Abstract
Objective
A considerable body of research links cognitive function to dopaminergic transmission in the prefrontal cortex, but less is known about cognition in relation to striatal dopamine D2/3 receptors in unmedicated patients with psychosis.
Methods
We investigated this association by obtaining PET recordings with the high-affinity D2/3 antagonist ligand [18F] fallypride in 15 medication-free patients with schizophrenia and 11 healthy controls. On the day of PET scanning, we undertook comprehensive neuropsychological testing and assessment of psychopathology using the Positive and Negative Syndrome Scale (PANSS).
Results
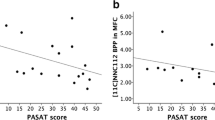
The patients’ performance in cognitive tests was significantly impaired in almost all domains. Irrespective of medication history, the mean [18F] fallypride binding potential (BPND) in the patient group tended to be globally 5–10% higher than that of the control group, but without reaching significance in any brain region. There were significant positive correlations between individual patient performance in the Trail Making Test (TMT(A) and TMT(B)) and Digit-Symbol-Substitution-Test with regional [18F] fallypride BPND, which remained significant after Bonferroni correction for the TMT(A) in caudate nucleus (CN) and for the TMT(B) in CN and putamen. No such correlations were evident in the control group.
Discussion
The association between better cognitive performance and greater BPND in schizophrenia patients may imply that relatively lower receptor occupancy by endogenous dopamine favors better sparing of cognitive function. Absence of comparable correlations in healthy controls could indicate a greater involvement of signaling at dopamine D2/3 receptors in certain cognitive functions in schizophrenia patients than in healthy controls.
Similar content being viewed by others
References
Addington J, Saeedi H, Addington D (2005) The course of cognitive functioning in first episode psychosis: changes over time and impact on outcome. Schizophr Res 78:35–43. https://doi.org/10.1016/j.schres.2005.05.008
Allen MD, Owens TE, Fong AK, Richards DR (2011) A functional neuroimaging analysis of the trail making test-B: implications for clinical application. Behav Neurol 24:159–171. https://doi.org/10.3233/BEN-2011-0278
Arbuthnott K, Frank J (2000) Trail making test, part B as a measure of executive control: validation using a set-switching paradigm. J Clin Exp Neuropsychol 22:518–528. https://doi.org/10.1076/1380-3395(200008)22:4;1-0;FT518
Backman L, Ginovart N, Dixon RA et al (2000) Age-related cognitive deficits mediated by changes in the striatal dopamine system. Am J Psychiatry 157:635–637. https://doi.org/10.1176/ajp.157.4.635
Bäckman L, Nyberg L, Lindenberger U, Li SC, Farde L (2006) The correlative triad among aging, dopamine, and cognition: current status and future prospects. Neurosci Biobehav Rev 30:791–807. https://doi.org/10.1016/j.neubiorev.2006.06.005
Brunelin J, Fecteau S, Suaud-Chagny M-F (2013) Abnormal striatal dopamine transmission in schizophrenia. Curr Med Chem 20:397–404. https://doi.org/10.2174/0929867311320030011
Buchanan RW, Freedman R, Javitt DC, Abi-Dargham A, Lieberman JA (2007) Recent advances in the development of novel pharmacological agents for the treatment of cognitive impairments in schizophrenia. Schizophr Bull 33:1120–1130. https://doi.org/10.1093/schbul/sbm083
