Abstract
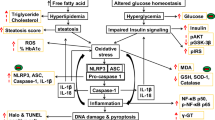
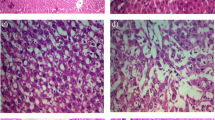
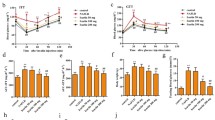
Glibenclamide (GLB), most widely used in the treatment of type II diabetes mellitus, inhibits K+ATP channel in pancreatic-β cells and releases insulin, while thioacetamide (TAA) is a well-known hepatotoxicant and most recommended for the induction of acute and chronic liver disease. The purpose of this study was to evaluate the hepatoprotective potential of GLB against TAA-induced hepatic damage in Wistar rats. TAA (200 mg/kg, ip, twice weekly) and GLB (1.25, 2.5, and 5 mg/kg/day, po) were administered for 6 consecutive weeks. Different biochemical, DNA damage, histopathological, TEM, immunohistochemical, and western blotting parameters were evaluated. GLB treatment has no effects on the TAA-induced significant decrease in body and liver weights. TAA treatment significantly increased liver index and treatment with GLB has no effect the same. TAA treatment altered the liver morphology, whereas treatment with GLB normalized the alteration in morphology. Further, significant increase in oxidative stress, apoptosis, and DNA damage was found in TAA-treated animals and GLB treatment significantly reduced these effects. TAA-induced plasma transaminases and serum ALP levels were significantly restored by GLB. Furthermore, histopathological findings showed the presence of lymphocyte infiltration, collagen deposition, bridging fibrosis, degeneration of portal triad, and necrosis in TAA-treated animals and GLB intervention significantly reduced the same. TEM images revealed that GLB significantly normalized the hepatic stellate cell morphology as well as restored the number of lipid droplets. GLB treatment significantly downregulated the expressions of TGF-β1, α-SMA, NLRP3, ASC, caspase-1, and IL-1β, and upregulated MMP-2 and catalase against TAA-induced liver damage. The outcomes of the present study confirmed that GLB ameliorated the liver damage induced by TAA.











Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine transaminase
- ASC:
-
Apoptosis-associated speck-like protein containing a caspase-recruiting domain
- AST:
-
Aspartate transaminase
- ATP:
-
Adenosine triphosphate
- CYP2E1:
-
Cytochrome P450 2E1
- DAB:
-
3,3′-Diaminobenzidine
- DMSO:
-
Dimethyl sulfoxide
- ECM:
-
Extracellular matrix
- GLB:
-
Glibenclamide
- HSC:
-
Hepatic stellate cell
- IL:
-
Interleukin
- IL-1β:
-
Interleukin-1 beta
- ip:
-
Intraperitoneal
- MT:
-
Masson’s trichrome
- MMP-2:
-
Matrix metalloproteinase-2
- NLRP3:
-
NACHT, LRR, and PYD domains-containing protein 3
- TMO:
-
Tail moment olive
- PAS:
-
Periodic acid-Schiff base
- po:
-
Per os (oral administration)
- PSR:
-
Picro Sirius Red
- PSRF:
-
PSR with fast green FCF
- SOD:
-
Superoxide dismutase
- T2DM:
-
Type II diabetes mellitus
- TAA:
-
Thioacetamide
- TEM:
-
Transmission electron microscopy
- TGF-β1:
-
Transforming growth factor beta1
- TL:
-
Tail length
- TM:
-
Tail moment
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
- US-FDA:
-
United States Food and Drug Administration
- WHO:
-
World Health Organization
- α-SMA:
-
Alpha-smooth muscle actin
- γ-GT:
-
γ-Glutamyl transferase
References
Aguillar-Gomes L, Lopes C, Barbieri D, Rocha T, Randazzo-Moura P (2014) Toxic effects of glibenclamide in fetuses of normoglycemic rats: an alternative therapy for gestational diabetes mellitus. Open Vet J 4:59–64
