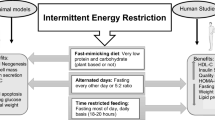
Abstract
The inappropriate dietary habits lead to the onset of age-related pathologies which include diabetes and cardiovascular ailments. Dietary restriction and nutritional therapy play an important role in the prevention of these chronic ailments. Preclinical research provides a basis for the therapeutic exploration of new dietary interventions for the clinical trials to potentiate the scientific management of diabetes and its related complications which further help in translating these nutritional improvements from bench to bedside. Within the same context, numerous therapeutically proved preclinical dietary interventions like high-fiber diet, caloric restriction, soy isoflavone-containing diets, etc., have shown the promising results for the management of diabetes and the associated complications. The focus of the present review is to highlight the various preclinical evidences of diet restriction for the management of diabetes and which will be helpful for enlightening the new ideas of nutritional therapy for future research exploration. In addition, some potential approaches are also discussed which are associated with various nutritional interventions to combat progressive diabetes and the associated disorders.

ᅟ
Similar content being viewed by others
Abbreviations
- AGES:
-
Advanced glycation end products
- AMPK:
-
AMP-activated protein kinase
- CR:
-
Caloric restriction
- DN:
-
Diabetic nephropathy
- DNA:
-
Deoxyribonucleic acid
- DR:
-
Dietary restriction
- ET:
-
Exercise training
- GLUT2:
-
Glucose transporter 2
- HbAIC:
-
Hemoglobin A1c or glycated hemoglobin
- HDL:
-
High-density lipoprotein
- HFD:
-
High-fat diet
- IGF-1:
-
Insulin-like growth factor 1
- KD:
-
Ketogenic diet
- LCKD:
-
Low-carbohydrate ketogenic diet
- LDL:
-
Low-density lipoprotein
- LFSMP:
-
Low-fat soy milk powder
- MDA:
-
Malondialdehyde
- MNT:
-
Medical nutrition therapy
- mTORC1:
-
Mammalian target of rapamycin complex 1
- NAD+:
-
Nicotinamide adenine dinucleotide
- OLETF:
-
Otsuka-Long-Evans-Tokushima fatty
- PPAR:
-
Peroxisome proliferator-activated receptor
- SIRT:
-
Sirtuins
- STZ:
-
Streptozotocin
- T2DM:
-
Type 2 diabetes mellitus
- TG:
-
Triglyceride
- WHO:
-
World Health Organization
References
Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti H (2011) Low carbohydrate ketogenic diet prevents the induction of diabetes using streptozotocin in rats. Exp Toxicol Pathol 63(7):663–669. https://doi.org/10.1016/j.etp.2010.05.008
Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti HM (2009) Therapeutic role of low-carbohydrate ketogenic diet in diabetes. Nutrition 25(11):1177–1185. https://doi.org/10.1016/j.nut.2009.04.004
American Dietetic Association (2008) Diabetes type 1 and 2 evidence-based nutrition practice guidelines for adults Chicago, IL. http://wwwadaevidencelibrary.com/topiccfm?=3252. Accessed 12 Aug 2017
Andrade EF, Lima AR, Nunes IE, Orlando DR, Gondim PN, Zangeronimo MG, Alves FH, Pereira LJ (2016) Exercise and beta-glucan consumption (Saccharomyces cerevisiae) improve the metabolic profile and reduce the Atherogenic Index in type 2 diabetic rats (HFD/STZ). Nutrients 8(12):792. https://doi.org/10.3390/nu8120792
Azadbakht L, Atabak S, Esmaillzadeh A (2008) Soy protein intake, cardiorenal indices, and C-reactive protein in type 2 diabetes with nephropathy. Diabetes Care 31(4):648–654. https://doi.org/10.2337/dc07-2065
