Abstract
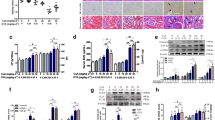
Ochratoxin A (OTA) is reported to induce nephrotoxicity and immunotoxicity in animals and humans. However, the underlying mechanism and the effects of OTA on DNA damage have not been reported until now. The present study aims to investigate OTA-induced cytotoxicity and DNA damage and the underlying mechanism in PK15 cells and PAMs. The results showed that OTA at 2.0–8.0 µg/mL for 24 h induced cytotoxicity and DNA damage in PK15 cells and PAMs as demonstrated by decreasing cell viabilities and mRNA levels of DNA repair genes (OGG1, NEIL1 and NEIL3), increasing LDH release, Annexin V staining cells, apoptotic nuclei and the accumulation of γ-H2AX foci. OTA at 2.0–8.0 µg/mL increased DNMT1 and SOCS3 mRNA expressions about 2–4 fold in PK15 cells or 1.3–2 fold in PAMs. OTA at 2.0–8.0 µg/mL increased DNMT1, SOCS3, JAK2 and STAT3 protein expressions in PK15 cells or PAMs. DNMT inhibitor (5-Aza-2-dc), promoted SOCS3 expression, inhibited JAK2 and STAT3 expression, alleviated cytotoxicity, apoptosis and DNA damage induced by OTA at 4.0 µg/mL in PK15 cells. While, in PAMs, 5-Aza-2-dc had no effects on SOCS3 expression induced by OTA at 4.0 µg/mL, but inhibited JAK2 and STAT3 expression, and alleviated cytotoxicity, apoptosis and DNA damage induced by OTA. JAK inhibitor (AG490) or STAT3-siRNA alleviated OTA-induced cytotoxicity and DNA damage in PK15 cells or PAMs. Taken together, nephrotoxicity instead of immunotoxicity of OTA is induced by targeting SOCS3 through DNMT1-mediated JAK2/STAT3 signaling pathway. These results provide a scientific and new explanation of the underlying mechanism of OTA-induced nephrotoxicity and immunotoxicity.











Similar content being viewed by others
References
Barnes JL, Zubair M, John K, Poirier MC, Martin FL (2018) Carcinogens and DNA damage. Biochem Soc Trans 46(5):1213–1224. https://doi.org/10.1042/BST20180519
Bestor TH (2000) The DNA methyltransferases of mammals. Hum Mol Genet 9(16):2395–2402
Bohrer RC, Dicks N, Gutierrez K, Duggavathi R, Bordignon V (2018) Double-strand DNA breaks are mainly repaired by the homologous recombination pathway in early developing swine embryos. FASEB J 32(4):1818–1829. https://doi.org/10.1096/fj.201700800R
Boosani CS, Dhar K, Agrawal DK (2015) Down-regulation of hsa-miR-1264 contributes to DNMT1-mediated silencing of SOCS3. Mol Biol Rep 42(9):1365–1376. https://doi.org/10.1007/s11033-015-3882-x
Chuturgoon A, Phulukdaree A, Moodley D (2014) Fumonisin B1 induces global DNA hypomethylation in HepG2 cells—an alternative mechanism of action. Toxicology 315:65–69. https://doi.org/10.1016/j.tox.2013.11.004
Ciarcia R, Damiano S, Squillacioti C et al (2016) Recombinant mitochondrial manganese containing superoxide dismutase protects against ochratoxin A-induced nephrotoxicity. J Cell Biochem 117(6):1352–1358. https://doi.org/10.1002/jcb.25425
Damiano S, Puzio MV, Squillacioti C et al (2018) Effect of rMnSOD on sodium reabsorption in renal proximal tubule in ochratoxin A-treated rats. J Cell Biochem 119(1):424–430. https://doi.org/10.1002/jcb.26197
Delgado-Ortega M, Melo S, Meurens F (2011) Expression of SOCS1-7 and CIS mRNA in porcine tissues. Vet Immunol Immunopathol 144(3–4):493–498. https://doi.org/10.1016/j.vetimm.2011.08.002
Dhar K, Rakesh K, Pankajakshan D, Agrawal DK (2013) SOCS3 promotor hypermethylation and STAT3-NF-kappaB interaction downregulate SOCS3 expression in human coronary artery smooth muscle cells. Am J Physiol Heart Circ Physiol 304(6):H776–H785. https://doi.org/10.1152/ajpheart.00570.2012
Fu RJ, He W, Wang XB et al (2017) DNMT1-maintained hypermethylation of Kruppel-like factor 5 involves in the progression of clear cell renal cell carcinoma. Cell Death Dis 8(7):e2952. https://doi.org/10.1038/cddis.2017.323