Al-Attar AM (2012) Attenuating effect of ginkgo biloba leaves extract on liver fibrosis induced by thioacetamide in mice. J Biomed Biotechnol 2012:9–9. https://doi.org/10.1155/2012/761450
Ansar MM, Ansari M (2006) Nitric oxide involvement in pancreatic beta cell apoptosis by glibenclamide. Nitric oxide : biology and chemistry 14:39–44. https://doi.org/10.1016/j.niox.2005.09.002
Bataller R, Brenner DA (2005) Liver fibrosis. J Clin Invest 115:209–218. https://doi.org/10.1172/jci200524282
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Beltrand J, Elie C, Busiah K, Fournier E, Boddaert N, Bahi-Buisson N, Vera M, Bui-Quoc E, Ingster-Moati I, Berdugo M, Simon A, Gozalo C, Djerada Z, Flechtner I, Treluyer JM, Scharfmann R, Cave H, Vaivre-Douret L, Polak M (2016) Erratum. Sulfonylurea therapy benefits neurological and psychomotor functions in patients with neonatal diabetes owing to potassium channel mutations. Diabetes Care 2015;38:2033-2041. Diabetes Care 39: 175 https://doi.org/10.2337/dc16-er01
Cai J, Lu S, Yao Z, Deng YP, Zhang LD, Yu JW, Ren GF, Shen FM, Jiang GJ (2014) Glibenclamide attenuates myocardial injury by lipopolysaccharides in streptozotocin-induced diabetic mice. Cardiovasc Diabetol 13:106. https://doi.org/10.1186/s12933-014-0106-y
Chukwunonso Obi B, Chinwuba Okoye T, Okpashi VE, Nonye Igwe C, Olisah Alumanah E (2016) Comparative study of the antioxidant effects of metformin, glibenclamide, and repaglinide in alloxan-induced diabetic rats. J Diabetes Res 2016. doi: https://doi.org/10.1155/2016/1635361, 1, 5
Czaja AJ (2014) Hepatic inflammation and progressive liver fibrosis in chronic liver disease. World J Gastroenterol: WJG 20:2515–2532. https://doi.org/10.3748/wjg.v20.i10.2515
Diwan V, Gobe G, Brown L (2014) Glibenclamide improves kidney and heart structure and function in the adenine-diet model of chronic kidney disease. Pharmacol Res 79:104–110. https://doi.org/10.1016/j.phrs.2013.11.007
Dixon LJ, Barnes M, Tang H, Pritchard MT, Nagy LE (2013) Kupffer cells in the liver. Compr Physiol 3:785–797. https://doi.org/10.1002/cphy.c120026
El-Mihi KA, Kenawy HI, El-Karef A, Elsherbiny NM, Eissa LA (2017) Naringin attenuates thioacetamide-induced liver fibrosis in rats through modulation of the PI3K/Akt pathway. Life Sci 187:50–57. https://doi.org/10.1016/j.lfs.2017.08.019
Friedman SL (2004) Mechanisms of disease: mechanisms of hepatic fibrosis and therapeutic implications. Nat Clin Pract Gastroenterol Hepatol 1:98–105. https://doi.org/10.1038/ncpgasthep0055
Gad SC, Cassidy CD, Aubert N, Spainhour B, Robbe H (2006) Nonclinical vehicle use in studies by multiple routes in multiple species. Int J Toxicol 25:499–521. https://doi.org/10.1080/10915810600961531
Hughes FM Jr, Vivar NP, Kennis JG, Pratt-Thomas JD, Lowe DW, Shaner BE, Nietert PJ, Spruill LS, Purves JT (2014) Inflammasomes are important mediators of cyclophosphamide-induced bladder inflammation. American journal of physiology Renal physiology 306:F299–F308. https://doi.org/10.1152/ajprenal.00297.2013
Kewcharoenwong C, Rinchai D, Utispan K, Suwannasaen D, Bancroft GJ, Ato M, Lertmemongkolchai G (2013) Glibenclamide reduces pro-inflammatory cytokine production by neutrophils of diabetes patients in response to bacterial infection. Sci Rep 3:3363. https://doi.org/10.1038/srep03363
Khan S, Jena G (2014a) Sodium butyrate, a HDAC inhibitor ameliorates eNOS, iNOS and TGF-beta1-induced fibrogenesis, apoptosis and DNA damage in the kidney of juvenile diabetic rats. Food Chem Toxicol 73:127–139. https://doi.org/10.1016/j.fct.2014.08.010