Azar ST, Beydoun HM, Albadri MR (2016) Benefits of ketogenic diet for management of type two diabetes: a review. J Obes Eat Disord 2:1–3
Bantle JP, Wylie-Rosett J, Albright AL, Apovian CM, Clark NG, Franz MJ, Hoogwerf BJ, Lichtenstein AH, Mayer-Davis E, Mooradian AD, Wheeler ML, American Diabetes Association (2008) Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 31(Suppl. 1):S61–S78
Barcelli UO, Weiss M, Beach D, Motz A, Thompson B (1990) High linoleic acid diets ameliorate diabetic nephropathy in rats. Am J Kidney Dis 16(3):244–251. https://doi.org/10.1016/S0272-6386(12)81024-9
Broderick TL, Jankowski M, Gutkowska J (2017) The effects of exercise training and caloric restriction on the cardiac oxytocin natriuretic peptide system in the diabetic mouse. Diabetes Metab Syndr Obes 10:27–36. https://doi.org/10.2147/DMSO.S115453
Campbell AP, Rains TM (2015) Dietary protein is important in the practical management of prediabetes and type 2 diabetes. J Nutr 145(1):164S–169S. https://doi.org/10.3945/jn.114.194878
Cappelli AP, Zoppi CC, Silveira LR, Batista TM, Paula FM, da Silva PM, Rafacho A, Barbosa-Sampaio HC, Boschero AC, Carneiro EM (2017) Reduced glucose-induced insulin secretion in low-protein-fed rats is associated with altered pancreatic islets redox status. J Cell Physiol
Chandalia M, Garg A, Lutjohann D, von Bergmann K, Grundy SM, Brinkley LJ (2000) Beneficial effects of high dietary fiber intake in patients with type 2 diabetes mellitus. N Engl J Med 342(19):1392–1398. https://doi.org/10.1056/NEJM200005113421903
Choi YE, Ahn SK, Lee WT, Lee JE, Park SH, Yoon BB, Park KA (2010) Soybeans ameliolate diabetic nephropathy in rats. Evid Based Complement Alternat Med 7(4):433–440. https://doi.org/10.1093/ecam/nen021
Collier GR, Kalmusky J, Guidici S (1985) Effect of slowly digested carbohydrates in type I diabetic children. Diabetes 32:33A
Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM, Allison DB, Cruzen C, Simmons HA, Kemnitz JW, Weindruch R (2009) Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 325(5937):201–204. https://doi.org/10.1126/science.1173635
Dashti HM, Al-Zaid NS, Mathew TC, Al-Mousawi M, Talib H, Asfar SK, Behbahani AI (2006) Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol Cell Biochem 286(1-2):1–9. https://doi.org/10.1007/s11010-005-9001-x
Fontana L, Partridge L, Longo VD (2010) Extending healthy life span-from yeast to humans. Science 328(5976):321–326. https://doi.org/10.1126/science.1172539
Fontana L, Weiss EP, Villareal DT, Klein S, Holloszy JO (2008) Long-term effects of calorie or protein restriction on serum IGF-1 and IGFBP-3 concentration in humans. Aging Cell 7(5):681–687. https://doi.org/10.1111/j.1474-9726.2008.00417.x
Franz MJ, Boucher JL, Green-Pastors J, Powers MA (2008) Evidence-based nutrition practice guidelines for diabetes and scope and standards of practice. J Am Diet Assoc 108(1):S52–S58. https://doi.org/10.1016/j.jada.2008.01.021
Franz MJ, Powers MA, Leontos C, Holzmeister LA, Kulkarni K, Monk A, Wedel N, Gradwell E (2010) The evidence for medical nutrition therapy for type 1 and type 2 diabetes in adults. J Am Diet Assoc 110(12):1852–1889. https://doi.org/10.1016/j.jada.2010.09.014
Friedman AN (2004) High-protein diets: potential effects on the kidney in renal health and disease. Am J Kidney Dis 44(6):950–962. https://doi.org/10.1053/j.ajkd.2004.08.020
