Abstract
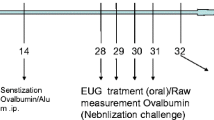
The overlapping between asthmatic subtypes, including both CD4+ T helper (TH)2 and TH17 cells, is found in the natural course of allergic asthma, especially in exacerbations and severe and insensitive forms to steroids, which are in need of new molecular therapies. In the TH2-subset mediated asthma, fenofibrate displays therapeutic promises, besides evidenced therapeutic effects on TH17-mediated colitis and myocarditis. Therefore, the effects of fenofibrate versus dexamethasone on IL-23/IL-17 axis in ovalbumin (OVA)/lipopolysaccharide (LPS)-induced airway inflammation and bronchial asthma in rats were explored. The OVA/LPS sensitization and challenge were performed for 28 days in male Wistar rats. After sensitization, fenofibrate (100 mg/kg/day) or dexamethasone (2.5 mg/kg/day) was orally administered from the day 15 to 28. Either fenofibrate or dexamethasone attenuated the severity of OVA/LPS-induced airway inflammation and bronchial asthma through significant ameliorations in the total serum immunoglobulin (Ig)E assay; the total and differential leukocytic counts in the bronchoalveolar lavage (BAL) fluid; the lung inflammatory cytokines such as interleukin (IL)-4, IL-13, IL-17, and IL-23, transforming growth factor (TGF)-β1, and tumor necrosis factor(TNF)-α levels; and the lung IL-17 and IL-23 expressions. In addition to the reduction in the inflammatory and fibrotic histopathological scores, fenofibrate significantly ameliorated the BAL neutrophilic count and the lung IL-17 and IL-23 expressions in comparison to dexamethasone. The suppression of IL-23/IL-17 axis could be considered a molecular therapeutic target for fenofibrate in OVA/LPS-induced airway inflammation and bronchial asthma. Combined therapeutic regimens of fenofibrate and steroids should be furtherly investigated in severe and resistant asthma.










Similar content being viewed by others
References
Arora P, Ansari SH, Anjum V, Mathur R, Ahmad S (2017) Investigation of anti-asthmatic potential of Kanakasava in ovalbumin-induced bronchial asthma and airway inflammation in rats. J Ethnopharmacol 197:242–249. https://doi.org/10.1016/j.jep.2016.07.082
Barnes PJ (2011) Pathophysiology of allergic inflammation. Immunol Rev 242(1):31–50. https://doi.org/10.1111/j.1600-065X.2011.01020.x
Becker J, Delayre-Orthez C, Frossard N, Pons F (2008) Regulation of peroxisome proliferator-activated receptor-alpha expression during lung inflammation. Pulm Pharmacol Ther 21(2):324–330. https://doi.org/10.1016/j.pupt.2007.08.001
Becker J, Delayre-Orthez C, Frossard N, Pons F (2006) Regulation of inflammation by PPARs: a future approach to treat lung inflammatory diseases? Fundam Clin Pharmacol 20(5):429–447. https://doi.org/10.1111/j.1472-8206.2006.00425.x
Becker J, Delayre-Orthez C, Frossard N, Pons F (2012) The peroxisome proliferator-activated receptor alpha agonist fenofibrate decreases airway reactivity to methacholine and increases endothelial nitric oxide synthase phosphorylation in mouse lung: fenofibrate and airway reactivity. Fundam Clin Pharmacol 26(3):340–346. https://doi.org/10.1111/j.1472-8206.2011.00935.x
Belvisi MG, Mitchell JA (2009) Targeting PPAR receptors in the airway for the treatment of inflammatory lung disease. Br J Pharmacol 158(4):994–1003. https://doi.org/10.1111/j.1476-5381.2009.00373.x
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, Weiner HL, Kuchroo VK (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441(7090):235–238. https://doi.org/10.1038/nature04753