Khan S, Jena G (2014b) Sodium valproate, a histone deacetylase inhibitor ameliorates cyclophosphamide-induced genotoxicity and cytotoxicity in the colon of mice. J Basic Clin Physiol Pharmacol: 1-11 https://doi.org/10.1515/jbcpp-2013-0134
Kim K-A, Park J-Y (2003) Inhibitory effect of glyburide on human cytochrome p450 isoforms in human liver microsomes. Drug Metab Dispos 31:1090–1092. https://doi.org/10.1124/dmd.31.9.1090
Kim NH, Lee S, Kang MJ, Jeong HG, Kang W, Jeong TC (2014) Protective effects of diallyl sulfide against thioacetamide-induced toxicity: a possible role of cytochrome P450 2E1. Biomol Ther 22:149–154. https://doi.org/10.4062/biomolther.2014.016
Kimberly WT, Battey TW, Pham L, Wu O, Yoo AJ, Furie KL, Singhal AB, Elm JJ, Stern BJ, Sheth KN (2014) Glyburide is associated with attenuated vasogenic edema in stroke patients. Neurocrit Care 20:193–201. https://doi.org/10.1007/s12028-013-9917-z
Kmiec Z (2001) Cooperation of liver cells in health and disease. Advances in anatomy, embryology, and cell biology 161: iii-xiii, 1-151
Koblihova E, Mrazova I, Vernerova Z, Ryska M (2014) Acute liver failure induced by thioacetamide: selection of optimal dosage in Wistar and Lewis rats. Physiol Res 63:491–503
Koh GC, Weehuizen TA, Breitbach K, Krause K, de Jong HK, Kager LM, Hoogendijk AJ, Bast A, Peacock SJ, van der Poll T, Steinmetz I, Wiersinga WJ (2013) Glyburide reduces bacterial dissemination in a mouse model of melioidosis. PLoS Negl Trop Dis 7:e2500. https://doi.org/10.1371/journal.pntd.0002500
Kwon MJ, Chung HS, Yoon CS, Lee EJ, Kim TK, Lee SH, Ko KS, Rhee BD, Kim MK, Park JH (2013) Low glibenclamide concentrations affect endoplasmic reticulum stress in INS-1 cells under glucotoxic or glucolipotoxic conditions. The Korean journal of internal medicine 28:339–346. https://doi.org/10.3904/kjim.2013.28.3.339
Lamkanfi M, Mueller JL, Vitari AC, Misaghi S, Fedorova A, Deshayes K, Lee WP, Hoffman HM, Dixit VM (2009) Glyburide inhibits the cryopyrin/Nalp3 inflammasome. J Cell Biol 187:61–70. https://doi.org/10.1083/jcb.200903124
Ledda-Columbano GM, Coni P, Curto M, Giacomini L, Faa G, Oliverio S, Piacentini M, Columbano A (1991) Induction of two different modes of cell death, apoptosis and necrosis, in rat liver after a single dose of thioacetamide. Am J Pathol 139:1099–1109
Lee SW, Kim SH, Min SO, Kim KS (2011) Ideal experimental rat models for liver diseases. Korean J Hepatobiliary Pancreat Surg 15:67–77. https://doi.org/10.14701/kjhbps.2011.15.2.67
Lee TF, Mak KM, Rackovsky O, Lin YL, Kwong AJ, Loke JC, Friedman SL (2010) Downregulation of hepatic stellate cell activation by retinol and palmitate mediated by adipose differentiation-related protein (ADRP). J Cell Physiol 223:648–657. https://doi.org/10.1002/jcp.22063
Li S, Tan HY, Wang N, Zhang ZJ, Lao L, Wong CW, Feng Y (2015) The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 16:26087–26124. https://doi.org/10.3390/ijms161125942
Liu Y, Meyer C, Xu C, Weng H, Hellerbrand C, ten Dijke P, Dooley S (2013) Animal models of chronic liver diseases. Am J Physiol Gastrointest Liver Physiol 304:G449–G468. https://doi.org/10.1152/ajpgi.00199.2012
Ludwig-Portugall I, Bartok E, Dhana E, Evers BD, Primiano MJ, Hall JP, Franklin BS, Knolle PA, Hornung V, Hartmann G, Boor P, Latz E, Kurts C (2016) An NLRP3-specific inflammasome inhibitor attenuates crystal-induced kidney fibrosis in mice. Kidney Int 90:525–539. https://doi.org/10.1016/j.kint.2016.03.035