Cabeza R, Nyberg L (2000) Imaging cognition II: an empirical review of 275 PET and fMRI studies. J Cogn Neurosci 12:1–47
Carter CS, MacDonald AW, Ross LL, Stenger VA (2001) Anterior cingulate cortex activity and impaired self-monitoring of performance in patients with schizophrenia: an event-related fMRI study. Am J Psychiatry 158:1423–1428. https://doi.org/10.1176/appi.ajp.158.9.1423
Cropley VL, Innis RB, Nathan PJ, Brown AK, Sangare JL, Lerner A, Ryu YH, Sprague KE, Pike VW, Fujita M (2008) Small effect of dopamine release and no effect of dopamine depletion on [18F]fallypride binding in healthy humans. Synapse 62:399–408. https://doi.org/10.1002/syn.20506
Cumming P, Xiong G, la Fougere C et al (2013) Surrogate markers for cerebral blood flow correlate with [(1)(8)F]-fallypride binding potential at dopamine D(2/3) receptors in human striatum. Synapse 67:199–203. https://doi.org/10.1002/syn.21630
Dunn JT, Clark-Papasavas C, Marsden P, Baker S, Cleij M, Kapur S, Kessler R, Howard R, Reeves SJ (2013) Establishing test-retest reliability of an adapted [(18)F]fallypride imaging protocol in older people. J Cereb Blood Flow Metab 33:1098–1103. https://doi.org/10.1038/jcbfm.2013.55
Fagerlund B, Pinborg LH, Mortensen EL, Friberg L, Baaré WFC, Gade A, Svarer C, Glenthøj BY (2013) Relationship of frontal D(2/3) binding potentials to cognition: a study of antipsychotic-naive schizophrenia patients. Int J Neuropsychopharmacol 16:23–36. https://doi.org/10.1017/S146114571200003X
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Floresco SB, Magyar O (2006) Mesocortical dopamine modulation of executive functions: beyond working memory. Psychopharmacology 188:567–585. https://doi.org/10.1007/s00213-006-0404-5
Floresco SB, Zhang Y, Enomoto T (2009) Neural circuits subserving behavioral flexibility and their relevance to schizophrenia. Behav Brain Res 204:396–409. https://doi.org/10.1016/j.bbr.2008.12.001
Glenthoj BY, Mackeprang T, Svarer C, Rasmussen H, Pinborg LH, Friberg L, Baaré W, Hemmingsen R, Videbaek C (2006) Frontal dopamine D(2/3) receptor binding in drug-naive first-episode schizophrenic patients correlates with positive psychotic symptoms and gender. Biol Psychiatry 60:621–629. https://doi.org/10.1016/j.biopsych.2006.01.010
Gold JM, Carpenter C, Randolph C, Goldberg TE, Weinberger DR (1997) Auditory working memory and Wisconsin Card Sorting Test performance in schizophrenia. Arch Gen Psychiatry 54:159–165. https://doi.org/10.1001/archpsyc.1997.01830140071013
Goldman-Rakic PS, Muly EC, Williams GV (2000) D1 receptors in prefrontal cells and circuits. Brain Res Rev 31:295–301. https://doi.org/10.1016/S0165-0173(99)00045-4
Gray JA, Roth BL (2007) Molecular targets for treating cognitive dysfunction in schizophrenia. Schizophr Bull 33:1100–1119
Green MF, Harvey PD (2014) Cognition in schizophrenia: past, present, and future. Schizophr Res Cogn 1:e1–e9. https://doi.org/10.1016/j.scog.2014.02.001
Green MF, Nuechterlein KH, Gold JM, Barch DM, Cohen J, Essock S, Fenton WS, Frese F, Goldberg TE, Heaton RK, Keefe RS, Kern RS, Kraemer H, Stover E, Weinberger DR, Zalcman S, Marder SR (2004) Approaching a consensus cognitive battery for clinical trials in schizophrenia: the NIMH-MATRICS conference to select cognitive domains and test criteria. Biol Psychiatry 56(5):301–307