Gower BA, Goss AM (2015) A lower-carbohydrate, higher-fat diet reduces abdominal and intermuscular fat and increases insulin sensitivity in adults at risk of type 2 diabetes. J Nutr 145(1):177S–183S. https://doi.org/10.3945/jn.114.195065
Hamid A, Ilyas M, Kalsoom S, Bhatty N (2017) Effects of wheat bran diet and maize bran diet on the random blood glucose and weight of alloxan induced diabetic rats. J Anim Plant Sci 27(1):325–330
Hannan JM, Rokeya B, Faruque O, Nahar N, Mosihuzzaman M, Khan AA, Ali L (2003) Effect of soluble dietary fibre fraction of Trigonella foenum graecum on glycemic, insulinemic, lipidemic and platelet aggregation status of type 2 diabetic model rats. J Ethnopharmacol 88(1):73–77. https://doi.org/10.1016/S0378-8741(03)00190-9
Hofmann SM, Dong HJ, Li Z, Cai W, Altomonte J, Thung SN, Zeng F, Fisher EA, Vlassara H (2002) Improved insulin sensitivity is associated with restricted intake of dietary glycoxidation products in the db/db mouse. Diabetes 51(7):2082–2089. https://doi.org/10.2337/diabetes.51.7.2082
Holman RR, Steemson J, Darling P, Turner RC (1987) No glycemic benefit from guar administration in NIDDM. Diabetes Care 10(1):68–71. https://doi.org/10.2337/diacare.10.1.68
Hussain TA, Mathew TC, Dashti AA, Asfar S, Al-Zaid N, Dashti HM (2012) Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition 28(10):1016–1021. https://doi.org/10.1016/j.nut.2012.01.016
Ikeda Y, Enomoto H, Tajima S, Izawa-Ishizawa Y, Kihira Y, Ishizawa K, Tomita S, Tsuchiya K, Tamaki T (2013) Dietary iron restriction inhibits progression of diabetic nephropathy in db/db mice. Am J Physiol Ren Physiol 304(7):F1028–F1036. https://doi.org/10.1152/ajprenal.00473.2012
Imai S, Guarente L (2010) Ten years of NAD-dependent SIR2 family deacetylases: implications for metabolic diseases. Trends Pharmacol Sci 31(5):212–220. https://doi.org/10.1016/j.tips.2010.02.003
Ismaiel M, Yang H, Cui M (2017) Evaluation of high fibers Okara and soybean bran as functional supplements for mice with experimentally induced type 2 diabetes. PJFNS
Jenkins A, Josse RG (1985) Effects of processing/preparation as the blood glucose response to potatoes in diabetic patients. Diabetes (Suppl) 34:32A
Jheng HF, Hirotsuka M, Goto T, Shibata M, Matsumura Y, Kawada T (2017) Dietary low-fat soy milk powder retards diabetic nephropathy progression via inhibition of renal fibrosis and renal inflammation. Mol Nutr Food Res 61(3)
Jürgens H, Neschen S, Ortmann S, Scherneck S, Schmolz K, Schüler G, Schmidt S, Blüher M, Klaus S, Perez-Tilve D, Tschöp M, Schürmann A, Joost HG (2007) Development of diabetes in obese, insulinresistant mice: essential role of dietary carbohydrate in ß-cell destruction. Diabetologia 50(7):1481–1489. https://doi.org/10.1007/s00125-007-0662-8
Kahn SE, Hull RL, Utzschneider KM (2006) Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444(7121):840–846. https://doi.org/10.1038/nature05482
Kim H, Kang H, Heo RW, Jeon BT, Yi CO, Shin HJ, Kim J, Jeong SY, Kwak W, Kim WH, Kang SS (2016) Caloric restriction improves diabetes-induced cognitive deficits by attenuating neurogranin-associated calcium signaling in high-fat diet-fed mice. J Cereb Blood Flow Metab 6:1098–1110
Kitada M, Ogura Y, Suzuki T, Sen S, Lee SM, Kanasaki K, Kume S, Koya D (2016) A very-low-protein diet ameliorates advanced diabetic nephropathy through autophagy induction by suppression of the mTORC1 pathway in Wistar fatty rats, an animal model of type 2 diabetes and obesity. Diabetologia 59(6):1307–1317. https://doi.org/10.1007/s00125-016-3925-4