Bi Y, Yang R (2012) Direct and indirect regulatory mechanisms in TH17 cell differentiation and functions: direct and indirect regulatory mechanisms in TH17 cell. Scand J Immunol 75(6):543–552. https://doi.org/10.1111/j.1365-3083.2012.02686.x
Bolton PB, Lefevre P, McDonald DM (1997) Salmeterol reduces early- and late-phase plasma leakage and leukocyte adhesion in rat airways. Am J Respir Crit Care Med 155(4):1428–1435. https://doi.org/10.1164/ajrccm.155.4.9105089
Busse WW, Holgate S, Kerwin E, Chon Y, Feng JY, Lin J, Lin SL (2013) Randomized, double-blind, placebo-controlled study of brodalumab, a human anti–IL-17 receptor monoclonal antibody, in moderate to severe asthma. Am J Respir Crit Care Med 188(11):1294–1302. https://doi.org/10.1164/rccm.201212-2318OC
Chakir J, Shannon J, Molet S, Fukakusa M, Elias J, Laviolette M, Boulet LP, Hamid Q (2003) Airway remodeling-associated mediators in moderate to severe asthma: effect of steroids on TGF-β, IL-11, IL-17, and type I and type III collagen expression. J Allergy Clin Immunol 111(6):1293–1298. https://doi.org/10.1067/mai.2003.1557
Cheng H, Xi Y, Chi X, Wu Y, Liu G (2016) Fenofibrate treatment of rats with experimental autoimmune myocarditis by alleviating Treg/Th17 disorder. Cent Eur J Immunol 1(1):64–70. https://doi.org/10.5114/ceji.2016.58817
Choy DF, Hart KM, Borthwick LA, Shikotra A, Nagarkar DR, Siddiqui S, Jia G, Ohri CM, Doran E, Vannella KM, Butler CA, Hargadon B, Sciurba JC, Gieseck RL, Thompson RW, White S, Abbas AR, Jackman J, Wu LC, Egen JG, Heaney LG, Ramalingam TR, Arron JR, Wynn TA, Bradding P (2015) TH2 and TH17 inflammatory pathways are reciprocally regulated in asthma. Sci Transl Med 7(301):301ra129. https://doi.org/10.1126/scitranslmed.aab3142
Chung Y, Chang SH, Martinez GJ, Yang XO, Nurieva R, Kang HS, Ma L, Watowich SS, Jetten AM, Tian Q, Dong C (2009) Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 30(4):576–587. https://doi.org/10.1016/j.immuni.2009.02.007
Delayre-Orthez C, Becker J, Auwerx J, Frossard N, Pons F (2008) Suppression of allergen-induced airway inflammation and immune response by the peroxisome proliferator-activated receptor-alpha agonist fenofibrate. Eur J Pharmacol 581(1-2):177–184. https://doi.org/10.1016/j.ejphar.2007.11.040
Delayre-Orthez C, Becker J, Guenon I, Lagente V, Auwerx J, Frossard N, Pons F (2005) PPARα downregulates airway inflammation induced by lipopolysaccharide in the mouse. Respir Res 6(1):91. https://doi.org/10.1186/1465-9921-6-91
Denning GM, Stoll LL (2006) Peroxisome proliferator-activated receptors: potential therapeutic targets in lung disease? Pediatr Pulmonol 41(1):23–34. https://doi.org/10.1002/ppul.20338
Dong F, Wang C, Duan J, Zhang W, Xiang D, Li M (2014) Puerarin attenuates ovalbumin-induced lung inflammation and hemostatic unbalance in rat asthma model. Evid Based Complement Alternat Med 2014:1–9. https://doi.org/10.1155/2014/726740
Dong L, Li H, Wang S, Li Y (2009) Different doses of lipopolysaccharides regulate the lung inflammation of asthmatic mice via TLR4 pathway in alveolar macrophages. J Asthma Off J Assoc Care Asthma 46(3):229–233. https://doi.org/10.1080/02770900802610050
Estrela GR, Wasinski F, Batista RO, Hiyane MI, Felizardo RJF, Cunha F, de Almeida DC, Malheiros DMAC, Câmara NOS, Barros CC, Bader M, Araujo RC (2017) Caloric restriction is more efficient than physical exercise to protect from cisplatin nephrotoxicity via PPAR-alpha activation. Front Physiol 8. https://doi.org/10.3389/fphys.2017.00116