Gründer G, Fellows C, Janouschek H et al (2008) Brain and plasma pharmacokinetics of aripiprazole in patients with schizophrenia: an [18F]fallypride PET study. Am J Psychiatry 165:988–995. https://doi.org/10.1176/appi.ajp.2008.07101574
Gründer G, Siessmeier T, Piel M, Vernaleken I, Buchholz HG, Zhou Y, Hiemke C, Wong DF, Rösch F, Bartenstein P (2003) Quantification of D2-like dopamine receptors in the human brain with 18F-desmethoxyfallypride. J Nucl Med 44:109–116
Hall H, Farde L, Halldin C et al (1996) Autoradiographic localization of extrastriatal D2-dopamine receptors in the human brain using [125I]epidepride. Synapse 23:115–123. https://doi.org/10.1002/(SICI)1098-2396(199606)23:2<115::AID-SYN7>3.0.CO;2-C
Harth S, Müller SV, Aschenbrenner S et al (2004) Regensburger Wortflüssigkeits-Test (RWT). Z Neuropsychol 15:315–321. https://doi.org/10.1024/1016-264x.15.4.315
Helmich RC, Derikx LC, Bakker M, Scheeringa R, Bloem BR, Toni I (2010) Spatial remapping of cortico-striatal connectivity in Parkinson’s disease. Cereb Cortex 20:1175–1186
Herath P, Kinomura S, Roland PE (2001) Visual recognition: evidence for two distinctive mechanisms from a PET study. Hum Brain Mapp 12:110–119
Heydebrand G, Weiser M, Rabinowitz J, Hoff AL, DeLisi L, Csernansky JG (2004) Correlates of cognitive deficits in first episode schizophrenia. Schizophr Res 68:1–9. https://doi.org/10.1016/S0920-9964(03)00097-5
Hietala J, Syvalahti E, Vuorio K et al (1994) Striatal D2 dopamine receptor characteristics in neuroleptic-naive schizophrenic patients studied with positron emission tomography. Arch Gen Psychiatry 51:116–123
Horacek J, Dockery C, Kopecek M, Spaniel F, Novak T, Tislerova B, Klirova M, Palenicek T, Höschl C (2006) Regional brain metabolism as the predictor of performance on the trail making test in schizophrenia. A 18FDG PET covariation study. Neuro Endocrinol Lett 27:587–594
Howes OD, Kambeitz J, Kim E, Stahl D, Slifstein M, Abi-Dargham A, Kapur S (2012) The nature of dopamine dysfunction in schizophrenia and what this means for treatment. Arch Gen Psychiatry 69:776–786. https://doi.org/10.1001/archgenpsychiatry.2012.169
Ishai A, Ungerleider LG, Martin A, Schouten JL, Haxby JV (1999) Distributed representation of objects in the human ventral visual pathway. Proc Natl Acad Sci U S A 96:9379–9384
Ishibashi K, Robertson CL, Mandelkern MA, et al (2013) The simplified reference tissue model with 18F-fallypride positron emission tomography: choice of reference region
Jacobson SC, Blanchard M, Connolly CC, Cannon M, Garavan H (2011) An fMRI investigation of a novel analogue to the Trail-Making Test. Brain Cogn 77:60–70. https://doi.org/10.1016/j.bandc.2011.06.001
Keefe RSE, Bilder RM, Davis SM, Harvey PD, Palmer BW, Gold JM, Meltzer HY, Green MF, Capuano G, Stroup TS, McEvoy J, Swartz MS, Rosenheck RA, Perkins DO, Davis CE, Hsiao JK, Lieberman JA, CATIE Investigators, Neurocognitive Working Group (2007) Neurocognitive effects of antipsychotic medications in patients with chronic schizophrenia in the CATIE trial. Arch Gen Psychiatry 64:633–647. https://doi.org/10.1001/archpsyc.64.6.633
Keefe RSE, Harvey PD (2012) Cognitive impairment in schizophrenia BT—novel antischizophrenia treatments. In: Geyer MA, Gross G (eds) Springer. Berlin Heidelberg, Berlin, Heidelberg, pp 11–37
