Abstract
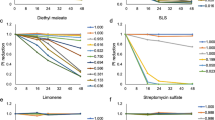
We established a luciferase reporter assay system, the Multi-ImmunoTox Assay (MITA), which can evaluate the effects of chemicals on the promoter activities of four cytokines: IL-2, IFN-γ, IL-1β, and IL-8. We previously reported that MITA correctly reflected the change in mRNA of human whole-blood cells treated with dexamethasone, cyclosporine, FK506, or several other immunosuppressive drugs. In this study, we combined MITA with the IL-8 Luc assay to detect skin sensitization chemicals (OECD 442E) (modified MITA: mMITA) and established a data set of 60 chemicals examined by mMITA. Using the mMITA results, chemicals can be classified based on the lowest observed effect level (LOEL) of chemicals in suppressing or augmenting the promoter activities of the four cytokines. Moreover, we demonstrated that K-means clustering and hierarchical clustering of the 60 chemicals based on the LOEL for their effects on IL-2 and IL-8 promoter activities and the judgment by the IL-8 Luc assay resulted in the same 6-cluster solution: cluster 1 with preferential suppression of IL-8, cluster 2 with suppression of IL-2 and a positive IL-8 Luc assay result, cluster 3 with suppression of both IL-2 and IL-8, cluster 4 with no effects on IL-2 or IL-8 and a negative IL-8 Luc assay result, cluster 5 with suppression of both IL-2 and IL-8 and a negative IL-8 Luc assay result, and cluster 6 with preferential suppression of IL-8. These data suggest that mMITA is a promising novel high-throughput approach for detecting unrecognized immunological effects of chemicals and for profiling their immunotoxic effects.




Similar content being viewed by others
References
World Health Organization (WHO)(2012) Guidance for immunotoxicity risk assessment for chemicals. WHO press, Geneva, Switzerland
Carfi M, Gennari A, Malerba I et al (2007) In vitro tests to evaluate immunotoxicity: a preliminary study. Toxicology 229(1–2):11–22. https://doi.org/10.1016/j.tox.2006.09.003
Furst DE, Clements PJ (2008) Immunosuppressives. In: Hochberg MC, Silman AJ, Smolen JS, Weinblatt ME, Weisman MH (eds) Rheumatology (Oxford), vol 1, 4th edn. Mosby Elsevier, Philadelphia, pp 471–480
Galbiati V, Mitjans M, Corsini E (2010) Present and future of in vitro immunotoxicology in drug development. J Immunotoxicol 7(4): 255–267. https://doi.org/10.3109/1547691X.2010.509848
Gennari A, Ban M, Braun A et al (2005) The use of in vitro systems for evaluating immunotoxicity: the report and recommendations of an ECVAM workshop. J Immunotoxicol 2(2):61–83. https://doi.org/10.1080/15476910590965832
Hartung T (2002) Comparison and validation of novel pyrogen tests based on the human fever reaction. Altern Lab Anim ATLA 30(Suppl 2):49–51
Kakeda M, Hiratsuka M, Nagata K et al (2005) Human artificial chromosome (HAC) vector provides long-term therapeutic transgene expression in normal human primary fibroblasts. Gene Ther 12(10):852–856. https://doi.org/10.1038/sj.gt.3302483
Kimura Y, Fujimura C, Ito Y et al (2014) Evaluation of the Multi-ImmunoTox Assay composed of 3 human cytokine reporter cells by examining immunological effects of drugs. Toxicol In Vitro 28(5):759–768. https://doi.org/10.1016/j.tiv.2014.02.013
Kimura Y, Fujimura C, Ito Y et al (2015) Optimization of the IL-8 Luc assay as an in vitro test for skin sensitization. Toxicol In Vitro 29(7):1816–1830. https://doi.org/10.1016/j.tiv.2015.07.006
Langezaal I, Hoffmann S, Hartung T et al (2002) Evaluation and prevalidation of an immunotoxicity test based on human whole-blood cytokine release. Altern Lab Anim ATLA 30(6):581–595
Lankveld DP, Van Loveren H, Baken KA et al (2010) In vitro testing for direct immunotoxicity: state of the art. Methods Mol Biol 598:401–423. https://doi.org/10.1007/978-1-60761-401-2_26
OECD (2017) OECD Test Gudeline for the Testing of Chemicals No.442E: In vitro skin sensitisation assays addressing the key event on activation of dendritic cells on the adverse outcome pathway for skin sensitisation. http://www.oecd-ilibrary.org/environment/oecd-guidelines-for-the-testing-of-chemicals-section-4-health-effects_20745788. Accessed 9 Oct 2017
Saag K (2008) Systemic glucocorticoids in rheumatology. In: Hochberg MC, Silman AJ, Smolen JS, Weinblatt ME, Weisman MH (eds) Rheumtology. vol 1, 4th edn. Mosby Elsevier, Philadelphia, pp 411–419
Saito R, Hirakawa S, Ohara H et al(2011) Nickel differentially regulates NFAT and NF-kappaB activation in T cell signaling. Toxicol Appl Pharmacol 254(3):245–255. https://doi.org/10.1016/j.taap.2011.04.017
Sarle W (1983) Cubic Clustering Criterion, SAS Technical Report A-108 Cary, NC: SAS Institute Inc
Takahashi T, Kimura Y, Saito R et al (2011) An in vitro test to screen skin sensitizers using a stable THP-1-derived IL-8 reporter cell line, THP-G8. Toxicol Sci 124(2):359–369. https://doi.org/10.1093/toxsci/kfr237
Wagner W, Walczak-Drzewiecka A et al (2006) Fluorescent cell chip a new in vitro approach for immunotoxicity screening. Toxicol Lett 162(1):55–70. https://doi.org/10.1016/j.toxlet.2005.10.017
Acknowledgements
This work was supported in part by the New Energy and Industrial Development Organization (NEDO), by a Grant-in-Aid for Challenging Exploratory Research from the Japan Society for the Promotion of Science (KAKENHI Grant no. 26670473), by Health Labor Sciences Research in Japan, by the Ministry of Economy, Science and Industry in Japan, and by the Japanese Society for Alternatives to Animal Experiments. We are grateful to Mika Watanabe, Miwako Kobayashi, and Kohji Yamakage, PhD (Hatano Research Institute, Food and Drug Safety Center, Hadano, Japan), Rie Yasuno, PhD and Yoshihiro Ohmiya, MD, PhD (Biomedical Research Institute, Advanced Industrial Science and Technology, Tsukuba, Japan), Tomoko Iwaki and Yoshihiro Nakajima, PhD (Health Research Institute, Advanced Industrial Science and technology, Takamatsu, Japan), Mayumi Kobayashi and Takashi Omori, PhD (Division of Biostatistics, Department of Social/Community Medicine and Health Science, Kobe University School of Medicine, Japan), and Hajime Kojima (Japanese Center for the Validation of Alternative Methods, Tokyo, Japan) for their kind advise in developing MITA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kimura, Y., Fujimura, C., Ito, Y. et al. Profiling the immunotoxicity of chemicals based on in vitro evaluation by a combination of the Multi-ImmunoTox assay and the IL-8 Luc assay. Arch Toxicol 92, 2043–2054 (2018). https://doi.org/10.1007/s00204-018-2199-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-018-2199-7