Gan F, Ren F, Chen X et al (2013) Effects of selenium-enriched probiotics on heat shock protein mRNA levels in piglet under heat stress conditions. J Agric Food Chem 61(10):2385–2391. https://doi.org/10.1021/jf300249j
Gan F, Xue H, Huang Y, Pan C, Huang K (2015a) Selenium alleviates porcine nephrotoxicity of ochratoxin a by improving selenoenzyme expression in vitro. PLoS One 10(3):e0119808. https://doi.org/10.1371/journal.pone.0119808
Gan F, Zhang Z, Hu Z et al (2015b) Ochratoxin A promotes porcine circovirus type 2 replication in vitro and in vivo. Free Radic Biol Med 80:33–47. https://doi.org/10.1016/j.freeradbiomed.2014.12.016
Gan F, Hou L, Zhou Y et al (2017a) Effects of ochratoxin A on ER stress, MAPK signaling pathway and autophagy of kidney and spleen in pigs. Environ Toxicol 32(10):2277–2286. https://doi.org/10.1002/tox.22443
Gan F, Zhou Y, Hou L, Qian G, Chen X, Huang K (2017b) Ochratoxin A induces nephrotoxicity and immunotoxicity through different MAPK signaling pathways in PK15 cells and porcine primary splenocytes. Chemosphere 182:630–637. https://doi.org/10.1016/j.chemosphere.2017.05.030
Gan F, Zhou Y, Qian G et al (2018) PCV2 infection aggravates ochratoxin A-induced nephrotoxicity via autophagy involving p38 signaling pathway in vivo and in vitro. Environ Pollut 238:656–662. https://doi.org/10.1016/j.envpol.2018.03.032
Hamilton JP (2011) Epigenetics: principles and practice. Dig Dis 29(2):130–135. https://doi.org/10.1159/000323874
Han J, Wang QC, Zhu CC et al (2016) Deoxynivalenol exposure induces autophagy/apoptosis and epigenetic modification changes during porcine oocyte maturation. Toxicol Appl Pharmacol 300:70–76. https://doi.org/10.1016/j.taap.2016.03.006
Hennemeier I, Humpf HU, Gekle M, Schwerdt G (2012) The food contaminant and nephrotoxin ochratoxin A enhances Wnt1 inducible signaling protein 1 and tumor necrosis factor-α expression in human primary proximal tubule cells. Mol Nutr Food Res 56(9):1375–1384
Huang L, Hu B, Ni J et al (2016) Transcriptional repression of SOCS3 mediated by IL-6/STAT3 signaling via DNMT1 promotes pancreatic cancer growth and metastasis. J Exp Clin Cancer Res 35:27. https://doi.org/10.1186/s13046-016-0301-7
Huang S, Zhou D, Li YX et al (2019) In vivo and in vitro effects of microRNA-221 on hepatocellular carcinoma development and progression through the JAK-STAT3 signaling pathway by targeting SOCS3. J Cell Physiol 234(4):3500–3514. https://doi.org/10.1002/jcp.26863
Ito F, Yamada Y, Shigemitsu A et al (2017) Role of oxidative stress in epigenetic modification in endometriosis. Reprod Sci. https://doi.org/10.1177/1933719117704909
Ji P (1987) Penicillium viridicatum, Penicillium verrucosum, and production of ochratoxin A. Appl Environ Microbiol 53:266–269
Kandhaya-Pillai R, Miro-Mur F, Alijotas-Reig J, Tchkonia T, Kirkland JL, Schwartz S (2017) TNFalpha-senescence initiates a STAT-dependent positive feedback loop, leading to a sustained interferon signature, DNA damage, and cytokine secretion. Aging 9(11):2411–2435. https://doi.org/10.18632/aging.101328
Kimura A, Ishida Y, Nosaka M et al (2016) Exaggerated arsenic nephrotoxicity in female mice through estrogen-dependent impairments in the autophagic flux. Toxicology 339:9–18. https://doi.org/10.1016/j.tox.2015.11.005
Klarić M, Rumora L, Ljubanović D, Pepeljnjak S (2008) Cytotoxicity and apoptosis induced by fumonisin B1, beauvericin and ochratoxin A in porcine kidney PK15 cells: effects of individual and combined treatment. Arch Toxicol 82(4):247–255
