Abstract
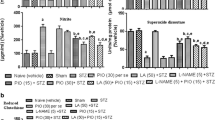
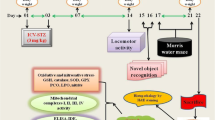
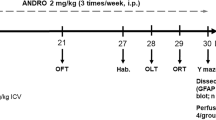
Ellagic acid (EGA)-enriched dietary supplements are widely acclaimed, owing to its versatile bioactivities. Previously, we reported that chronic administration of EGA prevented the impairment of cognitive abilities in rats using the intracerebroventricular-administered streptozotocin (STZ-ICV) model of Alzheimer’s disease. Impairment of phosphoinositide 3 (PI3)-kinase-regulated endothelial nitric oxide synthase (eNOS) activity by central administration of STZ in rodents instigates dementia. The aim of the present study was to delineate the role of PI3-kinase-eNOS activity in the prevention of STZ-ICV-induced memory dysfunctions by EGA. The Morris water maze and elevated plus maze tests were conducted, and brain oxidative stress markers (TBARS, GSH, SOD, CAT), nitrite, acetylcholinesterase (AChE), LDH, TNF-α and eNOS were quantified. Administration of EGA (35 mg/k, p.o.) for 4 weeks daily attenuated the STZ-ICV (3 mg/kg)-triggered increase of brain oxidative stress, nitrite and TNF-α levels; AChE and LDH activity; and decline of brain eNOS activity. The memory restoration by EGA in STZ-ICV-treated rats was conspicuously impaired by N(G)-nitro-l-arginine methyl ester (L-NAME) (20 mg/kg, 28 days) and wortmannin (5 μg/rat; ICV) treatments. Wortmannin (PI3-kinase inhibitor) and L-NAME groups manifested elevated brain oxidative stress, TNF-α content and AChE and LDH activity and diminished nitrite content. L-NAME (arginine-based competitive eNOS inhibitor) enhanced the eNOS expression (not activity) whereas wortmannin reduced the brain eNOS levels in EGA- and STZ-ICV-treated rats. However, the L-NAME group exhibited superior cognitive abilities in comparison to the wortmannin group. It can be concluded that EGA averted the memory deficits by precluding the STZ-ICV-induced loss of PI3-kinase-eNOS signalling in the brain of rats.









Similar content being viewed by others
Change history
17 April 2024
An Editorial Expression of Concern to this paper has been published: https://doi.org/10.1007/s00210-024-03096-6
References
Agrawal R, Tyagi E, Shukla R, Nath C (2011) Insulin receptor signaling in rat hippocampus: a study in STZ (ICV) induced memory deficit model. Eur Neuropsychopharmacol 21:261–273. https://doi.org/10.1016/j.euroneuro.2010.11.009
Austin SA, Santhanam AV, Hinton DJ, Choi DS, Katusic ZS (2013) Endothelial nitric oxide deficiency promotes Alzheimer’s disease pathology. J Neurochem 127:691–700. https://doi.org/10.1111/jnc.12334
Baki L, Shioi J, Wen P, Shao Z, Schwarzman A, Gama-Sosa M, Neve R, Robakis N (2004) PS1 activates PI3K thus inhibiting GSK-3 activity and tau overphosphorylation: effects of FAD mutations. EMBO J 23:2586–2596. https://doi.org/10.1038/sj.emboj.7600251
Bansal N, Yadav P, Kumar M (2017) Ellagic acid administration negated the development of streptozotocin-induced memory deficit in rats. Drug Res (Stuttg) 67:425–431. https://doi.org/10.1055/s-0043-108552
Bedse G, Di Domenico F, Serviddio G, Cassano T (2015) Aberrant insulin signaling in Alzheimer’s disease: current knowledge. Front Neurosci 9:204. https://doi.org/10.3389/fnins.2015.00204
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: current evidence and future directions. Alzheimers Dement 12:719–732. https://doi.org/10.1016/j.jalz.2016.02.010
