific capture of glycoproteins. Anal Bioanal Chem. 2013;405(15):5321–31.
Guo ZM, Jin Y, Liang T, Liu YF, Xu Q, Liang XM, et al. Synthesis, chromatographic evaluation and hydrophilic interaction/reversed-phase mixed-mode behavior of a “click β-cyclodextrin” stationary phase. J Chromatogr A. 2009;1216(2):257–63.
Pal B, Jaisankar P, Giri VS. Versatile reagent for reduction of azides to amines. Synth Commun. 2004;34(7):1317–23.
Zhu YL, Zhang WY, Li LX, Wu CY, Xing J. Preparation of a mixed-mode silica-based sorbent by click reaction and its application in the determination of primary aromatic amines in environmental water samples. Anal Methods. 2014;6(7):2102–11.
Bi W, Zhou J, Row KH. Solid phase extraction of lactic acid from fermentation broth by anion-exchangeable silica confined ionic liquids. Talanta. 2011;83(3):974–9.
Xu J, Zhang Z, He X-M, Wang R-Q, Hussain D, Feng Y-Q. Immobilization of zirconium-glycerolate nanowires on magnetic nanoparticles for extraction of urinary ribonucleosides. Microchim Acta. 2017;185(1):43.
Vieno NM, Tuhkanen T, Kronberg L. Analysis of neutral and basic pharmaceuticals in sewage treatment plants and in recipient rivers using solid phase extraction and liquid chromatography–tandem mass spectrometry detection. J Chromatogr A. 2006;1134(1):101–11.
Lu Q, Zhao Q, Yu QW, Feng YQ. Use of pollen solid-phase extraction for the determination of trans-resveratrol in peanut oils. J Agric Food Chem. 2015;63(19):4771–6.
Samadi S, Sereshti H, Assadi Y. Ultra-preconcentration and determination of thirteen organophosphorus pesticides in water samples using solid-phase extraction followed by dispersive liquid–liquid microextraction and gas chromatography with flame photometric detection. J Chromatogr A. 2012;1219:61–5.
Liu Y, Zhao E, Zhu W, Gao H, Zhou Z. Determination of four heterocyclic insecticides by ionic liquid dispersive liquid–liquid microextraction in water samples. J Chromatogr A. 2009;1216(6):885–91.
Li H, Ai L, Fan S, Wang Y, Sun D. Rapid determination of 18 glucocorticoids in serum using reusable on-line SPE polymeric monolithic column coupled with LC-quadrupole/orbitrap high-resolution mass spectrometer. J Chromatogr B. 2017;1065-1066:79–86.
Aresta A, Bianchi D, Calvano CD, Zambonin CG. Solid phase microextraction-liquid chromatography (SPME-LC) determination of chloramphenicol in urine and environmental water samples. J Pharm Biomed Anal. 2010;53(3):440–4.
Ahmad S, Tucker M, Spooner N, Murnane D, Gerhard U. Direct ionization of solid-phase microextraction fibers for quantitative drug bioanalysis: from peripheral circulation to mass spectrometry detection. Anal Chem. 2015;87(1):754–9.
Farhadi K, Firuzi M, Hatami M. Stir bar sorptive extraction of propranolol from plasma samples using a steel pin coated with a polyaniline and multiwall carbon nanotube composite. Microchim Acta. 2015;182(1):323–30.
Xue S-W, Li J, Xu L. Preparation of magnetic melamine-formaldehyde resin and its application to extract nonsteroidal anti-inflammatory drugs. Anal Bioanal Chem. 2017;409(12):3103–13.
Souza-Silva ÉA, Reyes-Garcés N, Gómez-Ríos GA, Boyacı E, Bojko B, Pawliszyn J. A critical review of the state of the art of solid-phase microextraction of complex matrices III. Bioanalytical and clinical applications. Trends Analyt Chem. 2015;71:249–64.
Honor AS, Pedersen-Bjergaard S. Bioanalysis of pharmaceuticals: sample preparation, separation techniques and mass spectrometry. 1st edn. Hoboken: John Wiley & Sons; 2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study has been approved by the Hospital of Wuhan University Ethics Committee and has been performed in accordance with the ethical standards.
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that there is no conflict of interest and all the authors have materially participated in the research and/or article preparation.
Electronic supplementary material
ESM 1
(PDF 340 kb)
Rights and permissions
About this article
Cite this article
Jin, S., Qiao, Y. & Xing, J. Ternary mixed-mode silica sorbent of solid-phase extraction for determination of basic, neutral and acidic drugs in human serum. Anal Bioanal Chem 410, 3731–3742 (2018). https://doi.org/10.1007/s00216-018-1037-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-1037-3