Lan J, Hua S, He X, Zhang Y (2010) DNA methyltransferases and methyl-binding proteins of mammals. Acta biochimica et biophysica Sinica 42(4):243–252
Lee HJ, Pyo MC, Shin HS, Ryu D, Lee KW (2018) Renal toxicity through AhR, PXR, and Nrf2 signaling pathway activation of ochratoxin A-induced oxidative stress in kidney cells. Food Chem Toxicol 122:59–68. https://doi.org/10.1016/j.fct.2018.10.004
Lim CW, Yoshinari T, Layne J, Chan SH (2015) Multi-mycotoxin screening reveals separate occurrence of aflatoxins and ochratoxin a in Asian rice. J Agric Food Chem 63(12):3104–3113. https://doi.org/10.1021/acs.jafc.5b00471
Lin YC, Owen N, Minko IG et al (2016) DNA polymerase zeta limits chromosomal damage and promotes cell survival following aflatoxin exposure. Proc Natl Acad Sci USA 113(48):13774–13779. https://doi.org/10.1073/pnas.1609024113
Liu D, Lin J, Su J, Chen X, Jiang P, Huang K (2018) Glutamine deficiency promotes PCV2 infection through induction of autophagy via activation of ROS-mediated JAK2/STAT3 signaling pathway. J Agric Food Chem 66(44):11757–11766. https://doi.org/10.1021/acs.jafc.8b04704
Mahalingaiah PK, Ponnusamy L, Singh KP (2017) Oxidative stress-induced epigenetic changes associated with malignant transformation of human kidney epithelial cells. Oncotarget 8(7):11127–11143. https://doi.org/10.18632/oncotarget.12091
Mai HN, Nguyen LTT, Shin EJ et al (2018) Astrocytic mobilization of glutathione peroxidase-1 contributes to the protective potential against cocaine kindling behaviors in mice via activation of JAK2/STAT3 signaling. Free Radic Biol Med 131:408–431. https://doi.org/10.1016/j.freeradbiomed.2018.12.027
Manderville RA, Wetmore SD (2017) Mutagenicity of Ochratoxin A: role for a carbon-linked C8-deoxyguanosine adduct? J Agric Food Chem 65(33):7097–7105. https://doi.org/10.1021/acs.jafc.6b03897
Mann JR (2014) Epigenetics and memigenetics. Cell Mol Life Sci 71(7):1117–1122. https://doi.org/10.1007/s00018-014-1560-0
Marquardt A, Al-Dabet MM, Ghosh S et al (2017) Farnesoid X receptor agonism protects against diabetic tubulopathy: potential add-on therapy for diabetic nephropathy. J Am Soc Nephrol 28(11):3182–3189. https://doi.org/10.1681/ASN.2016101123
Martins HM, Almeida I, Camacho C, Costa JM, Bernardo F (2012) A survey on the occurrence of ochratoxin A in feeds for swine and laying hens. Mycotoxin Res 28(2):107–110
Meerpoel C, Vidal A, di Mavungu JD et al (2018) Development and validation of an LC-MS/MS method for the simultaneous determination of citrinin and ochratoxin a in a variety of feed and foodstuffs. J Chromatogr A 1580:100–109. https://doi.org/10.1016/j.chroma.2018.10.039
Neuwirt H, Eder IE, Puhr M, Rudnicki M (2013) SOCS-3 is downregulated in progressive CKD patients and regulates proliferation in human renal proximal tubule cells in a STAT1/3 independent manner. Lab Investig J Tech Methods Pathol 93(1):123–134. https://doi.org/10.1038/labinvest.2012.154
Nozaki Y, Kinoshita K, Yano T et al (2012) Signaling through the interleukin-18 receptor alpha attenuates inflammation in cisplatin-induced acute kidney injury. Kidney Int 82(8):892–902. https://doi.org/10.1038/ki.2012.226
Palabiyik SS, Erkekoglu P, Zeybek ND et al (2013) Protective effect of lycopene against ochratoxin A induced renal oxidative stress and apoptosis in rats. Exp Toxicol Pathol 65(6):853–861. https://doi.org/10.1016/j.etp.2012.12.004
Raghubeer S, Nagiah S, Chuturgoon AA (2017) Acute Ochratoxin A exposure induces inflammation and apoptosis in human embryonic kidney (HEK293) cells. Toxicon 137:48–53. https://doi.org/10.1016/j.toxicon.2017.07.013
Stoev SD, Gundasheva D, Zarkov I et al (2012) Experimental mycotoxic nephropathy in pigs provoked by a mouldy diet containing ochratoxin A and fumonisin B1. Exp Toxicol Pathol 64(7):733–741