Mann DA, Smart DE (2002) Transcriptional regulation of hepatic stellate cell activation. Gut 50:891–896
Maremanda KP, Khan S, Jena G (2014) Zinc protects cyclophosphamide-induced testicular damage in rat: involvement of metallothionein, tesmin and Nrf2. Biochem Biophys Res Commun 445:591–596. https://doi.org/10.1016/j.bbrc.2014.02.055
Maremanda KP, Khan S, Jena GB (2016) Role of zinc supplementation in testicular and epididymal damages in diabetic rat: involvement of Nrf2, SOD1, and GPX5. Biol Trace Elem Res 173:452–464. https://doi.org/10.1007/s12011-016-0674-7
Marumo T, Hishikawa K, Yoshikawa M, Hirahashi J, Kawachi S, Fujita T (2010) Histone deacetylase modulates the proinflammatory and -fibrotic changes in tubulointerstitial injury. Am J Physiol Renal Physiol 298:F133–F141. https://doi.org/10.1152/ajprenal.00400.2009
Masarone M, Rosato V, Aglitti A, Bucci T, Caruso R, Salvatore T, Sasso FC, Tripodi MF, Persico M (2017) Liver biopsy in type 2 diabetes mellitus: steatohepatitis represents the sole feature of liver damage. PLoS One 12:e0178473. https://doi.org/10.1371/journal.pone.0178473
Meng N, Xia M, Lu YQ, Wang M, Boini KM, Li PL, Tang WX (2016) Activation of NLRP3 inflammasomes in mouse hepatic stellate cells during Schistosoma. J Infection Oncotarget 7:39316–39331. https://doi.org/10.18632/oncotarget.10044
Mirzamoradi M, Heidar Z, Faalpoor Z, Naeiji Z, Jamali R (2015) Comparison of glyburide and insulin in women with gestational diabetes mellitus and associated perinatal outcome: a randomized clinical trial. Acta Med Iran 53:97–103
Mokdad AA, Lopez AD, Shahraz S, Lozano R, Mokdad AH, Stanaway J, Murray CJL, Naghavi M (2014) Liver cirrhosis mortality in 187 countries between 1980 and 2010: a systematic analysis. BMC Med 12:145. https://doi.org/10.1186/s12916-014-0145-y
Moreira RK (2007) Hepatic stellate cells and liver fibrosis. Arch Pathol Lab Med 131:1728–1734. https://doi.org/10.1043/1543-2165(2007)131[1728:hscalf]2.0.co;2
Mridha AR, Wree A, Robertson AAB, Yeh MM, Johnson CD, Van Rooyen DM, Haczeyni F, Teoh NC, Savard C, Ioannou GN, Masters SL, Schroder K, Cooper MA, Feldstein AE, Farrell GC (2017) NLRP3 inflammasome blockade reduces liver inflammation and fibrosis in experimental NASH in mice. J Hepatol 66:1037–1046. https://doi.org/10.1016/j.jhep.2017.01.022
Mulder H, Schopman W Sr, van der Lely AJ (1991) Extrapancreatic insulin effect of glibenclamide. Eur J Clin Pharmacol 40:379–381
Ouyang X, Ghani A, Mehal WZ (2013) Inflammasome biology in fibrogenesis. Biochim Biophys Acta 1832:979–988. https://doi.org/10.1016/j.bbadis.2013.03.020
Puche JE, Saiman Y, Friedman SL (2013) Hepatic stellate cells and liver fibrosis. Comprehensive Physiology 3:1473–1492. https://doi.org/10.1002/cphy.c120035
Rowe IA (2017) Lessons from epidemiology: the burden of liver disease. Dig Dis 35:304–309. https://doi.org/10.1159/000456580
Sawada F, Inoguchi T, Tsubouchi H, Sasaki S, Fujii M, Maeda Y, Morinaga H, Nomura M, Kobayashi K, Takayanagi R (2008) Differential effect of sulfonylureas on production of reactive oxygen species and apoptosis in cultured pancreatic beta-cell line, MIN6. Metab Clin Exp 57:1038–1045. https://doi.org/10.1016/j.metabol.2008.01.038
Schmid D, Svoboda M, Sorgner A, Moravcevic I, Thalhammer T, Chiba P, Moslinger T (2011) Glibenclamide reduces proinflammatory cytokines in an ex vivo model of human endotoxinaemia under hypoxaemic conditions. Life Sci 89:725–734. https://doi.org/10.1016/j.lfs.2011.08.017
