Abstract
Rationale
The Food and Drug Administration is considering severely restricting the nicotine in cigarettes, to reduce smoking. A study showed that non-daily, intermittent smokers (ITS) randomized to very-low-nicotine-content cigarettes (VLNCCs) reduced their cigarette consumption.
Objectives
To assess whether increased smoking intensity of VLNCCs compensated for some of the reduced cigarette consumption.
Methods
After a 2-week baseline smoking their own-brand cigarettes, 118 ITS were randomized to VLNCCs (~ 1 mg nicotine/g tobacco), and 120 to normal-nicotine-content cigarettes (NNCCs; ~ 16 mg/g) for 10 weeks. Laboratory measures of smoking intensity—total puff volume and carbon monoxide (CO) boost—assessed single cigarettes smoked in up to three laboratory topography sessions. Field measures assessed returned cigarette butts, averaged over up to five 2-week intervals: the mass of tobacco burned (computed from residual mass of butts) and the intensity of smoking (by scanning of returned filters). Analysis was by mixed model random effects models using baseline values as covariates.
Results
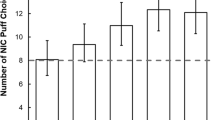
ITS in the VLNCC group puffed less smoke in topography sessions (−38.50 mL [−75.21, −1.78]; p < 0.04), but showed no difference in CO boost. Participants in the VLNCC group burned 0.02 [0.04, 0.002] grams less tobacco per cigarette (p < 0.03). Analysis of filters showed their smoking intensity declined over time, compared to NNCC participants (p < 0.04). “Cheating” by smoking normal cigarettes did not moderate these effects.
Conclusions
ITS did not increase their smoking intensity when switched to VLNCCs; indeed, their smoking intensity decreased. Reductions in cigarette consumption seen when ITS are switched to VLNCCs were not compensated by increased smoking intensity.

Similar content being viewed by others
References
Ashton H, Stepney R, Thompson JW (1979) Self-titration by cigarette smokers. BMJ 2:357–360
Benowitz N (2010) Nicotine addiction. N Engl J Med 362:2295–2203
Benowitz NL, Henningfield JE (1994) Establishing a nicotine threshold for addiction: the implications for tobacco regulation. N Engl J Med 331:123–125
Benowitz NL, Hall SM, Stewart S, Wilson M, Dempsey D, Pr J (2007) Nicotine and carcinogen exposure with smoking of progressively reduced nicotine content cigarette. Cancer Epidemiol Biomark Prev 16:2479–2485
Benowitz NL, Hukkanen J, Pr J (2009) Nicotine chemistry, metabolism, kinetics and biomarkers. Handb Exp Pharmacol 192:29–60
Benowitz NL, Nardone N, Hatsukami DK, Donny EC (2015) Biochemical estimation of noncompliance with smoking of very low nicotine content cigarettes. Cancer Epidemiol Biomark Prev 24:331–335
Cassidy RN, Tidey, J.W., Cao, Q., Colby, S.M., McClernon, F.J., Koopmeiners, J.S., Hatsukami, D., Donny, E.C. (2018) Age moderates smokers’ subjective response to very-low nicotine content cigarettes: evidence from a randomized controlled trial. Nicotine Tob Res Advance online publication
Donny EC, Houtsmuller EJ, Stitzer ML (2007) Smoking in the absence of nicotine: behavioral, subjective and physiological effects over 11 days. Addiction 102:324–334
Donny EC, Denlinger RL, Tidey JW, Koopmeiners JS, Benowitz NL, Vandrey RG, al'Absi M, Carmella SG, Cinciripini PM, Dermody SS, Drobes DJ, Hecht SS, Jensen J, Lane T, Le CT, McClernon FJ, Montoya ID, Murphy SE, Robinson JD, Stitzer ML, Strasser AA, Tindle H, Hatsukami DK (2015) Randomized trial of reduced-nicotine standards for cigarettes. N Engl J Med 373:1340–1349
Foulds J, Hobkirk, A., Wasserman, E., Richie, J., Veldheer, S., Krebs, N.M., Reinhart, L., Muscat, J. (2018) Estimation of compliance with exclusive smoking of very low nicotine content cigarettes using plasma cotinine. Prev Med. Advance online publication
Hasenfratz M, Baldinger B, Battig K (1993) Nicotine or tar titration in cigarette smoking behavior? Psychopharmacology 112:253–258
Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO (1991) The Fagerström test for nicotine dependence: a revision of the Fagerström tolerance questionnaire. Br J Addict 86:1119–1127
Henningfield JE, Benowitz NL, Slade J, Houston TP, Davis RM, Deitchman SD (1998) Reducing the addictiveness of cigarettes. Council on Scientific Affairs, American Medical Association. Tob Control 7: 281–293
Inoue-Choi M, Liao LM, Reyes-Guzman C, Hartge P, Caporaso N, Freedman ND (2017) Association of long-term, low-intensity smoking with all-cause and cause-specific mortality in the National Institutes of Health-AARP Diet and Health Study. JAMA Intern Med 177:87–95
Jamal A, King BA, Neff LJ, Whitmill J, Babb SD, Graffunder CM (2016) Current cigarette smoking among adults - United States, 2005-2015. Mmwr-Morbid Mortal W 65:1205–1211
Kozlowski LT (1981) Applications of some physical indicators of cigarette smoking. Addict Behav 6:213–219
O'Connor RJ, Kozlowski LT, Hammond D, Vance TT, Stitt JP, Cummings KM (2007) Digital image analysis of cigarette filter staining to estimate smoke exposure. Nicotine Tob Res 9:865–871
Polzin G, Xizheng Y, McCraw J, Ashley D, Watson C (2006) Comparison of optical, ultraviolet, and mass spectrometric methods for the determination of cigarette smoking patterns: can lower cost/higher thoughtput methods provide reliable data. Poster presented at The 13th World Conference on Tobacco OR Health, Washington, DC