Tikoo K, Ali IY, Gupta J, Gupta C (2009) 5-Azacytidine prevents cisplatin induced nephrotoxicity and potentiates anticancer activity of cisplatin by involving inhibition of metallothionein, pAKT and DNMT1 expression in chemical induced cancer rats. Toxicol Lett 191(2–3):158–166. https://doi.org/10.1016/j.toxlet.2009.08.018
Tsogbadrakh B, Ryu H, Ju KD et al (2019) AICAR, an AMPK activator, protects against cisplatin-induced acute kidney injury through the JAK/STAT/SOCS pathway. Biochem Biophys Res Commun. https://doi.org/10.1016/j.bbrc.2018.12.159
van der Merwe KJ, Steyn PS, Fourie L, Scott DB, Theron JJ (1965) Ochratoxin A, a toxic metabolite produced by Aspergillus ochraceus Wilh. Nature 205(976):1112–1113
Verma M, Chattopadhyay BD, Kumar S, Kumar K, Verma D (2014) DNA methyltransferase 1(DNMT1) induced the expression of suppressors of cytokine signaling3 (Socs3) in a mouse model of asthma. Mol Biol Rep 41(7):4413–4424. https://doi.org/10.1007/s11033-014-3312-5
Vermes I, Haanen C, Reutelingsperger C (2000) Flow cytometry of apoptotic cell death. J Immunol Methods 243(1–2):167–190
Wang M, Zhao J, Zhang N, Chen J (2016) Astilbin improves potassium oxonate-induced hyperuricemia and kidney injury through regulating oxidative stress and inflammation response in mice. Biomed Pharmacother 83:975–988. https://doi.org/10.1016/j.biopha.2016.07.025
Wormald S, Hilton DJ (2004) Inhibitors of cytokine signal transduction. J Biol Chem 279(2):821–824. https://doi.org/10.1074/jbc.R300030200
Xu H, Hao S, Gan F et al (2017) In vitro immune toxicity of ochratoxin A in porcine alveolar macrophages: a role for the ROS-relative TLR4/MyD88 signaling pathway. Chem Biol Interact 272:107–116. https://doi.org/10.1016/j.cbi.2017.05.016
Yen YH, Farooqi AA, Li KT et al (2014) Methanolic extracts of solieria robusta inhibits proliferation of oral cancer Ca9-22 cells via apoptosis and oxidative stress. Molecules 19(11):18721–18732. https://doi.org/10.3390/molecules191118721
Zain ME (2011) Impact of mycotoxins on humans and animals. J Saudi Chem Soc 15(2):129–144. https://doi.org/10.1016/j.jscs.2010.06.006
Zhou Y, Gan F, Hou L, Zhou X, Adam Ibrahim YA, Huang K (2017) Modulations of DNMT1 and HDAC1 are involved in the OTA-induced cytotoxicity and apoptosis in vitro. Chem Biol Interact 278:170–178. https://doi.org/10.1016/j.cbi.2017.10.020
Zhu CC, Hou YJ, Han J, Cui XS, Kim NH, Sun SC (2014) Zearalenone exposure affects epigenetic modifications of mouse eggs. Mutagenesis 29(6):489–495. https://doi.org/10.1093/mutage/geu033
Acknowledgements
This study was funded by the Natural Science Foundation of Jiangsu Province (BK20160736), National Natural Science Foundation of China (31602123, 31772811), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (Jiangsu, China).
Author information
Authors and Affiliations
Contributions
Study concept and design: KH. Acquisition of data: FG, XZ, ZJ, LH, KH. Analysis and interpretation of data: FG, XZ, ZJ, XC, CP. Drafting of the manuscript: FG, KH. Critical revision of the manuscript for important intellectual content: KH, FG. Statistical analysis: FG, KH. Obtained funding: KH and FG. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We do not have any commercial associations that might create a conflict of interest in connection with this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gan, F., Zhou, X., Zhou, Y. et al. Nephrotoxicity instead of immunotoxicity of OTA is induced through DNMT1-dependent activation of JAK2/STAT3 signaling pathway by targeting SOCS3. Arch Toxicol 93, 1067–1082 (2019). https://doi.org/10.1007/s00204-019-02434-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02434-5