Chen HT, Ruan NY, Chen JC, Lin TY (2012) Dopamine D2 receptor-mediated Akt/PKB signalling: initiation by the D2S receptor and role in quinpirole-induced behavioural activation. ASN Neuro 4:371–382. https://doi.org/10.1042/AN20120013
Claiborne A (1985) Catalase activity. In: Greenwald RA (ed) CRC handbook of methods for oxygen radical research, 3rd edn. CRC, Boca Raton, pp 283–284
Ding Y, Zhang B, Zhou K, Chen M, Wang M, Jia Y, Song Y, Li Y, Wen A (2014) Dietary ellagic acid improves oxidant-induced endothelial dysfunction and atherosclerosis: role of Nrf2 activation. Int J Cardiol 175:508–514. https://doi.org/10.1016/j.ijcard.2014.06.045
Doens D, Fernandez PL (2014) Microglia receptors and their implications in the response to amyloid β for Alzheimer’s disease pathogenesis. J Neuroinflammation 11:48. https://doi.org/10.1186/1742-2094-11-48
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Ellman GL, Courtney KD, VJr A, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Ferreira-Vieira TH, Guimaraes IM, Silva FR, Ribeiro FM (2016) Alzheimer’s disease: targeting the cholinergic system. Curr Neuropharmacol 14:101–115. https://doi.org/10.2174/1570159X13666150716165726
Franke TF (2008) PI3K/Akt: getting it right matters. Oncogene 27:6473–6488. https://doi.org/10.1038/onc.2008.313
Gao C, Liu Y, Jiang Y, Ding J, Li L (2014) Geniposide ameliorates learning memory deficits, reduces tau phosphorylation and decreases apoptosis via GSK3β pathway in streptozotocin-induced Alzheimer rat model. Brain Pathol 24:261–269. https://doi.org/10.1111/bpa.12116
Guha M, Mackman N (2002) The phosphatidylinositol 3-kinase-Akt pathway limits lipopolysaccharide activation of signaling pathways and expression of inflammatory mediators in human monocytic cells. J Biol Chem 277:32124–32132. https://doi.org/10.1074/jbc.M203298200
Harry GJ, Sills R, Schlosser MJ, Maier WE (2001) Neurodegeneration and glia response in rat hippocampus following nitro-L-arginine methyl ester (L-NAME). Neurotox Res 3:307–319
Horecker BL, Kornberg A (1948) The extinction coefficient of the reduced band of pyridine nucleotides. J Biol Chem 175:385–390
Kiasalari Z, Heydarifard R, Khalili M, Afshin-Majd S, Baluchnejadmojarad T, Zahedi E, Sanaierad A, Roghani M (2017) Ellagic acid ameliorates learning and memory deficits in a rat model of Alzheimer’s disease: an exploration of underlying mechanisms. Psychopharmacology 234:1841–1852. https://doi.org/10.1007/s00213-017-4589-6
Kong J, Ren G, Jia N, Wang Y, Zhang H, Zhang W, Chen B, Cao Y (2013) Effects of nicorandil in neuroprotective activation of PI3K/AKT pathways in a cellular model of Alzheimer’s disease. Eur Neurol 70:233–241. https://doi.org/10.1159/000351247
Landete JM (2011) Ellagitannins, ellagic acid and their derived metabolites: a review about source, metabolism, functions and health. Food Res Int 44:1150–1160. https://doi.org/10.1016/j.foodres.2011.04.027
Lee HJ, Ryu JM, Jung YH, Lee SJ, Kim JY, Lee SH, Hwang IK, Seong JK, Han HJ (2016) High glucose upregulates BACE1-mediated Aβ production through ROS-dependent HIF-1α and LXRα/ABCA1-regulated lipid raft reorganization in SK-N-MC cells. Sci Rep 6:36746. https://doi.org/10.1038/srep36746
