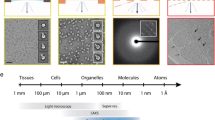
Abstract
The 2017 Nobel Prize in Chemistry was awarded to Jacques Dubochet, Joachim Frank, and Richard Henderson for “developing cryo-electron microscopy (cryo-EM) for the high-resolution structure determination of biomolecules in solution.” This feature article summarizes some of the major achievements leading to the development of cryo-EM and recent technological breakthroughs that have transformed the method into a mainstream tool for structure determination.


Similar content being viewed by others
References
Henderson R, Unwin PNT. Three-dimensional model of purple membrane obtained by electron microscopy. Nature. 1975;257:28–32. https://doi.org/10.1038/257028a0.
De Rosier DJ, Klug A. Reconstruction of three dimensional structures from electron micrographs. Nature. 1968;217:130–4. https://doi.org/10.1038/217130a0.
Henderson R, Baldwin JM, Ceska TA, Zemlin F, Beckmann E, Downing KH. Model for the structure of bacteriorhodopsin based on high-resolution electron cryo-microscopy. J Mol Biol. 1990;213:899–929. https://doi.org/10.1016/S0022-2836(05)80271-2.
Frank J. Averaging of low exposure electron micrographs of non-periodic objects. Ultramicroscopy. 1975;1:159–62. https://doi.org/10.1016/S0304-3991(75)80020-9.
van Heel M, Frank J. Use of multivariates statistics in analysing the images of biological macromolecules. Ultramicroscopy. 1981;6:187–94. https://doi.org/10.1016/S0304-3991(81)80197-0.
Radermacher M, Wagenknecht T, Verschoor A, Frank J. A new 3-D reconstruction scheme applied to the 50S ribosomal subunit of E. coli. J Microsc. 1986;141:RP1–2. https://doi.org/10.1111/j.1365-2818.1986.tb02693.x.
Taylor KA, Glaeser RM. Electron diffraction of frozen, hydrated protein crystals. Science (80- ). 1974;186:1036–7. https://doi.org/10.1126/science.186.4168.1036.
Dubochet J, McDowall AW. Vitrification of pure water for electron microscopy. J Microsc. 1981;124:3–4. https://doi.org/10.1111/j.1365-2818.1981.tb02483.x.
Adrian M, Dubochet J, Lepault J, McDowall AW. Cryo-electron microscopy of viruses. Nature. 1984;308:32–6. https://doi.org/10.1038/308032a0.
Dubochet J, Adrian M, Chang JJ, Homo JC, Lepault J, McDowall AW, et al. Cryo-electron microscopy of vitrified specimens. Q Rev Biophys. 1988;21:129–228. https://doi.org/10.1017/S0033583500004297.
Brilot AF, Chen JZ, Cheng A, Pan J, Harrison SC, Potter CS, et al. Beam-induced motion of vitrified specimen on holey carbon film. J Struct Biol. 2012;177:630–7. https://doi.org/10.1016/j.jsb.2012.02.003.
Campbell MG, Cheng A, Brilot AF, Moeller A, Lyumkis D, Veesler D, et al. Movies of ice-embedded particles enhance resolution in electron cryo-microscopy. Structure. 2012;20:1823–8. https://doi.org/10.1016/j.str.2012.08.026.
Grant T, Grigorieff N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. Elife. 2015;4. https://doi.org/10.7554/eLife.06980.
Sigworth FJ. A maximum-likelihood approach to single-particle image refinement. J Struct Biol. 1998;122:328–39. https://doi.org/10.1006/jsbi.1998.4014.
Scheres SHW. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J Struct Biol. 2012;180:519–30. https://doi.org/10.1016/j.jsb.2012.09.006.
Bai X, Fernandez IS, McMullan G, Scheres SH. Ribosome structures to near-atomic resolution from thirty thousand cryo-EM particles. elife. 2013;2:e00461. https://doi.org/10.7554/eLife.00461.
Liao M, Cao E, Julius D, Cheng Y. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature. 2013;504:107–12. https://doi.org/10.1038/nature12822.
Merk A, Bartesaghi A, Banerjee S, Falconieri V, Rao P, Davis MI, et al. Breaking cryo-EM resolution barriers to facilitate drug discovery. Cell. 2016;165:1698–707. https://doi.org/10.1016/j.cell.2016.05.040.
Khoshouei M, Radjainia M, Baumeister W, Danev R. Cryo-EM structure of haemoglobin at 3.2 Å determined with the Volta phase plate. Nat Commun. 2017;8. https://doi.org/10.1038/ncomms16099.
Danev R, Buijsse B, Khoshouei M, Plitzko JM, Baumeister W. Volta potential phase plate for in-focus phase contrast transmission electron microscopy. Proc Natl Acad Sci. 2014;111:15635–40. https://doi.org/10.1073/pnas.1418377111.
Taylor KA, Glaeser RM. Retrospective on the early development of cryoelectron microscopy of macromolecules and a prospective on opportunities for the future. Q Rev Biophys. 1988;163:214–23. https://doi.org/10.1016/j.jsb.2008.06.004.
Noble AJ, Dandey VP, Wei H, Brasch J, Chase J, Acharya P, et al. Routine single particle cryoEM sample and grid characterization by tomography. bioRxiv 230276. 2017. https://doi.org/10.1101/230276.
Dandey VP, Wei H, Zhang Z, Tan YZ, Acharya P, Eng ET, et al. Spotiton: new features and applications. bioRxiv 230151. 2017. https://doi.org/10.1101/230151.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, P.S. The 2017 Nobel Prize in Chemistry: cryo-EM comes of age. Anal Bioanal Chem 410, 2053–2057 (2018). https://doi.org/10.1007/s00216-018-0899-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-018-0899-8