Kessler RM, Ansari MS, Riccardi P, Li R, Jayathilake K, Dawant B, Meltzer HY (2005) Occupancy of striatal and extrastriatal dopamine D2/D3 receptors by olanzapine and haloperidol. Neuropsychopharmacology 30:2283–2289. https://doi.org/10.1038/sj.npp.1300836
Kestler LP, Walker E, Vega EM (2001) Dopamine receptors in the brains of schizophrenia patients: a meta-analysis of the findings. Behav Pharmacol 12:355–371
Kortte KB, Horner MD, Windham WK (2002) The trail making test, part B: cognitive flexibility or ability to maintain set? Appl Neuropsychol 9:106–109. https://doi.org/10.1207/S15324826AN0902_5
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. NeuroImage 4:153–158. https://doi.org/10.1006/nimg.1996.0066
Langer O, Halldin C, Dollé F, Swahn CG, Olsson H, Karlsson P, Hall H, Sandell J, Lundkvist C, Vaufrey F, Loc'h C, Crouzel C, Mazière B, Farde L (2017) Carbon-11 epidepride: a suitable radioligand for PET investigation of striatal and extrastriatal dopamine D2 receptors. Nucl Med Biol 26:509–518. https://doi.org/10.1016/S0969-8051(99)00005-0
Laruelle M (1998) Imaging dopamine transmission in schizophrenia. A review and meta-analysis. Q J Nucl Med 42:211–221
Lawrence AD, Weeks RA, Brooks DJ, et al (1998) The relationship between striatal dopamine receptor binding and cognitive performance in Huntington’s disease. Brain : a journal of neurology 121 (Pt 7). England: 1343–1355.
Leeson VC, Barnes TRE, Harrison M, Matheson E, Harrison I, Mutsatsa SH, Ron MA, Joyce EM (2010) The relationship between IQ, memory, executive function, and processing speed in recent-onset psychosis: 1-year stability and clinical outcome. Schizophr Bull 36:400–409. https://doi.org/10.1093/schbul/sbn100
Lindsberg J, Poutiainen E, Kalska H (2009) Clarifying the diversity of first-episode psychosis: neuropsychological correlates of clinical symptoms. Nord J Psychiatry 63:493–500. https://doi.org/10.3109/08039480903118182
MacPherson SE, Cox SR, Dickie DA et al (2017) Processing speed and the relationship between Trail Making Test-B performance, cortical thinning and white matter microstructure in older adults. Cortex 95:92–103. https://doi.org/10.1016/j.cortex.2017.07.021
Mahurin RK, Velligan DI, Hazleton B, Mark Davis J, Eckert S, Miller AL (2006) Trail making test errors and executive function in schizophrenia and depression. Clin Neuropsychol 20:271–288. https://doi.org/10.1080/13854040590947498
Mehta MA, Montgomery AJ, Kitamura Y, et al. (2008) Dopamine D2 receptor occupancy levels of acute sulpiride challenges that produce working memory and learning impairments in healthy volunteers. Psychopharmacology 196(1). Germany: 157–165. https://doi.org/10.1007/s00213-007-0947-0
Meyer EC, Carrion RE, Cornblatt BA, Addington J, Cadenhead KS, Cannon TD, McGlashan TH, Perkins DO, Tsuang MT, Walker EF, Woods SW, Heinssen R, Seidman LJ, the NAPLS group (2014) The relationship of neurocognition and negative symptoms to social and role functioning over time in individuals at clinical high risk in the first phase of the North American Prodrome Longitudinal Study. Schizophr Bull 40:1452–1461. https://doi.org/10.1093/schbul/sbt235
Minzenberg MJ, Laird AR, Thelen S, Carter CS, Glahn DC (2009) Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch Gen Psychiatry 66:811–822. https://doi.org/10.1001/archgenpsychiatry.2009.91