Sheth KN, Elm JJ, Molyneaux BJ, Hinson H, Beslow LA, Sze GK, Ostwaldt AC, Del Zoppo GJ, Simard JM, Jacobson S, Kimberly WT (2016) Safety and efficacy of intravenous glyburide on brain swelling after large hemispheric infarction (GAMES-RP): a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Neurol 15:1160–1169. https://doi.org/10.1016/s1474-4422(16)30196-x
Sheth KN, Kimberly WT, Elm JJ, Kent TA, Yoo AJ, Thomalla G, Campbell B, Donnan GA, Davis SM, Albers GW, Jacobson S, del Zoppo G, Simard JM, Stern BJ, Mandava P (2014) Exploratory analysis of glyburide as a novel therapy for preventing brain swelling. Neurocrit Care 21:43–51. https://doi.org/10.1007/s12028-014-9970-2
Simard JM, Geng Z, Woo SK, Ivanova S, Tosun C, Melnichenko L, Gerzanich V (2009a) Glibenclamide reduces inflammation, vasogenic edema, and caspase-3 activation after subarachnoid hemorrhage. J Cereb Blood Flow Metab 29:317–330. https://doi.org/10.1038/jcbfm.2008.120
Simard JM, Yurovsky V, Tsymbalyuk N, Melnichenko L, Ivanova S, Gerzanich V (2009b) Protective effect of delayed treatment with low-dose glibenclamide in three models of ischemic stroke. Stroke 40:604–609. https://doi.org/10.1161/strokeaha.108.522409
Socci DJ, Bjugstad KB, Jones HC, Pattisapu JV, Arendash GW (1999) Evidence that oxidative stress is associated with the pathophysiology of inherited hydrocephalus in the H-Tx rat model. Exp Neurol 155:109–117. https://doi.org/10.1006/exnr.1998.6969
Sokolovska J, Isajevs S, Sugoka O, Sharipova J, Paramonova N, Isajeva D, Rostoka E, Sjakste T, Kalvinsh I, Sjakste N (2012) Comparison of the effects of glibenclamide on metabolic parameters, GLUT1 expression, and liver injury in rats with severe and mild streptozotocin-induced diabetes mellitus. Medicina (Kaunas, Lithuania) 48: 532-543
Soliman HM, Selim AO (2012) Role of hepatic stellate cells in fibrogenesis in a model of pomegranate-treated fatty liver induced by junk food in male albino rats immunohistochemical and electron microscopic study. Egyptian Journal of Histology 35:54–66. https://doi.org/10.1097/01.EHX.0000410947.56955.e0
Song R, Chen L, Chen Y, Si X, Liu Y, Liu Y, Irwin DM (2017) Comparison of glyburide and insulin in the management of gestational diabetes: a meta-analysis 12: e0182488 https://doi.org/10.1371/journal.pone.0182488
Spirli C, Fiorotto R, Song L, Santos-Sacchi J, Okolicsanyi L, Masier S, Rocchi L, Vairetti MP, De Bernard M, Melero S, Pozzan T, Strazzabosco M (2005) Glibenclamide stimulates fluid secretion in rodent cholangiocytes through a cystic fibrosis transmembrane conductance regulator-independent mechanism. Gastroenterology 129:220–233. https://doi.org/10.1053/j.gastro.2005.03.048
Szabo G, Petrasek J (2015) Inflammasome activation and function in liver disease. Nat Rev Gastroenterol Hepatol 12:387–400. https://doi.org/10.1038/nrgastro.2015.94
Tsai MK, Lin YL, Huang YT (2010) Effects of salvianolic acids on oxidative stress and hepatic fibrosis in rats. Toxicol Appl Pharmacol 242:155–164. https://doi.org/10.1016/j.taap.2009.10.002
Uno Y, Kojima H, Omori T, Corvi R, Honma M, Schechtman LM, Tice RR, Beevers C, De Boeck M, Burlinson B, Hobbs CA, Kitamoto S, Kraynak AR, McNamee J, Nakagawa Y, Pant K, Plappert-Helbig U, Priestley C, Takasawa H, Wada K, Wirnitzer U, Asano N, Escobar PA, Lovell D, Morita T, Nakajima M, Ohno Y, Hayashi M (2015) JaCVAM-organized international validation study of the in vivo rodent alkaline comet assay for detection of genotoxic carcinogens: II. Summary of definitive validation study results. Mutat Res Genet Toxicol Environ Mutagen 786-788:45–76. https://doi.org/10.1016/j.mrgentox.2015.04.010
