Abstract
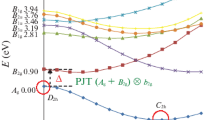
The bent D2d structures of the four-membered homo-cyclic compounds A4 (A = O, S, Se, Te, Po) were examined computationally to understand the pseudo-Jahn–Teller effect (PJTE). To do this, ab initio geometry optimizations and corresponding frequency calculations (at the MP2/cc-pVQZ-(PP) level of theory) show that all A4 compounds under-consideration are unstable in their planar (D4h) configuration. The ground state and six low-lying non-degenerate and degenerate electronic excited states were computed at the CASSCF (6,7)/cc-pVQZ-(PP) along the bending normal coordinate connecting the D4h and D2d geometries; these represent the adiabatic potential energy surfaces (APESs). Based on the APESs, the coupling between the ground state (1A1g) and the 1B2u excited state is demonstrated to be the reason for the planar structure bends from the high-symmetry D4h geometry into the lower-symmetry D2d stable equilibrium configuration. The solution to the PJTE (1A1g + 1B2u) ⊗ b2u problem is useful to answer the question of “how instability rises in A4 planar configuration?”. Although all A4 compounds in the series are non-planar with D4h symmetry, but geometrical optimizations and frequency calculations show that coordination of two noble gas cations (NG+ = He+, Ne+ and Ar+) above and below the σh plane of the A4 (A = O, S, Se) ring could restore ring planarity in (A4 NG)2+ complexes. The PJTE is also quenched in the A42+ (A = O, S, Se, Te, Po) cation and dication series, and planarity of the rings is also restored, i.e., the high-symmetry D4h structure becomes the equilibrium configuration.




Similar content being viewed by others
References
Steudel R, Eckert B (2003) Top Curr Chem 230:1–79 (Steudel R (2003) Top Curr Chem 231:31–98)
Steudel R (1982) Top Curr Chem 102:149–176
Picquenard E, Boumedien MS, Corset J (1993) J Mol Struct 293:63–66
Boumedien MS, Corset J, Picquenard E (1999) J Raman Spectrosc 30:463–472
Brabson GD, Mielke Z, Andrews L (1991) J Phys Chem 95:79–86
Hassanzadeh P, Andrews L (1992) J Phys Chem 96:6579–6585
Meyer B, Stroyer-Hansen T, Oommen TV (1972) J Mol Spectrosc 42:335–343
Meyer B, Stroyer-Hansen T (1972) J Phys Chem 76:3968–3969
Weser G, Hensel F, Warren WW, Bunsenges B (1978) J Phys Chem 82:588–593
Krasnopolsky VA (1987) Adv Space Res 7:25–27
Berkowitz J, Chupka WA (1964) J Chem Phys 40:287–295
Drowart J, Goldfinger P, Detry D, Rickert H, Keller H (1968) Adv Mass Spectrom 4:499–510
Berkowitz J, Lifshitz C (1968) J Chem Phys 48:4346–4350
Lewis GN (1924) J Am Chem Soc 46:2027–2032
Lundegaard LF, Weck G, McMahon MI, Desgreniers S, Loubeyre P (2006) Nature 443:201–204
Krossing I (2003) Top Curr Chem 230:135–152
Stephens PJ (1969) Chem Comm 1615:1496–1506
Barr J, Gillespie RJ, Kapoor R, Malhotra KC (1968) Can J Chem 46:149–159
Barr J, Gillespie RJ, Kapoor R, Pez GP (1968) J Am Chem Soc 90:6855–6856
Brown ID, Crump DB, Gillespie RJ, Santry DP (1968) Chem Comm 602:853–854
Steudel R (2003) Top Curr Chem 231:203–230
Quelch GE, Schaefer HF, Marsden CJ (1990) J Am Chem Soc 112:8719–8733
Wong MW, Steudel R (2003) Chem Phys Lett 379:162–169
Raghavachari K, Rohlfing CM, Binkley JS (1990) J Chem Phys 93:5862–5874
Zakrzcwski VG, von Niessen W (1994) Theor Chim Acta 88:75–83
Abboud JLM, Essefar M, Herreros M, Mo O, Molina MT, Notario R, Yanez M (1998) J Phys Chem A 102:7996–8003