Kluth O, Mirhashemi F, Scherneck S, Kaiser D, Kluge R, Neschen S, Joost HG, Schürmann A (2011) Dissociation of lipotoxicity and glucotoxicity in a mouse model of obesity associated diabetes: role of forkhead box O1 (FOXO1) in glucose-induced beta cell failure. Diabetologia 54(3):605–616. https://doi.org/10.1007/s00125-010-1973-8
Knight DC, Eden JA (1996) A review of the clinical effects of phytoestrogens. Obstet Gynecol 87(5):897–904
Koh GY, Rowling MJ, Schalinske KL, Grapentine K, Loo YT (2016) Consumption of dietary resistant starch partially corrected the growth pattern despite hyperglycemia and compromised kidney function in streptozotocin-induced diabetic rats. J Agric Food Chem 64(40):7540–7545. https://doi.org/10.1021/acs.jafc.6b03808
Kuiper GG, Lemmen JG, Carlsson BO, Corton JC, Safe SH, Van Der Saag PT, Van Der Burg B, Gustafsson JA (1998) Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor β. Endocrinology 139(10):4252–4263. https://doi.org/10.1210/endo.139.10.6216
Kume S, Thomas MC, Koya D (2012) Nutrient sensing, autophagy, and diabetic nephropathy. Diabetes 61(1):23–29. https://doi.org/10.2337/db11-0555
Lee JS (2006) Effects of soy protein and genistein on blood glucose, antioxidant enzyme activities, and lipid profile in streptozotocin-induced diabetic rats. Life Sci 79(16):1578–1584. https://doi.org/10.1016/j.lfs.2006.06.030
Lee SM (1982) The effect of a high fibre diet on diabetic nephropathy in the db/db mouse. Diabetologia 22(5):349–353
Lee SM, Bressler R (1981) Prevention of diabetic nephropathy by diet control in the db/db mouse. Diabetes 30(2):106–111. https://doi.org/10.2337/diab.30.2.106
Leiter EH, Coleman DL, Ingram DK, Reynolds MA (1983) Influence of dietary carbohydrate on the induction of diabetes in C57BL/KsJ-db/db diabetes mice. J Nutr 113(1):184–195
Li J, Kaneko T, Qin LQ, Wang J, Wang Y, Sato A (2003) Long-term effects of high dietary fiber intake on glucose tolerance and lipid metabolism in GK rats: comparison among barley, rice, and cornstarch. Metabolism 52(9):1206–1210. https://doi.org/10.1016/S0026-0495(03)00159-8
Li N, Guenancia C, Rigal E, Hachet O, Chollet P, Desmoulins L, Leloup C, Rochette L, Vergely C (2016) Short-term moderate diet restriction in adulthood can reverse oxidative, cardiovascular and metabolic alterations induced by postnatal overfeeding in mice. Sci Rep 6
Lu MP, Wang R, Song X, Chibbar R, Wang X, Wu L, Meng QH (2008) Dietary soy isoflavones increase insulin secretion and prevent the development of diabetic cataracts in streptozotocin-induced diabetic rats. Nutr Res 28(7):464–471
Madar Z (1983) Effect of brown rice and soybean dietary fiber on the control of glucose and lipid metabolism in diabetic rats. Am J Clin Nutr 38(3):388–393
Markovic TP, Jenkins AB, Campbell LV, Furler SM, Kraegen EW, Chisholm DJ (1998) The determinants of glycemic responses to diet restriction and weight loss in obesity and NIDDM. Diabetes Care 21(5):687–694. https://doi.org/10.2337/diacare.21.5.687
Minamiyama Y, Bito Y, Takemura S, Takahashi Y, Kodai S, Mizuguchi S, Nishikawa Y, Suehiro S, Okada S (2007) Calorie restriction improves cardiovascular risk factors via reduction of mitochondrial reactive oxygen species in type II diabetic rats. J Pharmacol Exp Ther 320(2):535–543