Van Doren SR (2015) Matrix metalloproteinase interactions with collagen and elastin. Matrix Biol 0: 224–231 https://doi.org/10.1016/j.matbio.2015.01.005, 44-46
Wan X, Xu C, Yu C, Li Y (2016) Role of NLRP3 inflammasome in the progression of NAFLD to NASH. Can J Gastroenterol Hepatol 2016, doi: https://doi.org/10.1155/2016/6489012, 1, 7
Watanabe A, Sohail MA, Gomes DA, Hashmi A, Nagata J, Sutterwala FS, Mahmood S, Jhandier MN, Shi Y, Flavell RA, Mehal WZ (2009) Inflammasome-mediated regulation of hepatic stellate cells. Am J Physiol Gastrointest Liver Physiol 296:G1248–G1257. https://doi.org/10.1152/ajpgi.90223.2008
WHO (2015) The top 10 cause of death in lower-middle-income economies 2015. WHO & world bank available online on http://wwwwhoint/mediacentre/factsheets/fs310/en/index1html last accessed on september 9, 2017
Wree A, Eguchi A, McGeough MD, Pena CA, Johnson CD, Canbay A, Hoffman HM, Feldstein AE (2014) NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology (Baltimore, Md) 59:898–910. https://doi.org/10.1002/hep.26592
Wynn TA (2008) Cellular and molecular mechanisms of fibrosis. J Pathol 214:199–210. https://doi.org/10.1002/path.2277
York JM, Castellanos KJ, Cabay RJ, Fantuzzi G (2014) Inhibition of the NLRP3 inflammasome reduces the severity of experimentally-induced acute pancreatitis in obese mice. Transl Res 164:259–269. https://doi.org/10.1016/j.trsl.2014.06.012
Zhang G, Lin X, Zhang S, Xiu H, Pan C, Cui W (2017a) A protective role of glibenclamide in inflammation-associated injury. 2017: 3578702 https://doi.org/10.1155/2017/3578702
Zhang Z, Zhao S, Yao Z, Wang L, Shao J, Chen A, Zhang F, Zheng S (2017b) Autophagy regulates turnover of lipid droplets via ROS-dependent Rab25 activation in hepatic stellate cell. Redox Biol 11:322–334. https://doi.org/10.1016/j.redox.2016.12.021
Zhu L, Cortes P, Hassett C, Taube DW, Yee J (2012) Glibenclamide induces collagen IV catabolism in high glucose-stimulated mesangial cells. Exp Diabetes Res 2012:183535–183539. https://doi.org/10.1155/2012/183535
Acknowledgements
We wish to acknowledge the financial assistance received from National Institute of Pharmaceutical Education and Research (NIPER), S.A.S. Nagar, India, for carrying out the above experimentation. The authors would like to acknowledge Mr. Vinod Kumar for TEM image analysis. We are also grateful to Dr. Sabbir Khan, Dr. Krishna Prahlad Maremanda, and Dr. Venkateswara Rao Amara for their timely advice and technical support.
Author information
Authors and Affiliations
Contributions
DKD conceived, conducted the experiments, analyzed the data, and wrote the manuscript. GBJ conceived the idea, reviewed the manuscript, and administered the project. Finally, both the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
The experimental protocol was approved by the Institutional Animal Ethics Committee (IAEC) against approval number IAEC/16/54.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 17354 kb)
Rights and permissions
About this article
Cite this article
Dwivedi, D.K., Jena, G.B. Glibenclamide protects against thioacetamide-induced hepatic damage in Wistar rat: investigation on NLRP3, MMP-2, and stellate cell activation. Naunyn-Schmiedeberg's Arch Pharmacol 391, 1257–1274 (2018). https://doi.org/10.1007/s00210-018-1540-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1540-2