Morris ED, Constantinescu CC, Sullivan JM, Normandin MD, Christopher LA (2010) Noninvasive visualization of human dopamine dynamics from PET images. NeuroImage 51:135–144. https://doi.org/10.1016/j.neuroimage.2009.12.082
Mukherjee J, Christian BT, Dunigan KA, Shi B, Narayanan TK, Satter M, Mantil J (2002) Brain imaging of 18F-fallypride in normal volunteers: blood analysis, distribution, test-retest studies, and preliminary assessment of sensitivity to aging effects on dopamine D-2/D-3 receptors. Synapse 46:170–188. https://doi.org/10.1002/syn.10128
Mukherjee J, Yang ZY, Das MK, Brown T (1995) Fluorinated benzamide neuroleptics--III. Development of (S)-N-[(1-allyl-2-pyrrolidinyl)methyl]-5-(3-[18F]fluoropropyl)-2, 3-dimethoxybenzamide as an improved dopamine D-2 receptor tracer. Nucl Med Biol 22:283–296
Nordstrom AL, Farde L, Eriksson L, Halldin C (1995) No elevated D2 dopamine receptors in neuroleptic-naive schizophrenic patients revealed by positron emission tomography and [11C]N-methylspiperone. Psychiatry Res 61:67–83
Nuechterlein KH, Green MF, Kern RS et al (2008) The MATRICS consensus cognitive battery, part 1: test selection, reliability, and validity. Am J Psychiatry 165:203–213. https://doi.org/10.1176/appi.ajp.2007.07010042
Nyberg L, Karalija N, Salami A, et al. (2016) Dopamine D2 receptor availability is linked to hippocampal-caudate functional connectivity and episodic memory. Proceedings of the National Academy of Sciences of the United States of America 113(28). United States: 7918–7923. https://doi.org/10.1073/pnas.1606309113
Oda Y, Kanahara N, Iyo M (2015) Alterations of dopamine D2 receptors and related receptor-interacting proteins in schizophrenia: the pivotal position of dopamine supersensitivity psychosis in treatment-resistant schizophrenia. Int J Mol Sci 16:30144–30163. https://doi.org/10.3390/ijms161226228
Olsson H, Halldin C, Farde L (2004) Differentiation of extrastriatal dopamine D2 receptor density and affinity in the human brain using PET. NeuroImage 22:794–803. https://doi.org/10.1016/j.neuroimage.2004.02.002
Pavese N, Andrews TC, Brooks DJ, et al. (2003) Progressive striatal and cortical dopamine receptor dysfunction in Huntington’s disease: a PET study. Brain : a journal of neurology 126(Pt 5). England: 1127–1135.
Rajji TK, Mulsant BH, Nakajima S, Caravaggio F, Suzuki T, Uchida H, Gerretsen P, Mar W, Pollock BG, Mamo DC, Graff-Guerrero A (2017) Cognition and dopamine D2 receptor availability in the striatum in older patients with schizophrenia. Am J Geriatr Psychiatry 25:1–10. https://doi.org/10.1016/j.jagp.2016.08.001
Reeves SJ, Grasby PM, Howard RJ, Bantick RA, Asselin MC, Mehta MA (2005) A positron emission tomography (PET) investigation of the role of striatal dopamine (D2) receptor availability in spatial cognition. NeuroImage 28:216–226. https://doi.org/10.1016/j.neuroimage.2005.05.034
Reitan RM (1958) Validity of the trail making test as an indicator of organic brain damage. Percept Mot Skills 8:271–276. https://doi.org/10.2466/PMS.8.7.271-276
Robinson JL, Laird AR, Glahn DC, Blangero J, Sanghera MK, Pessoa L, Fox PM, Uecker A, Friehs G, Young KA, Griffin JL, Lovallo WR, Fox PT (2012) The functional connectivity of the human caudate: an application of meta-analytic connectivity modeling with behavioral filtering. NeuroImage 60:117–129. https://doi.org/10.1016/j.neuroimage.2011.12.010