Li ST, Pan J, Hua XM, Liu H, Shen S, Liu JF, Li B, Tao BB, Ge XL, Wang XH, Shi JH, Wang XQ (2014) Endothelial nitric oxide synthase protects neurons against ischemic injury through regulation of brain-derived neurotrophic factor expression. CNS Neurosci Ther 20:154–164. https://doi.org/10.1111/cns.12182
Lipinska L, Klewicka E, Sojka M (2014) The structure, occurrence and biological activity of ellagitannins: a general review. Acta Sci Pol Technol Aliment 13:289–299
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mansouri MT, Farbood Y, Naghizadeh B, Shabani S, Mirshekar MA, Sarkaki A (2016) Beneficial effects of ellagic acid against animal models of scopolamine- and diazepam-induced cognitive impairments. Pharm Biol 54:1947–1953. https://doi.org/10.3109/13880209.2015.1137601
Meng Y, Wang W, Kang J, Wang X, Sun L (2017) Role of the PI3K/AKT signalling pathway in apoptotic cell death in the cerebral cortex of streptozotocin-induced diabetic rats. Exp Ther Med 13:2417–2422. https://doi.org/10.3892/etm.2017.4259
Mohan S, Wu CC, Shin S, Fung HL (2012) Continuous exposure to L-arginine induces oxidative stress and physiological tolerance in cultured human endothelial cells. Amino Acids 43:1179–1188. https://doi.org/10.1007/s00726-011-1173-y
Moosavi M, Abbasi L, Zarifkar A, Rastegar K (2014) The role of nitric oxide in spatial memory stages, hippocampal ERK and CaMKII phosphorylation. Pharmacol Biochem Behav 122:164–172. https://doi.org/10.1016/j.pbb.2014.03.021
Morris RGM (1984) Development of a water-maze procedure for studying spatial learning in the rats. J Neurosci Methods 11:47–60
Murphy MP (1999) Nitric oxide and cell death. Biochim Biophys Acta 1411:401–414
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Ou HC, Lee WJ, Lee SD, Huang CY, Chiu TH, Tsai KL, Hsu WC, Sheu WH (2010) Ellagic acid protects endothelial cells from oxidized low-density lipoprotein-induced apoptosis by modulating the PI3K/Akt/eNOS pathway. Toxicol Appl Pharmacol 248:134–143. https://doi.org/10.1016/j.taap.2010.07.025
Paul V, Ekambaram P (2011) Involvement of nitric oxide in learning & memory processes. Indian J Med Res 133:471–478
Paxinos G, Watson CR, Emson PC (1980) AChE-stained horizontal sections of the rat brain in stereotaxic coordinates. J Neurosci Methods 3:129–149
Petanceska SS, Gandy S (1999) The phosphatidylinositol 3-kinase inhibitor wortmannin alters the metabolism of the Alzheimer’s amyloid precursor protein. J Neurochem 73:2316–2320
Prast H, Fischer H, Werner E, Werner-Felmayer G, Philippu A (1995) Nitric oxide modulates the release of acetylcholine in the ventral striatum of the freely moving rat. Naunyn Schmiedeberg’s Arch Pharmacol 352:67–73
Provias J, Jeynes B (2008) Neurofibrillary tangles and senile plaques in Alzheimer’s brains are associated with reduced capillary expression of vascular endothelial growth factor and endothelial nitric oxide synthase. Curr Neurovasc Res 5:199–205. https://doi.org/10.2174/156720208785425729
Rajasekar N, Nath C, Hanif K, Shukla R (2017) Intranasal insulin improves cerebral blood flow, Nrf-2 expression and BDNF in STZ (ICV)-induced memory impaired rats. Life Sci 173:1–10. https://doi.org/10.1016/j.lfs.2016.09.020
Resende RR, Adhikari A (2009) Cholinergic receptor pathways involved in apoptosis, cell proliferation and neuronal differentiation. Cell Commun Signal 7:20. https://doi.org/10.1186/1478-811X-7-20