Mirhashemi F, Kluth O, Scherneck S, Vogel H, Kluge R, Schürmann A, Joost HG, Neschen S (2008) High-fat, carbohydrate-free diet markedly aggravates obesity but prevents beta cell loss and diabetes in the obese, diabetes-susceptible db/db strain. Obes Facts 1(6):292–297. https://doi.org/10.1159/000176064
Mori S, Kawano K, Hirashima T, Natori T (1996) Relationships between diet control and the development of spontaneous type II diabetes and diabetic nephropathy in OLETF rats. Diabetes Res Clin Pract 33(3):145–152. https://doi.org/10.1016/0168-8227(96)01290-9
Nangaku M, Izuhara Y, Usuda N, Inagi R, Shibata T, Sugiyama S, Kurokawa K, de Strihou CV, Miyata T (2005) In a type 2 diabetic nephropathy rat model, the improvement of obesity by a low calorie diet reduces oxidative/carbonyl stress and prevents diabetic nephropathy. Nephrol Dial Transplant 20(12):2661–2669. https://doi.org/10.1093/ndt/gfi096
Ohneda M, Inman LR, Unger RH (1995) Caloric restriction in obese pre-diabetic rats prevents beta-cell depletion, loss of beta-cell GLUT 2 and glucose incompetence. Diabetologia 38(2):173–179. https://doi.org/10.1007/BF00400091
Okauchi N, Mizuno A, Yoshimoto S, Zhu M, Sano T, Shima K (1995) Is caloric restriction effective in preventing diabetes mellitus in the Otsuka Long Evans Tokushima fatty rat, a model of spontaneous non-insulin-dependent diabetes mellitus? Diabetes Res Clin Pract 27(2):97–106. https://doi.org/10.1016/0168-8227(95)01029-D
Otoda T, Kanasaki K, Koya D (2014) Low-protein diet for diabetic nephropathy. Curr Diab Rep 14(9):523. https://doi.org/10.1007/s11892-014-0523-z
Oudot C, Lajoix AD, Jover B, Rugale C (2013) Dietary sodium restriction prevents kidney damage in high fructose-fed rats. Kidney Int 83(4):674–683. https://doi.org/10.1038/ki.2012.478
Poplawski MM, Mastaitis JW, Isoda F, Grosjean F, Zheng F, Mobbs CV (2011) Reversal of diabetic nephropathy by a ketogenic diet. PLoS One 6(4):e18604. https://doi.org/10.1371/journal.pone.0018604
Razzaque MS, Shimokawa I, Nazneen A, Liu D, Naito T, Higami Y, Taguchi T (1999) Life-long dietary restriction modulates the expression of collagens and collagen-binding heat shock protein 47 in aged Fischer 344 rat kidney. Histochem J 31(2):123–132. https://doi.org/10.1023/A:1003578928487
Shapiro H, Theilla M, Attal-Singer J, Singer P (2011) Effects of polyunsaturated fatty acid consumption in diabetic nephropathy. Nat Rev Nephrol 7(2):110–121. https://doi.org/10.1038/nrneph.2010.156
Sheard NF, Clark NG, Brand-Miller JC, Franz MJ, Pi-Sunyer FX, Mayer-Davis E, Kulkarni K, Geil P (2004) Dietary carbohydrate (amount and type) in the prevention and management of diabetes: a statement by the american diabetes association. Diabetes Care 27(9):2266–2271. https://doi.org/10.2337/diacare.27.9.2266
Steinberg GR, Kemp BE (2009) AMPK in health and disease. Physiol Rev 89(3):1025–1078. https://doi.org/10.1152/physrev.00011.2008
Surwit RS, Kuhn CM, Cochrane C, Mc Cubbin JA, Feinglos MN (1988) Diet-induced type II diabetes in C57BL/6J mice. Diabetes 37(9):1163–1167. https://doi.org/10.2337/diab.37.9.1163
Tikoo K, Tripathi DN, Kabra DG, Sharma V, Gaikwad AB (2007) Intermittent fasting prevents the progression of type I diabetic nephropathy in rats and changes the expression of Sir2 and p53. FEBS Lett 581(5):1071–1078. https://doi.org/10.1016/j.febslet.2007.02.006
Tonshoff B, Kaskel FJ, Moore LC (1998) Effects of insulin-like growth factor I on the renal juxtamedullary microvasculature. Am J Phys 274:F120–F128