Elwood W, Lötvall JO, Barnes PJ, Chung KF (1991) Characterization of allergen-induced bronchial hyperresponsiveness and airway inflammation in actively sensitized brown-Norway rats. J Allergy Clin Immunol 88(6):951–960. https://doi.org/10.1016/0091-6749(91)90253-K
Guo Y, Huang H, Zeng D et al (2013) Interleukin (IL)-4 induces production of cytokine-induced neutrophil chemoattractants (CINCs) and intercellular adhesion molecule (ICAM)-1 in lungs of asthmatic rats. J Huazhong Univ Sci Technol Med Sci 33(4):470–478. https://doi.org/10.1007/s11596-013-1144-5
Haddad EB, Birrell M, McCluskie K, Ling A, Webber SE, Foster ML, Belvisi MG (2001) Role of p38 MAP kinase in LPS-induced airway inflammation in the rat. Br J Pharmacol 132(8):1715–1724. https://doi.org/10.1038/sj.bjp.0704022
Halwani R, Al-Muhsen S, Al-Jahdali H, Hamid Q (2011) Role of transforming growth factor-β in airway remodeling in asthma. Am J Respir Cell Mol Biol 44(2):127–133. https://doi.org/10.1165/rcmb.2010-0027TR
Hastie AT, Moore WC, Meyers DA, Vestal PL, Li H, Peters SP, Bleecker ER, National Heart, Lung, and Blood Institute Severe Asthma Research Program (2010) Analyses of asthma severity phenotypes and inflammatory proteins in subjects stratified by sputum granulocytes. J Allergy Clin Immunol 125(5):1028–1036.e13. https://doi.org/10.1016/j.jaci.2010.02.008
Huang YJ, Boushey HA (2013) The bronchial microbiome and asthma phenotypes. Am J Respir Crit Care Med 188(10):1178–1180. https://doi.org/10.1164/rccm.201309-1702ED
Jung CH, Lee JY, Cho CH, Kim CJ (2007) Anti-asthmatic action of quercetin and rutin in conscious guinea-pigs challenged with aerosolized ovalbumin. Arch Pharm Res 30(12):1599–1607. https://doi.org/10.1007/BF02977330
Ke X, Huang J, Chen Q, Hong S, Zhu D (2010) Protective effects of combined Mycobacterium bovis BCG and interleukin-12 vaccination on airway inflammation in a murine model of allergic asthma. Clin Invest Med 33(3):196–202. https://doi.org/10.25011/cim.v33i3.13726
Khanna D, FitzGerald JD, Khanna PP et al (2012) 2012 American College of Rheumatology guidelines for management of gout Part I: Systematic non-pharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res 64(10):1431–1446. https://doi.org/10.1002/acr.21772
Kim S-H, Hong J-H, Lee Y-C (2013) Ursolic acid, a potential PPARγ agonist, suppresses ovalbumin-induced airway inflammation and Penh by down-regulating IL-5, IL-13, and IL-17 in a mouse model of allergic asthma. Eur J Pharmacol 701(1-3):131–143. https://doi.org/10.1016/j.ejphar.2012.11.033
Krysiak R, Gdula-Dymek A, Okopien B (2013) The effect of fenofibrate on lymphocyte release of proinflammatory cytokines and systemic inflammation in simvastatin-treated patients with atherosclerosis and early glucose metabolism disturbances. Basic Clin Pharmacol Toxicol 112(3):198–202. https://doi.org/10.1111/bcpt.12003
Kumari A, Dash D, Singh R (2015) Lipopolysaccharide (LPS) exposure differently affects allergic asthma exacerbations and its amelioration by intranasal curcumin in mice. Cytokine 76(2):334–342. https://doi.org/10.1016/j.cyto.2015.07.022
Kurucz I, Szelenyi I (2006) Current animal models of bronchial asthma. Curr Pharm Des 12(25):3175–3194. https://doi.org/10.2174/138161206778194169
Lambrecht BN, Hammad H (2014) The immunology of asthma. Nat Immunol 16(1):45–56. https://doi.org/10.1038/ni.3049