Seamans JK, Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74:1–58. https://doi.org/10.1016/j.pneurobio.2004.05.006
Seeman P (2013) Are dopamine D2 receptors out of control in psychosis? Prog Neuro-Psychopharmacol Biol Psychiatry 46:146–152. https://doi.org/10.1016/j.pnpbp.2013.07.006
Siessmeier T, Zhou Y, Buchholz H-G, Landvogt C, Vernaleken I, Piel M, Schirrmacher R, Rösch F, Schreckenberger M, Wong DF, Cumming P, Gründer G, Bartenstein P (2005) Parametric mapping of binding in human brain of D2 receptor ligands of different affinities. J Nucl Med 46:964–972
Simpson EH, Kellendonk C, Kandel E (2010) A possible role for the striatum in the pathogenesis of the cognitive symptoms of schizophrenia. Neuron 65:585–596. https://doi.org/10.1016/j.neuron.2010.02.014
Suridjan I, Rusjan P, Addington J, Wilson A, Houle S, Mizrahi R (2013) Dopamine D2 and D3 binding in people at clinical high risk for schizophrenia, antipsychotic-naive patients and healthy controls while performing a cognitive task. J Psychiatry Neurosci 38:98–106. https://doi.org/10.1503/jpn.110181
Takahashi H (2013) PET neuroimaging of extrastriatal dopamine receptors and prefrontal cortex functions. J Physiol 107:503–509. https://doi.org/10.1016/j.jphysparis.2013.07.001
Tamminga CA (2006) The neurobiology of cognition in schizophrenia. J Clin Psychiatry 67(Suppl 9):9–42
Tune LE, Wong DF, Pearlson G, Strauss M, Young T, Shaya EK, Dannals RF, Wilson AA, Ravert HT, Sapp J, Cooper T, Chase GA, Wagner HN Jr (1993) Dopamine D2 receptor density estimates in schizophrenia: a positron emission tomography study with 11C-N-methylspiperone. Psychiatry Res 49:219–237
Ventura J, Hellemann GS, Thames AD, Koellner V, Nuechterlein KH (2009) Symptoms as mediators of the relationship between neurocognition and functional outcome in schizophrenia: a meta-analysis. Schizophr Res 113:189–199. https://doi.org/10.1016/j.schres.2009.03.035
Ventura J, Thames AD, Wood RC, Guzik LH, Hellemann GS (2010) Disorganization and reality distortion in schizophrenia: a meta-analysis of the relationship between positive symptoms and neurocognitive deficits. Schizophr Res 121:1–14. https://doi.org/10.1016/j.schres.2010.05.033
Vernaleken I, Janouschek H, Raptis M, Hellmann S, Veselinovic T, Bröcheler A, Boy C, Cumming P, Hiemke C, Rösch F, Schäfer WM, Gründer G (2010) Dopamine D2/3 receptor occupancy by quetiapine in striatal and extrastriatal areas. Int J Neuropsychopharmacol 13:951–960
Vernaleken I, Klomp M, Moeller O, Raptis M, Nagels A, Rösch F, Schaefer WM, Cumming P, Gründer G (2013) Vulnerability to psychotogenic effects of ketamine is associated with elevated D2/3-receptor availability. Int J Neuropsychopharmacol 16:745–754. https://doi.org/10.1017/S1461145712000764
Vernaleken I, Peters L, Raptis M, Lin R, Buchholz HG, Zhou Y, Winz O, Rösch F, Bartenstein P, Wong DF, Scháfer WM, Gründer G (2011) The applicability of SRTM in [(18)F]fallypride PET investigations: impact of scan durations. J Cereb Blood Flow Metab 31:1958–1966. https://doi.org/10.1038/jcbfm.2011.73
Veselinović T, Vernaleken I, Janouschek H, Kellermann T, Paulzen M, Cumming P, Gründer G (2015) Effects of anticholinergic challenge on psychopathology and cognition in drug-free patients with schizophrenia and healthy volunteers. Psychopharmacology 232:1607–1617. https://doi.org/10.1007/s00213-014-3794-9