Tsukahara H, Gordienko DV, Tonshoff B, Gelato MC, Goligorsky MS (1994) Direct demonstration of insulin-like growth factor-I-induced nitric oxide production by endothelial cells. Kidney Int 45(2):598–604. https://doi.org/10.1038/ki.1994.78
Ugochukwu NH, Bagayoko ND, Antwi ME (2004) The effects of dietary caloric restriction on antioxidant status and lipid peroxidation in mild and severe streptozotocin-induced diabetic rats. Clin Chim Acta 348(1):121–129. https://doi.org/10.1016/j.cccn.2004.05.005
Ugochukwu NH, Mukes JD, Figgers CL (2006) Ameliorative effects of dietary caloric restriction on oxidative stress and inflammation in the brain of streptozotocin-induced diabetic rats. Clin Chim Acta 370(1):165–173. https://doi.org/10.1016/j.cca.2006.02.003
Uusitupa M, Siitonen O, Savolainen K, Silvasti M, Penttila I, Parviainen M (1989) Metabolic and nutritional effects of long-term use of guar gum in the treatment of noninsulin-dependent diabetes of poor metabolic control. Am J Clin Nutr 49(2):345–351
Wagner JD, Zhang L, Shadoan MK, Kavanagh K, Chen H, Tresnasari K, Kaplan JR, Adams MR (2008) Effects of soy protein and isoflavones on insulin resistance and adiponectin in male monkeys. Metabolism 57(7 Suppl 1):S24–S31. https://doi.org/10.1016/j.metabol.2008.04.001
Wellen KE, Thompson CB (2010) Cellular metabolic stress: considering how cells respond to nutrient excess. Mol Cell 40(2):323–332. https://doi.org/10.1016/j.molcel.2010.10.004
World Health Organization (2016) Global report on diabetes. Geneva. Available from http://apps.who.int/iris/bitstream/10665/204871/1/9789241565257_eng.pdf?u. Accessed 5 May 2017
Zheng F, He C, Cai W, Hattori M, Steffes M, Vlassara H (2002) Prevention of diabetic nephropathy in mice by a diet low in glycoxidation products. Diabetes Metab Res Rev 18(3):224–237. https://doi.org/10.1002/dmrr.283
Zoncu R, Efeyan A, Sabatini DM (2011) mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol 12(1):21–35. https://doi.org/10.1038/nrm3025
Acknowledgements
We want to acknowledge the Department of Pharmaceutical Sciences and Drug Research, Punjabi University, Patiala and UGC, New Delhi for extending the necessary research facilities for our work. We also want to acknowledge Elegant Foreign Language Institute (EFLI) IELTS Institute Sunam, Punjab, India for English writing and editing.
Funding
The authors are thankful to DST-SERB, New Delhi for providing financial assistance under project no. EMR/2014/000438 to accomplish this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Chemical compounds studied in this article
Streptozotocin (PubChem CID:29327)
Highlights
• Diet restriction has beneficial effects on insulin action and glycemic control in obesity and mild T2DM
• Soy diet shown to produce anti-diabetic effect by acting through the estrogen receptors.
• Preclinical evidences of diet restriction and dietary approaches in the management of diabetes and their associated complications.
• Other highlight of this review is about nutritional interventions which combat progressive diabetes and the associated disorders.
Rights and permissions
About this article
Cite this article
Krishan, P., Bedi, O. & Rani, M. Impact of diet restriction in the management of diabetes: evidences from preclinical studies. Naunyn-Schmiedeberg's Arch Pharmacol 391, 235–245 (2018). https://doi.org/10.1007/s00210-017-1453-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-017-1453-5