Lee JW, Bajwa PJ, Carson MJ, Jeske DR, Cong Y, Elson CO, Lytle C, Straus DS (2007) Fenofibrate represses interleukin-17 and interferon-gamma expression and improves colitis in interleukin-10-deficient mice. Gastroenterology 133(1):108–123. https://doi.org/10.1053/j.gastro.2007.03.113
Li X-Z, Feng J-T, C-P H et al (2010) Effects of Arkadia on airway remodeling through enhancing TGF-β signaling in allergic rats. Lab Investig 90(7):997–1003. https://doi.org/10.1038/labinvest.2010.78
Linz PE, Lovato LC, Byington RP, O’Connor PJ, Leiter LA, Weiss D, Force RW, Crouse JR, Ismail-Beigi F, Simmons DL, Papademetriou V, Ginsberg HN, Elam MB (2014) Paradoxical reduction in HDL-C with fenofibrate and thiazolidinedione therapy in type 2 diabetes: the ACCORD Lipid trial. Diabetes Care 37(3):686–693. https://doi.org/10.2337/dc13-0790
Lowe APP, Thomas RS, Nials AT, Kidd EJ, Broadley KJ, Ford WR (2017) Route of administration affects corticosteroid sensitivity of a combined ovalbumin and lipopolysaccharide model of asthma exacerbation in guinea pigs. J Pharmacol Exp Ther 362(2):327–337. https://doi.org/10.1124/jpet.117.241927
McKeage K, Keating GM (2011) Fenofibrate: a review of its use in dyslipidaemia. Drugs 71(14):1917–1946. https://doi.org/10.2165/11208090-000000000-00000
Mizutani N, Goshima H, Nabe T, Yoshino S (2012) Complement C3a-induced IL-17 plays a critical role in an IgE-mediated late-phase asthmatic response and airway hyperresponsiveness via neutrophilic inflammation in mice. J Immunol 188(11):5694–5705. https://doi.org/10.4049/jimmunol.1103176
Moraes LA, Piqueras L, Bishop-Bailey D (2006) Peroxisome proliferator-activated receptors and inflammation. Pharmacol Ther 110(3):371–385. https://doi.org/10.1016/j.pharmthera.2005.08.007
Morishima Y, Ano S, Ishii Y, Ohtsuka S, Matsuyama M, Kawaguchi M, Hizawa N (2013) Th17-associated cytokines as a therapeutic target for steroid-insensitive asthma. Clin Dev Immunol 2013:1–9. https://doi.org/10.1155/2013/609395
Muti A, Bolfa P, Muti L, Muresan A (2014) Vitamin E attenuates the degree of histopathological lung damage following toluene diisocyanate administration in rats
Ogura H, Murakami M, Okuyama Y, Tsuruoka M, Kitabayashi C, Kanamoto M, Nishihara M, Iwakura Y, Hirano T (2008) Interleukin-17 promotes autoimmunity by triggering a positive-feedback loop via interleukin-6 induction. Immunity 29(4):628–636. https://doi.org/10.1016/j.immuni.2008.07.018
Palomares Ó, Sánchez-Ramón S, Dávila I, Prieto L, Pérez de Llano L, Lleonart M, Domingo C, Nieto A (2017) How IgE axis contributes to the continuum of allergic asthma and anti-IgE therapies. Int J Mol Sci 18(6):1328. https://doi.org/10.3390/ijms18061328
Park SJ, Lee YC (2010) Interleukin-17 regulation: an attractive therapeutic approach for asthma. Respir Res 11(1):78. https://doi.org/10.1186/1465-9921-11-78
Rehnberg M, Ramnegård M, Krutrök N et al (2015) The role of lL-17 in the OVA-LPS driven model of lung inflammation. Eur Respir J 46:PA4010. https://doi.org/10.1183/13993003.congress-2015.PA4010
Robinson D, Humbert M, Buhl R, Cruz AA, Inoue H, Korom S, Hanania NA, Nair P (2017) Revisiting type 2-high and type 2-low airway inflammation in asthma: current knowledge and therapeutic implications. Clin Exp Allergy 47(2):161–175. https://doi.org/10.1111/cea.12880
Samah M, El-Aidy AE-R, Tawfik MK, Ewais MMS (2012) Evaluation of the antifibrotic effect of fenofibrate and rosiglitazone on bleomycin-induced pulmonary fibrosis in rats. Eur J Pharmacol 689(1-3):186–193. https://doi.org/10.1016/j.ejphar.2012.05.026