Reyes-Guzman CM, Pfeiffer RM, Lubin J, Freedman ND, Cleary SD, Levine PH, Caporaso NE (2017) Determinants of light and intermittent smoking in the United States: results from three pooled national health surveys. Cancer Epidemiol Biomark Prev 26:228–239
Schane RE, Ling PM, Glantz SA (2010) Health effects of light and intermittent smoking: a review. Circulation 121:1518–1522
Shiffman S, Scholl SM (2017) Increases in cigarette consumption and decreases in smoking intensity when non-daily smokers are provided with free cigarettes. Nicotine Tob Res: Advance online publication
Shiffman S, Dunbar MS, Scholl SM, Tindle HA (2012a) Smoking motives of daily and non-daily smokers: a profile analysis. Drug Alcohol Depend 126:362–368
Shiffman S, Tindle H, Li X, Scholl S, Dunbar M, Mitchell-Miland C (2012b) Characteristics and smoking patterns of intermittent smokers. Exp Clin Psychopharmacol 20:264–277
Shiffman S, Dunbar MS, Benowitz NL (2014) A comparison of nicotine biomarkers and smoking patterns in daily and nondaily smokers. Cancer Epidemiol Biomark Prev 23:1264–1272
Shiffman S, Dunbar MS, Tindle HA, Ferguson SG (2015) Nondaily smokers' experience of craving on days they do not smoke. J Abnorm Psychol 124:648–659
Shiffman S, Kurland B, Scholl S, Mao J (2018) Non-daily smokers’ changes in cigarette consumption with very-low-nicotine-content cigarettes: a randomized double-blind clinical trial. JAMA Psychiatry. Advance online publication
Sobell LC, Sobell MB, Maisto SA (1985) Time-line follow-back assessment methods. In: Lettieri DJ, Sayers MA, Nelson JE (eds) NIAA treatment handbook series: Vol 2, alcoholism treatment assessment research instruments. National Institute on Alcoholism and Drug Abuse DHHS Publications, Washington, D.C.
Stolerman IP, Jarvis MJ (1995) The scientific case that nicotine is addictive. Psychopharmacology 117:2–10
Strasser AA, O'Connor RJ, Mooney ME, Wileyto EP (2006) Digital image analysis of cigarette filter stains as an indicator of compensatory smoking. Cancer Epidemiol Biomark Prev 15:2565–2569
Substance Abuse and Mental Health Services Administration (2014) Results from the 2013 National Survey on Drug Use and Health: national findings. In: NSDUH Series H-48 HPNS- (ed). Substance Abuse and Mental Health Service Administration, 2014., Rockville, MD
Tindle HA, Shiffman S (2011) Smoking cessation behavior among intermittent smokers versus daily smokers. Am J Public Health 101:e1–e3
Tobacco Product Standard for Nicotine Level of Combusted Cigarettes; HHS Advance notice of proposed rulemaking, 83 Fed. Reg. 11818 (March 16, 2018) (to be codified at 21 C.F.R. pt. 1130)
Acknowledgements
The authors are grateful to Allison Brown for the assistance overseeing the study, to David Colarusso, Corinne Hogge, and Ian Jutsum, research assistants who conducted research sessions, to James Moorehead for the data management and preparation, to Alexsys Hoesch for the administrative assistance, to Dr. Esa Davis for the medical oversight, and to Dr. Clifford Watson for providing the filter-scanning software and training and consultation on the scanning. We also appreciate the contributions of members of the study Data and Safety Monitoring Board, Peter Callas (University of Vermont), Matthew Carpenter (Medical University of South Carolina), Jonathan Foulds (Pennsylvania State University), and John Hughes (University of Vermont).
Funding
This work was supported by a grant (S. Shiffman) from the National Cancer Institute (NCI) at the National Institutes of Health (NIH) and the Center for Tobacco Products (CTP) at the Food and Drug Administration (FDA), awarded as a supplement to grant number P30CA047904. The grant supported use of the UPMC Hillman Cancer Center Biostatistics Shared Resource Facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the University of Pittsburgh Institutional Review Board and participants provided written informed consent.
Conflict of interest
SS, through Pinney Associates, consults on tobacco cessation and harm reduction (including nicotine replacement therapy and digital vapor products; by contract, combusted cigarettes are excluded) to Niconovum USA, RJ Reynolds Vapor Company, and RAI Services Company, all subsidiaries of Reynolds American, Inc. and British American Tobacco. Previously, SS consulted to NJOY on e-cigarettes, and to GlaxoSmithKline Consumer Healthcare on smoking cessation medications and treatments. SS holds patents for a novel nicotine smoking cessation medication that is not under commercial development. Other authors report no competing interests.
Disclaimer
NCI and FDA, had no role in design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or the FDA.
Rights and permissions
About this article
Cite this article
Shiffman, S., Mao, J.M., Kurland, B.F. et al. Do non-daily smokers compensate for reduced cigarette consumption when smoking very-low-nicotine-content cigarettes?. Psychopharmacology 235, 3435–3441 (2018). https://doi.org/10.1007/s00213-018-5056-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-5056-8