Volkow ND, Gur RC, Wang G-J, Fowler JS, Moberg PJ, Ding YS, Hitzemann R, Smith G, Logan J (1998) Association between declines in brain dopamine activity with age and cognitive and motor impairment in healthy individuals. Am J Psychiatry 155:344–349. https://doi.org/10.1176/ajp.155.3.344
Wechsler D (1997) WAIS-III administration and scoring manual
Weinberger DR, Laruelle M (2002) Neuropharmacological imaging in schizophrenia. In: Davis KL, Charney D, Coyle JTCN (eds) Neuropsychopharmacology: the fifth generation of progress. Lippincott, Williams, & Wilkins. Pennsylvania, Philadelphia
Wong DF, Wagner HNJ, Tune LE et al (1986) Positron emission tomography reveals elevated D2 dopamine receptors in drug-naive schizophrenics. Science 234:1558–1563
Yang YK, Yeh TL, Chiu NT, Lee IH, Chen PS, Lee LC, Jeffries KJ (2004a) Association between cognitive performance and striatal dopamine binding is higher in timing and motor tasks in patients with schizophrenia. Psychiatry Res 131:209–216. https://doi.org/10.1016/j.pscychresns.2003.07.002
Yang YK, Yu L, Yeh TL, Chiu NT, Chen PS, Lee IH, SPECT study (2004b) Associated alterations of striatal dopamine D2/D3 receptor and transporter binding in drug-naive patients with schizophrenia: a dual-isotope SPECT study. Am J Psychiatry 161:1496–1498. https://doi.org/10.1176/appi.ajp.161.8.1496
Yoder KK, Hutchins GD, Morris ED, Brashear A, Wang C, Shekhar A (2004) Dopamine transporter density in schizophrenic subjects with and without tardive dyskinesia. Schizophr Res 71:371–375. https://doi.org/10.1016/j.schres.2004.03.015
Funding
This study was supported by the German Research Association (Deutsche Forschungsgemeinschaft, DFG; KFO-112/2-1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Vernaleken has served on the speakers’ bureau of Bristol-Myers Squibb (New York, NY), Eli Lilly (Indianapolis, Ind), and GlaxoSmithKline (London, UK). Dr. Gründer has served as a consultant for Allergan (Dublin, Ireland), Boehringer Ingelheim (Ingelheim, Germany), Eli Lilly (Indianapolis, Ind, USA), Janssen-Cilag (Neuss, Germany), Lundbeck (Copenhagen, Denmark), Ono Pharmaceuticals (Osaka, Japan), Otsuka (Chiyoda, Japan), Recordati (Milan, Italy), Roche (Basel, Switzerland), Servier (Paris, France), and Takeda (Osaka, Japan). He has served on the speakers’ bureau of Eli Lilly, Janssen Cilag, Neuraxpharm (Langenfeld, Germany), Roche, Servier, and Trommsdorf (Aachen, Germany). He has received grant support from Boehringer Ingelheim and Roche. He is co-founder of Mind and Brain Institute GmbH (Zornheim, Germany) and Brainfoods GmbH (Zornheim, Germany). Dr. Veselinović, Dr. Janouschek, Prof. Cumming, Dr. Paulzen, and Dr. Mottaghy declare no conflicts of interest.
Electronic supplementary material
Figure 1
Scatter plots showing the distributions of the D2/3 receptor availability ([18F]fallypride BPND) in the patients and the controls. Depicted are values for two striatal regions (caudate nucleus, putamen) and two extrastriatal regions (inferior temporal gyrus, thalamus). (PDF 49 kb)
Rights and permissions
About this article
Cite this article
Veselinović, T., Vernaleken, I., Janouschek, H. et al. The role of striatal dopamine D2/3 receptors in cognitive performance in drug-free patients with schizophrenia. Psychopharmacology 235, 2221–2232 (2018). https://doi.org/10.1007/s00213-018-4916-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4916-6