Sharma S, Sharma A, Kumar S, Sharma SK, Ghosh B (2006) Association of TNF haplotypes with asthma, serum IgE levels, and correlation with serum TNF-α levels. Am J Respir Cell Mol Biol 35(4):488–495. https://doi.org/10.1165/rcmb.2006-0084OC
Shoseyov D (2005) Treatment of ovalbumin-induced experimental allergic bronchitis in rats by inhaled inhibitor of secretory phospholipase A2. Thorax 60(9):747–753. https://doi.org/10.1136/thx.2005.043695
Silva JAF, Ferrucci DL, Peroni LA, Abrahão PGS, Salamene AF, Rossa-Junior C, Carvalho HF, Stach-Machado DR (2012) Sequential IL-23 and IL-17 and increased Mmp8 and Mmp14 expression characterize the progression of an experimental model of periodontal disease in type 1 diabetes. J Cell Physiol 227(6):2441–2450. https://doi.org/10.1002/jcp.22979
Simpson JL, Scott R, Boyle MJ, Gibson PG (2006) Inflammatory subtypes in asthma: assessment and identification using induced sputum. Respirol Carlton Vic 11(1):54–61. https://doi.org/10.1111/j.1440-1843.2006.00784.x
Singh A, Yamamoto M, Ruan J, Choi J, Gauvreau GM, Olek S, Hoffmueller U, Carlsten C, FitzGerald J, Boulet LP, O'Byrne PM, Tebbutt SJ (2014) Th17/Treg ratio derived using DNA methylation analysis is associated with the late phase asthmatic response. Allergy Asthma Clin Immunol 10(1):32. https://doi.org/10.1186/1710-1492-10-32
Steinke JW, Borish L (2001) Th2 cytokines and asthma—interleukin-4: its role in the pathogenesis of asthma, and targeting it for asthma treatment with interleukin-4 receptor antagonists. Respir Res 2(2):66–70. https://doi.org/10.1186/rr40
Szelenyi I (2000) Animal models of bronchial asthma. Inflamm Res Off J Eur Histamine Res Soc Al 49(12):639–654. https://doi.org/10.1007/s000110050642
Tarantino N, Santoro F, De Gennaro L, Correale M, Guastafierro F, Gaglione A, di Biase M, Brunetti ND (2017) Fenofibrate/simvastatin fixed-dose combination in the treatment of mixed dyslipidemia: safety, efficacy, and place in therapy. Vasc Health Risk Manag Volume 13:29–41. https://doi.org/10.2147/VHRM.S95044
Vazquez-Tello A, Halwani R, Hamid Q, Al-Muhsen S (2013) Glucocorticoid receptor-beta up-regulation and steroid resistance induction by IL-17 and IL-23 cytokine stimulation in peripheral mononuclear cells. J Clin Immunol 33(2):466–478. https://doi.org/10.1007/s10875-012-9828-3
Wang L-P, Wang Y-W, Wang B-Z, et al (2014) Expression of interleukin-17A in lung tissues of irradiated mice and the influence of dexamethasone. In: Sci. World J. https://www.hindawi.com/journals/tswj/2014/251067/. Accessed 15 Oct 2017
Wang Y-H, Wills-Karp M (2011) The potential role of interleukin-17 in severe asthma. Curr Allergy Asthma Rep 11(5):388–394. https://doi.org/10.1007/s11882-011-0210-y
Acknowledgments
Thanks to Dr. Radwa A. Noureldin, assistant lecturer of Radiology, Faculty of Medicine, Suez Canal University, Egypt, for language proofing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Statement on welfare of animals
All animal care and experimental procedures were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (MD, USA) and were approved by the Institutional Animal Care and Use Committee at the Faculty of Medicine, Suez Canal University, Egypt.
Rights and permissions
About this article
Cite this article
Elaidy, S.M., Essawy, S.S., Hussain, M.A. et al. Modulation of the IL-23/IL-17 axis by fenofibrate ameliorates the ovalbumin/lipopolysaccharide-induced airway inflammation and bronchial asthma in rats. Naunyn-Schmiedeberg's Arch Pharmacol 391, 309–321 (2018). https://doi.org/10.1007/s00210-017-1459-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-017-1459-z