Rickard NS, Gibbs ME, Ng KT (1999) Inhibition of the endothelial isoform of nitric oxide synthase impairs long-term memory formation in the chick. Learn Mem 6:458–466
Sastry KV, Moudgal RP, Mohan J, Tyagi JS, Rao GS (2002) Spectrophotometric determination of serum nitrite and nitrate by copper-cadmium alloy. Anal Biochem 306:79–82
Sharma N, Deshmukh R, Bedi KL (2010) SP600125, a competitive inhibitor of JNK attenuates streptozotocin induced neurocognitive deficit and oxidative stress in rats. Pharmacol Biochem Behav 96:386–394. https://doi.org/10.1016/j.pbb.2010.06.010
Son H, Hawkins RD, Martin K, Kiebler M, Huang PL, Fishman MC, Kandel ER (1996) Long-term potentiation is reduced in mice that are doubly mutant in endothelial and neuronal nitric oxide synthase. Cell 87:1015–1023
Traystman RJ, Moore LE, Helfaer MA, Davis S, Banasiak K, Williams M, Hurn PD (1995) Nitro-L-arginine analogues. Dose- and time-related nitric oxide synthase inhibition in brain. Stroke 26:864–869
Tyagi E, Agrawal R, Nath C, Shukla R (2010) Cholinergic protection via alpha7 nicotinic acetylcholine receptors and PI3K-Akt pathway in LPS-induced neuroinflammation. Neurochem Int 56:135–142. https://doi.org/10.1016/j.neuint.2009.09.011
Utkan T, Yazir Y, Karson A, Bayramgurler D (2015) Etanercept improves cognitive performance and increases eNOS and BDNF expression during experimental vascular dementia in streptozotocin-induced diabetes. Curr Neurovasc Res 12:135–146. https://doi.org/10.2174/1567202612666150311111340
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2:489–501. https://doi.org/10.1038/nrc839
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858
Winterbourn CC, Hawkins RE, Brian M, Carrell RW (1975) The estimation of red cell superoxide dismutase activity. J Lab Clin Med 85:337–341
Yang Y, Ma D, Xu W, Chen F, Du T, Yue W, Shao S, Yuan G (2016) Exendin-4 reduces tau hyperphosphorylation in type 2 diabetic rats via increasing brain insulin level. Mol Cell Neurosci 70:68–75. https://doi.org/10.1016/j.mcn.2015.10.005
Yoshida T, Amakura Y, Yoshimura M (2010) Structural features and biological properties of ellagitannins in some plant families of the order Myrtales. Int J Mol Sci 11:79–106. https://doi.org/10.3390/ijms11010079
Zhao Y, Hu X, Liu Y, Dong S, Wen Z, He W, Zhang S, Huang Q, Shi M (2017) ROS signaling under metabolic stress: cross-talk between AMPK and AKT pathway. Mol Cancer 16:79. https://doi.org/10.1186/s12943-017-0648-1
Acknowledgements
The authors are thankful to the management of ASBASJSM College of Pharmacy, Bela, for providing the necessary research facilities and IKG Punjab Technical University, Kapurthala.
Funding
This research was assisted by a grant from AICTE, New Delhi (India), under Research Promotion Scheme.
Author information
Authors and Affiliations
Contributions
Prof. (Dr.) Nitin Bansal designed this study. Manish Kumar (PhD research scholar) conducted the research and analysed and interpreted the data. Both authors wrote the initial and final draft of the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kumar, M., Bansal, N. Ellagic acid prevents dementia through modulation of PI3-kinase-endothelial nitric oxide synthase signalling in streptozotocin-treated rats. Naunyn-Schmiedeberg's Arch Pharmacol 391, 987–1001 (2018). https://doi.org/10.1007/s00210-018-1524-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1524-2