Chen MD, Liu ML, Luo HB, Zhang QE, Au CT (2001) J Mol Struct (THEOCHEM) 548:133–141
Millefiori S, Alparone A (2001) J Phys Chem A 105:9489–9497
Seidl ET, Schaefer HF (1988) J Chem Phys 88:7043–7049
Seidl ET, Schaefer HF (1992) J Chem Phys 96:1176–1182
Hernandez-Lamoneda R, Ramirez-Solis A (2000) J Chem Phys 113:4139–4145
Hernandez-Lamoneda R, Ramirez-Solis A (2004) J Chem Phys 120:10084–10088
Adamantides V, Neisius D, Verhaegen G (1980) Chem Phys 48:215–220
Gadzhiev OB, Ignatov SK, Kulikov MY, Feigin AM, Razuvaev AG, Sennikov PG, Schrems O (2013) J Chem Theo Comput 9:247–262
Bersuker IB (2006) The Jahn-Teller Effect. Cambridge University Press, Cambridge
Bersuker IB (2013) Chem Rev 113:1351–1390
Blancafort L, Bearpark MJ, Robb MA (2006) Mol Phys 104:2007–2010
Kim JH, Lee Z (2014) Appl Microscopy 44:123–132
Gromov EV, Trofimov AB, Vitkovskaya NM, Schirmer J, Koppel H (2003) J Chem Phys 119:737–751
Jose D, Datta A (2011) Phys Chem Chem Phys 13:7304–7311
Ilkhani AR (2015) J Theo Comput Chem 6:1550045
Ilkhani AR, Gorinchoy NN, Bersuker IB (2015) Chem Phys 460:106–110
Liu Y, Bersuker IB, Garcia-Fernandez P, Boggs JE (2012) J Phys Chem A 116:7564–7570
Ghosh M, Datta A (2018) Bull Mater Sci 41:117
Hermoso W, Ilkhani AR, Bersuker IB (2014) Comput Theo Chem 1049:109–114
Ilkhani AR (2017) Russian J Phys Chem A 91:1743–1751
Liu Y, Bersuker IB, Boggs JE (2013) Chem Phys 417:26–29
Ilkhani AR, Hermoso W, Bersuker IB (2015) Chem Phys 460:75–82
Bhattacharyya K, Surendran A, Chowdhury C, Datta A (2016) Phys Chem Chem Phys 18:31160–31167
Ilkhani AR, Monajjemi M (2015) Comput Theo Chem 1074:19–25
Ilkhani AR (2017) Quim Nova 40:491–495
Ilkhani AR (2015) J Mol Struc 1098:21–25
Ivanov AS, Bozhenko KV, Boldyrev AI (2012) Inorg Chem 51:8868–8872
Ilkhani AR, Wang Z (2019) Theo Chem Acc 138:14
Ilkhani AR (2019) J Chem Sci 131:37
Hampel C, Peterson KA, Werner HJ (1992) Chem Phys Lett 190:1–12
Deegan MJ, Knowles PJ (1994) Chem Phys Lett 227:321–326
Head-Gordon M, Pople JA, Frisch MJ (1988) Chem Phys Lett 153:503–506
Wilson AK, Woon DE, Peterson KA, Dunning TH Jr (1999) J Chem Phys 110:7667–7676
Dunning TH (1989) J Chem Phys 90:1007–1023
Woon DE, Dunning TH (1993) J Chem Phys 98:1358–1371
Peterson KA (2003) J Chem Phys 119:11099–11113
Dolg M (1996) Chem Phys Lett 250:75–79
Dolg M (1996) J Chem Phys 104:4061–4067
Werner HJ, Meyer W (1981) J Chem Phys 74:5794–5801
Werner HJ, Meyer W (1980) J Chem Phys 73:2342–2356
Werner HJ, Knowles PJ (1985) J Chem Phys 82:5053–5063
Werner HJ, Knowles PJ, Manby FR, Schutz M (2015) MOLPRO version 2015.1.22, a package of ab initio programs. https://www.molpro.net
Acknowledgements
This work was financially supported by the Yazd Branch, Islamic Azad University, and it has been enabled in part with support from Westgrid (www.westgrid.ca) and Compute/Calcul Canada (www.computecanada.ca). The author would like to acknowledge Dr. Alex Brown (University of Alberta) for his helpful discussion and the kindly support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflict of interest in this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ilkhani, A.R. Non-planarity in four-membered homo-cyclic compounds A4 (A = O, S, Se, Te, Po) and restoring their planarity: a study of the pseudo-Jahn–Teller effect. Theor Chem Acc 139, 99 (2020). https://doi.org/10.1007/s00214-020-02615-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-020-02615-x